Abstract
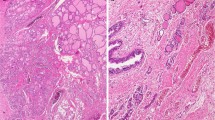
It has been a subject of much debate whether thyroid follicular cells originate from the ultimobranchial body, in addition to median thyroid primordium. Ultimobranchial remnants are detected in normal dogs, rats, mice, cattle, bison and humans and also in mutant mice such as Eya1 homozygotes, Hox3 paralogs homozygotes, Nkx2.1 heterozygotes and FRS2α2F/2F. Besides C cells, follicular cell lineages immunoreactive for thyroglobulin are located within these ultimobranchial remnants. In dogs, the C cell complexes, i.e., large cell clusters consisting of C cells and undifferentiated cells, are present together with parathyroid IV and thymus IV in or close to the thyroid lobe. In addition, follicular cells in various stages of differentiation, including follicular cell groups and primitive and minute follicles storing colloid, are intermingled with C cells in some complexes. This review elaborates the transcription factors and signaling molecules involved in folliculogenesis and it is supposed why the follicular cells in the ultimobranchial remnants are sustained in immature stages. Pax8, a transcription factor crucial for the development of follicular cells, is expressed in the fourth pharyngeal pouch and the ultimobranchial body in human embryos. Pax8 expression is also detected in the ultimobranchial remnants of Eya1 and Hes1 null mutant mice. To determine whether the C cells and follicular cells in the ultimobranchial remnants consist of dual lineage cells or are derived from the common precursor, the changes of undifferentiated cells in dog C cell complexes are examined after chronically induced hypercalcemia or antithyroid drug treatment.






Similar content being viewed by others
References
Andersson L, Westerlund J, Liang S, Carlsson T, Amendola E, Fagman H, Nilsson M (2011) Role of EphA4 receptor signaling in thyroid development: regulation of folliculogenesis and propagation of the C-cell lineage. Endocrinology 152:1154–1164
Antonica F, Kasprzyk DF, Opitz R, Lacovino M, Liao XH, Dumitrescu AM, Refetoff S, Peremans K, Manto M, Kyba M, Costagliola S (2012) Generation of functional thyroid from embryonic stem cells. Nature 491:66–71
Autelitano F, Santeusanio G, Tondo UD, Costantino M, Renda F, Autelitano M (1987) Immunohistochemical study of solid cell nests of the thyroid gland found from an autopsy study. Cancer 59:477–483
Bignami A, Raju T, Dahl D (1982) Localization of vimentin, the nonspecific intermediate filament protein, in embryonal glia and in early differentiation neurons: in vivo and in vitro immunofluorescence study of the rat embryo with vimentin and neurofilament antisera. Dev Biol 91:286–295
Burstein DE, Nagi C, Wang BY, Unger P (2004) Immunohistochemical detection of p53 homolog p63 in solid cell nests, papillary thyroid carcinoma, and Hashimoto’s thyroiditis: a stem cell hypothesis of papillary carcinoma oncogenesis. Hum Pathol 35:465–473
Calvert R (1972) Electron microscopic observations on the contribution of the ultimobranchial bodies to thyroid histogenesis in the rat. Am J Anat 133:269–290
Cameselle-Teijeiro J, Varela-Durán J, Sambade C, Villanueva JP, Varela-Núnez R, Sobriho-Simoes M (1994) Solid cell nests of the thyroid: light microscopy and immunohisochemical profile. Hum Pathol 25:684–693
Carre A, Rachdi L, Tron E, Richard B, Castanet M et al (2011) Hes1 is required for appropriate morphogenesis and differentiation during mouse thyroid gland development. PLoS One 6:e16752
