Abstract
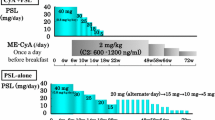
Cyclosporin A (CsA) is an effective therapy for children with long-lasting nephrotic syndrome (NS). Long-term treatment can result in chronic CsA nephropathy (CsAN) and there is controversy concerning its incidence and severity. Trough levels are commonly used to monitor the drug concentration. We report a retrospective clinical and histological analysis of 18 children (12 males, 6 females) with steroid-dependent nephrotic syndrome (15 patients) and partially steroid-sensitive nephrotic syndrome (3 patients) treated with CsA for a long-term period (mean 4.9 years, range 2.2–6.9). Before CsA treatment all patients had normal creatinine clearance. CsA was started at a dose of 5 mg/kg per day administered orally in two divided doses and adjusted to maintain the mean CsA blood concentration between 250 and 350 ng/ml obtained from abbreviated area under the curve (AUC). A renal biopsy was performed after a mean period of 3.9 years (range 2.2–6.2) from the start of CsA treatment. Tubular, interstitial, and arteriolar lesions were evaluated in order to assess CsAN. The mean CsA dose and the mean CsA blood concentration were 4.4 mg/kg per day (range 3.6–5.8) and 276.6 ng/ml (range 162–346), respectively. No child had a worsening creatinine clearance during CsA treatment and follow-up after CsA discontinuation. If compared with the year before the start of CsA treatment, NS relapses and prednisone (PDN) dose significantly decreased during CsA treatment, 4/year versus 0.8/year (P <0.0001) and 0.9 mg/kg per day versus 0.2 mg/kg per day (P <0.0001), respectively. Histological analysis showed 15 patients with minimal change disease and 3 with focal segmental glomerulosclerosis. Clear-cut lesions diagnostic of CsAN were never found and only mild lesions were observed in 5 children (suggestive of CsAN in 2 patients and consistent with CsAN in 3 patients). Long-term CsA treatment is confirmed to be effective in preventing NS relapses and reducing PDN dose. Renal function is not a reliable indicator of CsAN. With the mean CsA blood concentrations used in our patients CsAN presented a low incidence (28%) and was generally mild. Renal biopsy should be performed 2–3 years from the start of long-term CsA treatment, especially if the mean CsA blood concentrations are not regularly monitored.


Similar content being viewed by others
References
Kahan BD (1989) Cyclosporine. N Engl J Med 321:1725–1738
Tejani A (1986) Cyclosporin-induced remission of relapsing nephrotic syndrome in childhood (abstract). Kidney Int 29:206
Clasen W, Kindler J, Mihatsch MJ, Sieberth HG (1988) Long-term treatment of minimal change nephrotic syndrome with cyclosporin: a control biopsy study. Nephrol Dial Transplant 3:733–737
Neahaus TJ, Burger HR, Klingler M, Fanconi A, Leumann EP (1992) Long-term low-dose cyclosporin A in steroid dependent nephrotic syndrome of childhood. Eur J Pediatr 151:775–778
Ponticelli C, Edefonti A, Rizzoni G, Rinaldi S, Gusmano R, Lama G, Zacchello G, Confalonieri R, Altieri P, Bettinelli A, Maschio G, Cinotti GA, Fuiano G, Schena FP, Castellani A, Della Casa-Alberighi O (1993) Cyclosporin versus cyclophosphamide for patients with steroid-dependent and frequently relapsing idiopathic nephrotic syndrome: a multicenter randomized controlled trial. Nephrol Dial Transplant 8:1326–1332
Meyrier A (1995) Use of cyclosporin in the treatment of idiopathic nephrotic sindrome in adults. Contrib Nephrol 114:28–48
Gregory MJ, Smoyer WE, Sedman A, Kershaw DB, Valentini RP, Johnson K, Bunchman T (1996) Long-term cyclosporine therapy for pediatric nephrotic syndrome: a clinical and histologic analysis. J Am Soc Nephrol 7:543–549