Castro-Munozledo F, Meza-Aguilar DG, Dominguez-Castillo R, Hernández-Zequinely V, Sánchez-Guzimán E (2017) Vimentin as a marker of early differentiating, highly motile corneal epithelial cells. J Cell Physiol 232:818–830
Chisaka O, Kameda Y (2005) Hoxa3 regulates the proliferation and differentiation of the third pharyngeal arch mesenchyme in mice. Cell Tissue Res 320:77–89
Colin IM, Denef JF, Lengelé B, Many MC, Gérard AC (2013) Recent insights into the cell biology of thyroid angiofollicular units. Endocr Rev 34:209–238
Conde E, Moreno AM, Martin-Lacave I, Galera AFH (1992) Immunocytochemical study of the ultimobranchial tubule in Wistar rats. Anat Histol Embryol 21:94–100
De Felice M, Di Lauro R (2004) Thyroid development and its disorders: genetics and molecular mechanisms. Endocr Rev 25:722–746
De Felice M, Di Lauro R (2011) Minireview: intrinsic and extrinsic factors in thyroid gland development: an update. Endocrinology 152:2948–2956
Fagman H, Grӓnde M, Gritli-Linde A, Nilsson M (2004) Genetic deletion of Sonic hedgehog causes hemiagenesis and ectopic development of the thyroid in mouse. Am J Pathol 164:1865–1872
Fagman H, Andersson L, Nilsson M (2006) The developing mouse thyroid: embryonic vessel contacts and parenchymal growth pattern during specification, budding, migration, and lobulation. Dev Dyn 235:444–455
Frezzetti D, Reale C, Cali G, Nitsch L, Fagman H, Nilsson O, Scarfò M, De Vita G, Di Lauro R (2011) The microRNA-processing enzyme Dicer is essential for thyroid function. PLoS One 6:e27648
Greenburg G, Hay ED (1988) Cytoskeleton and thyroglobulin expression change during transformation of thyroid epithelium to mesenchyme-like cells. Development 102:605–622
Hadari YR, Gotoh N, Kouhara H, Lax I, Schlessinger J (2001) Critical role for the docking-protein FRS2α in FGF receptor-mediated signal transduction pathways. Proc Natl Acad Sci U S A 98:8578–8583
Hales M, Rosenau W, Okerlund MD, Galante M (1982) Carcinoma of the thyroid with a mixed medullary and follicular pattern: morphologic, immunohistochemical, and clinical laboratory studies. Cancer 50:1352–1359
Harach HR (1985) Solid cell nests of the thyroid. An anatomical survey and immunohistochemical study for the presence of thyroglobulin. Acta Anat 122:249–253
Harach HR (1988) Solid cell nests of the thyroid. J Pathol 155:191–200
Harmon BG, Kelley LC (2001) Immunohistochemistry of ultimobranchial thyroid carcinomas in seven slaughtered cows and one bull. J Vet Diagn Investig 13:101–105
Hick AC, Delmarcelle AS, Bouquet M, Klotz S, Copetti T et al (2013) Reciprocal epithelial:endothelial paracrine interactions during thyroid development govern follicular organization and C-cells differentiation. Dev Biol 381:227–240
Jacobs JW, Lund PK, Potts JT Jr, Bell NH, Habener JF (1981) Procalcitonin is a glycoprotein. J Biol Chem 256:2803–2807
Janzer RC, Weber E, Hedinger C (1979) The relation between solid cell nests and C cells of the thyroid gland: an immunohistochemical and morphometric investigation. Cell Tissue Res 197:295–312
Johansson E, Andersson L, Ornros J, Carlsson T, Ingeson-Carlsson C et al (2015) Revising the embryonic origin of thyroid C cells. Development 142:3519–3528
Johnston D, Hatzis D, Sunday ME (1998) Expression of v-Ha-ras driven by the calcitonin/calcitonin gene-related peptide promotor: a novel transgenic murine model for medullary thyroid carcinoma. Oncogene 16:167–177
Kameda Y (1970) Increased mitotic activity of the parafollicular cells of the dog thyroid in experimentally induced hypercalcemia. Arch Histol Jpn 32:179–192
Kameda Y (1971) The occurrence of a special parafollicular cell complex in and beside the dog thyroid gland. Arch Histol Jpn 33:115–132
Kameda Y (1973) Electron microscopic studies on the parafollicular cells and parafollicular cell complexes in the dog. Arch Histol Jpn 36:89–105
Kameda Y (1974a) Light and electron microscopic alterations of the dog parafollicular cells induced by antithyroid drug. Arch Histol Jpn 36:205–220
Kameda Y (1974b) Relationship between the thyroid parafollicular cells and pituitary gland. Arch Histol Jpn 37:225–244
Kameda Y (1977) Electron microscopical and immunohistochemical study on parafollicular cell complex with reference to parafollicular cell as a paraneuron. Arch Histol Jpn 40(suppl):133–145
Kameda Y (1982a) The cysts in C cell complexes of dog thyroids studied by immunoperoxidase staining and autoradiography. Arch Histol Jpn 45:437–448
Kameda Y (1982b) Degranulation and appearance of vesicular inclusions in canine C cells after administration of antithyroid drug. Cell Tissue Res 225:315–318
Kameda Y (1983) Distribution of C cells in monkey thyroid glands as studied by the immunoperoxidase method using anti-calcitonin and anti-C-thyroglobulin antisera. Arch Histol Jpn 46:221–228
Kameda Y (1984) Dog thyroid glands after chronic administration of antithyroid drugs. Am J Pathol 117:316–325
Kameda Y (1987) Immunohistochemical demonstration of keratins in the cysts of thyroid glands, parathyroid glands, and C-cell complexes of the dog. Amer J Anat 180:87–99
Kameda Y (1995a) Evidence to support the distal vagal ganglion as the origin of C cells of the ultimobranchial gland in the chick. J Comp Neurol 359:1–14
Kameda Y (1995b) Co-expression of vimentin and 19S-thyroglobulin in follicular cells located in the C-cell complex of dog thyroid gland. J Histochem Cytochem 43:1097–1106
Kameda Y (1996) Differential distribution of S-100 protein and vimentin in the hypophyseal pars tuberalis of the guinea pig. J Histochem Cytochem 44:501–510
Kameda Y (2016) Cellular and molecular events on the development of mammalian thyroid C cells. Dev Dyn 245:323–341
Kameda Y (2017) Morphological and molecular evolution of the ultimobranchial gland of nonmammalian vertebrates, with special reference to the chicken C cells. Dev Dyn 246:719–739
Kameda Y, Ikeda A (1980a) Immunohistochemical study of the C-cell complex of dog thyroid glands with reference to the reactions of calcitonin, C-thyroglobulin and 19S thyroglobulin. Cell Tissue Res 208:405–415
Kameda Y, Ikeda A (1980b) Immunohistochemical study of C-cell complexes in dogs after induced hypercalcemica, antithyroid drug treatment and hypophysectomy. Cell Tissue Res 208:417–432
Kameda Y, Shigemoto H, Ikeda A (1980) Development and cytodifferentiation of C cell complexes in dog fetal thyroid. Cell Tissue Res 206:403–415
Kameda Y, Ikeda K, Ikeda A (1981) Uptake of radioiodine in follicles of dog C-cell complexes studied by autoradiograph and immunoperoxidase staining. Anat Rec 200:461–470
Kameda Y, Nishimaki T, Chisaka O, Iseki S, Sucov HM (2007a) Expression of the epithelial marker E-cadherin by thyroid C cells and their precursors during murine development. J Histochem Cytochem 55:1075–1088
Kameda Y, Nishimaki T, Miura M, Jiang SX, Guillemot F (2007b) Mash1 regulates the development of C cells in mouse thyroid glands. Dev Dyn 236:262–270
Kameda Y, Ito M, Nishimaki T, Gotoh N (2009) FRS2α is required for the separation, migration, and survival of pharyngeal-endoderm derived organs including thyroid, ultimobranchial body, parathyroid, and thymus. Dev Dyn 238:503–513