Hino S, Takemura T, Okada M, Murakami K, Yagi K, Fukushima K, Yoshioka K (1998) Follow-up study of children with nephrotic syndrome treated with a long-term moderate dose of cyclosporine. Am J Kidney Dis 31:932–939
Singh A, Tejani C, Tejani A (1999) One-center experience with cyclosporine in refractory nephrotic sindrome in children. Pediatr Nephrol 13:26–32
Inoue Y, Iijima K, Nakamura H, Yoshikawa N (1999) Two-year cyclosporin treatment in children with steroid-dependent nephrotic syndrome. Pediatr Nephrol 13:33–38
Yoshikawa N, Iijima K, and Ito H (1999) Cyclosporin (Cyclosporine) treatment in children with steroid-dependent nephrotic syndrome. Clin Exp Nephrol 3 [Suppl]:S27–S33
Seikaly MG, Prashner H, Nolde-Hurlbert B, Browne R (2000) Long-term clinical and pathological effect of cyclosporin in children with nephrosis. Pediatr Nephrol 14:214–217
Ganesan V, Milford DV, Taylor CM, Hulton SA, Parvaresh S, Ramani P (2002) Cyclosporin-related nephrotoxicity in children with nephrotic syndrome. Pediatr Nephrol 17:225–226
Ponticelli C, Rizzoni G, Edefonti A, Altieri P, Rivolta E, Rinaldi S, Ghio L, Lusvarghi E, Gusmano R, Locatelli F, Pasquali S, Castellani A, Della Casa-Alberighi O (1993) A randomized trial of cyclosporine in steroid-resistant idiopathic nephrotic syndrome. Kidney Int 43:1377–1384
Lieberman KV, Tejani A (1996) A randomized double-blind, placebo-controlled trial of cyclosporine in steroid-resistant idiopathic focal segmental glomerulosclerosis in children. J Am Soc Nephrol 7:56–63
Okada M, Hino S, Takemura T, Fukushima K, Yoshioka K (1999) Cyclosporine therapy in children with steroid-resistant nephrotic syndrome. Clin Exp Nephrol 3:34–39
International Study of Kidney Disease in Children (1982) Early identification of frequent relapsers among children with minimal change nephrotic syndrome. J Pediatr 101:514–518
Amante AJ, Kahan BD (1996) Abbreviated area under the curve strategy for monitoring cyclosporin microemulsion therapy in immediate posttransplant period. Clin Chem 42:1294–1296
Schwartz GJ, Brion LP, Spitzer A (1987) The use of plasma creatinine concentration for estimating glomerular filtration rate in infants, children, and adolescents. Pediatr Clin North Am 34:571–586
Habib R, Niaudet P (1994) Comparison between pre and post treatment renal biopsies in children receiving cyclosporine for idiopathic nephrosis. Clin Nephrol 42:1446–1456
Mihatsch MJ, Antonovych T, Bohman SO, Habib R, Helmchen U, Noel LH, Sibley RK, Kemeny E, Feutren G (1994) Cyclosporine A nephropathy: standardization of the evaluation of kidney biopsies. Clin Nephrol 41:23–32
Iijima K, Hamahira K, Kobayashi A (2000) Immunohistochemical analysis of renin activity in chronic cyclosporine nephropathy in childhood nephrotic syndrome. J Am Soc Nephrol 11:2265–2271
Hamahira K, Iijima K, Tanaka R (2001) Recovery from cyclosporine associated arteriolopathy in childhood nephrotic syndrome. Pediatr Nephrol 16:723–727
Iijima K, Hamahira K, Tanaka R, Kobayashi A, Nozu K, Nakamura H, Yoshikawa N (2002) Risk factors for cyclosporine-induced tubulointerstitial lesions in children with minimal change nephrotic syndrome. Kidney Int 61:1801–1805
Levy GA (2001) C2 monitoring strategy for optimizing cyclosporin immunosuppression from the Neoral formulation. BioDrugs 15:279–290
Dello Strologo L, Pontesilli C, Rizzoni G, Tozzi AE (2003) C2 monitoring: a reliable tool in pediatric renal transplants recipients. Transplantation 76:444–445
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rinaldi, S., Sesto, A., Barsotti, P. et al. Cyclosporine therapy monitored with abbreviated area under curve in nephrotic syndrome. Pediatr Nephrol 20, 25–29 (2005). https://doi.org/10.1007/s00467-004-1618-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-004-1618-6