Kameda Y, Saitoh T, Nemoto N, Katoh T, Iseki S, Fujimura T (2013) Hes1 is required for the development of pharyngeal organs and survival of neural crest-derived mesenchymal cells in pharyngeal arches. Cell Tissue Res 353:9–25
Kimura S, Hara Y, Pineau T, Fernandez-Salguero P, Fox CH, Ward JM, Gonzalez FJ (1996) The T/ebp null mouse: thyroid-specific enhancer-binding protein is essential for the organogenesis of the thyroid, lung, ventral forebrain, and pituitary. Genes Dev 10:60–69
Koster MI, Roop DR (2004) The role of p63 in development and differentiation of the epidermis. J Dermatol Sci 34:3–9
Koumarianou P, Goméz-López G, Santisteban P (2017) Pax8 controls thyroid follicular polarity through cadherin-16. J Cell Sci 130:219–231
Kusakabe T, Hoshi N, Kimura S (2006a) Origin of the ultimobranchial body cyst: T/ebp/Nkx2.1 expression is required for development and fusion of the ultimobranchial body to the thyroid. Dev Dyn 235:1300–1309
Kusakabe T, Kawaguchi A, Hoshi N, Kawaguchi R, Hoshi S, Kimura S (2006b) Thyroid-specific enhancer-binding protein/NKX2.1 is required for the maintenance of ordered architecture and function of the differentiated thyroid. Mol Endocrinol 20:1796–1809
Lane EB, Hogan BL, Kurkinen M, Garrels JI (1983) Co-expression of vimentin and cytokeratins in parietal endodermal cells of early mouse embryo. Nature 303:701–704
Lazzaro D, Price M, De Felice M, Di Lauro R (1991) The transcription factor TTF-1 is expressed at the onset of thyroid and lung morphogenesis and in restricted regions of the foetal brain. Development 113:1093–1104
Le Douarin N, Fontaine J, Le Lièvre C (1974) New studies on the neural crest origin of the avian ultimobranchial glandular cells—interspecific combinations and cytochemical characterization of C cells based on the uptake of biogenic amine precursors. Histochemistry 38:297–305
Le Lièvre CS, Le Douarin NM (1975) Mesenchymal derivatives of the neural crest: analysis of chimaeric quail and chick embryos. J Embryol Exp Morph 34:125–154
Liang S, Johansson E, Barila G, Altschuler DL, Fagman H, Nilsson M (2018) A branching morphogenesis program governs embryonic growth of the thyroid gland. Development 145:dev146829
Ljungberg O, Nilsson PO (1985) Hyperplastic and neoplastic changes in ultimobranchial remnants and in parafollicular (C) cells in bulls: a histologic and immunohistochemical study. Vet Pathol 22:95–103
Ljungberg O, Ericsson UB, Bondeson L, Thorell J (1983) A compound follicular-parafollicular cell carcinoma of the thyroid: a new tumor entity? Cancer 52:1053–1061
Macias MJ, Martin-Malpartida P, Massagué J (2015) Structural determinants of SMAD function in TGF-β signaling. Trends Biochem Sci 40:296–308
Manley NR, Capecchi MR (1995) The role of Hoxa-3 in mouse thymus and thyroid development. Development 121:1989–2003
Manley NR, Capecchi MR (1998) Hox group3 paralogs regulate the development and migration of the thymus, thyroid, and parathyroid glands. Dev Biol 195:1–15
Mansouri A, Chowdhury K, Gruss P (1998) Follicular cells of the thyroid gland require Pax8 gene function. Nature Genet 19:87–90
Martin-Lacave I, Borrero MJ, Utrilla JC, Fernández-Santos JM, de Miguel M, Morillo J, Guerrero JM, Garcia-Marin R, Conde E (2009) C cells evolve at the same rhythm as follicular cells when thyroidal status changes in rats. J Anat 214:301–309
Matias-Guiu X (1999) Mixed medullary and follicular carcinoma of the thyroid. Am J Pathol 155:1413–1418
Mizukami Y, Nonomura A, Michigishi T, Noguchi M, Hashimoto T, Nakamura S, Ishizaki T (1994) Solid cell nests of the thyroid; a histologic and immunohistochemical study. Am J Clin Pathol 101:186–191
Morillo-Bernal J, Fernández-Santos JM, Utrilla JC, de Miguel M, Garcia-Marin R, Martin-Lacave I (2009) Functional expression of the thyrotropin receptor in C cells: new insights into their involvement in the hypothalamic-pituitary-thyroid axis. J Anat 215:150–158
Nilsson M, Fagman H (2017) Development of the thyroid gland. Development 144:2123–2140
Nilsson M, Williams D (2016) On the origin of cells and derivation of thyroid cancer: C cell story revisited. Eur Thyroid J 5:79–93
Noel M, Delehaye MC, Segond N, Lasmoles F, Caillou B, Gardet P, Fragu P, Moukhtar MS (1991) Study of calcitonin and thyroglobulin gene expression in human mixed follicular and medullary thyroid carcinoma. Thyroid 1:249–256
Ohuchi H, Hori Y, Yamasaki M, Harada H, Sekine K, Kato S, Itoh N (2000) FGF10 acts as a major ligand for FGF receptor 2 IIIb in mouse multi-organ development. Biochem Biophys Res Commun 277:643–649
Ozaki T, Nagashimma K, Kusakabe T, Kakudo K, Kimura S (2011) Development of thyroid gland and ultimobranchial body cyst in independent of p63. Lab Investig 91:138–146
Pagan R, Martin I, Alonso A, Llobera M, Vilaró S (1996) Vimentin filaments follow the preexisting cytokeratin network during epithelial-mesenchymal transition of cultured neonatal rat hepatocytes. Exp Cell Res 222:333–344
Papotti M, Negro F, Carney JA, Bussolati G, Lloyd RV (1997) Mixed medullary-follicular carcinoma of the thyroid. A morphological, immunohistochemical and in situ hybridization analysis of 11 cases. Virchows Arch 430:397–405
Parlato R, Rosica A, Rodriguez-Mallon A, Affuso A, Postiglione MP, Arra C, Mansouri A, Kimura S, Di Lauro R, De Felice M (2004) An integrated regulatory network controlling survival and migration in thyroid organogenesis. Dev Biol 276:464–475
Pasca di Magliano M, Di Lauro R, Zannini M (2000) Pax8 has a key role in thyroid cell differentiation. Proc Natl Acad Sci U S A 97:13144–13149
Peng TC, Cooper CW, Petrusz P, Volpert EM (1975) Identification of C-cells in normal and goitrous rat thyroid tissues using antiserum to rat thyrocalcitonin and the immunoperoxidase bridge technique. Endocrinology 97:1537–1544
Pfaltz M, Hedinger CE, Mühlethaler JP (1983) Mixed medullary and follicular carcinoma of the thyroid. Virch Arch A Pathol Anat Histopathol 400:53–59
Preto A, Cameselle-Teijeiro J, Moldes-Boullosa J, Soares P, Cameselle-Teijeiro JF et al (2004) Telomerase expression and proliferative activity suggest a stem cell role for thyroid solid cell nests. Mod Pathol 17:819–826
Reis-Filho JS, Preto A, Soares P, Ricardo S, Cameselle-Teijeiro J, Sobrinho-Simoes M (2003) P63 expression in solid cell nests of the thyroid: further evidence for a stem cell origin. Mod Pathol 16:43–48
Reynolds L, Jones K, Winton DJ, Cranston A, Houghton C, Howard L, Ponder BAJ, Smith DP (2001) C-cell and thyroid epithelial tumours and altered follicular development in transgenic mice expressing the long isoform of Men 2A RET. Oncogene 20:3986–3994
Rios Moreno MJ, Galera-Ruiz H, De Miguel M, López MIC, Illanes M, Galera-Davidson H (2011) Immunohistochemical profile of solid cell nest of thyroid gland. Endocr Pathol 22:35–39
Rodriguez W, Jin L, Janssens V, Pierreux C, Hick AC, Urizar E, Costagliola S (2012) Deletion of the RNaseIII enzyme Dicer in thyroid follicular cells causes hypothyroidism with signs of neoplastic alterations. PLoS One 7:e29929
Sawicki B, Zabel M (1997) Immunocytochemical study of parafollicular cells of the thyroid and ultimobranchial remnants of the European bison. Acta Histochem 99:223–230
Schmid KW (2015) Histopathology of C cells and medullary thyroid carcinoma. Recent Results Cancer Rec 204:41–60
Senoo M, Pinto F, Crum CP, McKeon F (2007) p63 is essential for the proliferative potential of stem cells in stratified epithelia. Cell 129:523–536
Silberschmidt D, Rodriguez-Mallon A, Mithboakar P, Cali G, Amendola E et al (2011) In vivo role of different domains and of phosphorylation in the transcription factor Nkx2-1. BMC Dev Biol 11:9
Teshima TH, Lourenco SV, Tucker AS (2016) Multiple cranial organ defects after conditionally knocking out Fgf10 in the neural crest. Front Physiol 7:488
Trueba SS, Augé J, Mattei G, Etchevers H, Martinovic J, Czernichow P, Vekemans M, Polak M, Attie-Bitach (2005) PAX8, TITF1, and FOXE1 gene expression patterns during human development: new insights into human thyroid development and thyroid dysgenesis-associated malformations. J Clin Endocrinol Metab 90:455–462
Vázquez-Román V, Utrilla JC, Fernández-Santos JM, Martin-Lacave I (2017) Immunohistochemical profiling of the ultimobranchial remnants in the rat postnatal thyroid gland. J Morphol 278:1114–1124
Velez-delValle C, Marsch-Moreno M, Castro-Munozledo, Galván-Mendoza IJ, Kuri-Harcuch W (2016) Epithelial cell migration requires the interaction between the vimentin and keratin intermediate filaments. Sci Rep 6:24389
Villacorte M, Delmarcelle AS, Lernoux M, Bouquet M et al (2016) Thyroid follicle development requires Smad1/5- and endothelial cell-dependent basement membrane assembly. Development 143:1958–1970
Volante M, Papotti M, Roth J, Saremaslani P, Speel EJM, Lloyd RV, Carney JA, Heitz PU, Bussolati G, Komminoth P (1999) Mixed medullary-follicular thyroid carcinoma. Am J Pathol 155:1499–1509
Westerlund J, Andersson L, Carlsson T, Fagman H, Nilsson M (2013) Misguided migration of C cell precursors to extra-thyroidal locations related to defective pharyngeal pouch development in Shh deficient mice. Cell Dev Biol 2(issue 4):1000129
Williams ED, Toyn CE, Harach R (1989) The ultimobranchial gland and congenital thyroid abnormalities in man. J Pathol 159:135–141
Wollman SH, Hilfer R (1977) Embryologic origin of various epithelial cell types in the thyroid gland of the rat. Anat Rec 189:467–478
Wollman SH, Hilfer R (1978) Embryologic origin of the various epithelial cell types in the second kind of thyroid follicle in the C3H mouse. Anat Rec 191:111–122
Wollman SH, Nève P (1971a) Postnatal development and properties of ultimobranchial follicles in the rat thyroid. Anat Rec 171:247–258
Wollman SH, Nève P (1971b) Ultimobranchial follicles in the thyroid glands of rats and mice. Recent Progr Horm Res 27:213–234
Xu PX, Zheng W, Laclef C, Maire P, Maas RL, Peters H, Xu X (2002) Eya1 is required for the morphogenesis of mammalian thymus, parathyroid and thyroid. Development 129:3033–3044
Yang A, Schweitzer R, Sun D, Kaghad M, Walker N et al (1999) p63 is essential for regenerative proliferation in limb, craniofacial and epithelial development. Nature 398:714–718
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kameda, Y. Follicular cell lineage in persistent ultimobranchial remnants of mammals. Cell Tissue Res 376, 1–18 (2019). https://doi.org/10.1007/s00441-018-02982-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-018-02982-9