Abstract
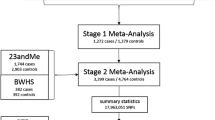
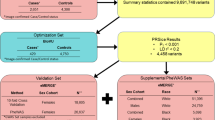
Uterine fibroid (UFs) affect 77 % of women by menopause and account for $9.4 billion in healthcare costs each year. Although UFs are heritable, genetic risk is poorly understood. The first genome-wide association study (GWAS) of UFs was recently performed in a Japanese population, with reported genome-wide significance for single nucleotide polymorphisms (SNPs) across three chromosomal regions. We tested these SNPs for association with UFs in US cohorts. Women were enrolled in the Right from the Start (RFTS) cohort and the BioVU DNA repository. UF status in both cohorts was determined by pelvic imaging. We tested 65 candidate and haplotype-tagging SNPs for association with UFs presence using logistic regression in RFTS and the top three GWAS-associated SNPs in BioVU. We also combined association results from both cohorts using meta-analysis. 1,086 European American (EA) cases and 1,549 controls were examined. Two SNP associations replicated [blocked early in transport 1 homolog (BET1L) rs2280543, RFTS–BioVU meta-odds ratio (OR) = 0.67 95 % confidence interval (CI) 0.38–0.96, Q = 0.70, I = 0, p = 6.9 × 10−3; trinucleotide repeat containing 6B (TNRC6B) rs12484776, RFTS–BioVU meta-OR = 1.21, 95 % CI 1.07–1.35, Q = 0.24, I = 28.37, p = 8.7 × 10−3). Meta-analyses combining evidence from RFTS, BioVU, and prior GWAS showed little heterogeneity in effect sizes across studies, with meta-p values between 7.45 × 10−8 and 3.89 × 10−9, which were stronger than prior GWAS and supported associations observed for all previously identified loci. These data suggest common variants increase risk for UF in both EA and Japanese populations. However, further research is needed to assess the role of these genes across other racial groups.
Similar content being viewed by others
References
Baird DD, Dunson DB (2003) Why is parity protective for uterine fibroids? Epidemiology 14:247–250
Baird DD, Dunson DB, Hill MC, Cousins D, Schectman JM (2003) High cumulative incidence of uterine leiomyoma in black and white women: ultrasound evidence. Am J Obstet Gynecol 188:100–107
Baird DD, Dunson DB, Hill MC, Cousins D, Schectman JM (2007) Association of physical activity with development of uterine leiomyoma. Am J Epidemiol 165:157–163
Barbieri CE, Baca SC, Lawrence MS, Demichelis F, Blattner M, Theurillat JP, White TA, Stojanov P, Van AE, Stransky N, Nickerson E, Chae SS, Boysen G, Auclair D, Onofrio RC, Park K, Kitabayashi N, MacDonald TY, Sheikh K, Vuong T, Guiducci C, Cibulskis K, Sivachenko A, Carter SL, Saksena G, Voet D, Hussain WM, Ramos AH, Winckler W, Redman MC, Ardlie K, Tewari AK, Mosquera JM, Rupp N, Wild PJ, Moch H, Morrissey C, Nelson PS, Kantoff PW, Gabriel SB, Golub TR, Meyerson M, Lander ES, Getz G, Rubin MA, Garraway LA (2012) Exome sequencing identifies recurrent SPOP, FOXA1 and MED12 mutations in prostate cancer. Nat Genet 44:685–689
Barrett JC, Fry B, Maller J, Daly MJ (2005) Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21:263–265
Bonatz G, Frahm SO, Andreas S, Heidorn K, Jonat W, Parwaresch R (1998) Telomere shortening in uterine leiomyomas. Am J Obstet Gynecol 179:591–596
Borengasser SJ, Lau F, Kang P, Blackburn ML, Ronis MJ, Badger TM, Shankar K (2011) Maternal obesity during gestation impairs fatty acid oxidation and mitochondrial SIRT3 expression in rat offspring at weaning. PLoS ONE 6:e24068
Burnett-Hartman AN, Fitzpatrick AL, Kronmal RA, Psaty BM, Jenny NS, Bis JC, Tracy RP, Kimura M, Aviv A (2012) Telomere-associated polymorphisms correlate with cardiovascular disease mortality in Caucasian women: the Cardiovascular Health Study. Mech Ageing Dev 133:275–281
Capel F, Viguerie N, Vega N, Dejean S, Arner P, Klimcakova E, Martinez JA, Saris WH, Holst C, Taylor M, Oppert JM, Sorensen TI, Clement K, Vidal H, Langin D (2008) Contribution of energy restriction and macronutrient composition to changes in adipose tissue gene expression during dietary weight-loss programs in obese women. J Clin Endocrinol Metab 93:4315–4322
Cha PC, Takahashi A, Hosono N, Low SK, Kamatani N, Kubo M, Nakamura Y (2011) A genome-wide association study identifies three loci associated with susceptibility to uterine fibroids. Nat Genet 43:447–450
Choudhury M, Jonscher KR, Friedman JE (2011) Reduced mitochondrial function in obesity-associated fatty liver: SIRT3 takes on the fat. Aging (Albany NY) 3:175–178
Cramer SF, Patel A (1990) The frequency of uterine leiomyomas. Am J Clin Pathol 94:435–438
Dragomir AD, Schroeder JC, Connolly A, Kupper LL, Hill MC, Olshan AF, Baird DD (2010) Potential risk factors associated with subtypes of uterine leiomyomata. Reprod Sci 17:1029–1035
Eggert SL, Huyck KL, Somasundaram P, Kavalla R, Stewart EA, Lu AT, Painter JN, Montgomery GW, Medland SE, Nyholt DR, Treloar SA, Zondervan KT, Heath AC, Madden PA, Rose L, Buring JE, Ridker PM, Chasman DI, Martin NG, Cantor RM, Morton CC (2012) Genome-wide linkage and association analyses implicate FASN in predisposition to uterine leiomyomata. Am J Hum Genet 91:621–628
El-Gharib MN, Elsobky ES (2010) Cytogenetic aberrations and the development of uterine leiomyomata. J Obstet Gynaecol Res 36:101–107
Estrada K, Krawczak M, Schreiber S, van Duijn K, Stolk L, van Meurs JB, Liu F, Penninx BW, Smit JH, Vogelzangs N, Hottenga JJ, Willemsen G, de Geus EJ, Lorentzon M, von Eller-Eberstein H, Lips P, Schoor N, Pop V, de KJ, Hofman A, Aulchenko YS, Oostra BA, Ohlsson C, Boomsma DI, Uitterlinden AG, van Duijn CM, Rivadeneira F, Kayser M (2009) A genome-wide association study of northwestern Europeans involves the C-type natriuretic peptide signaling pathway in the etiology of human height variation. Hum Mol Genet 18:3516–3524
Faerstein E, Szklo M, Rosenshein NB (2001) Risk factors for uterine leiomyoma: a practice-based case–control study. II. Atherogenic risk factors and potential sources of uterine irritation. Am J Epidemiol 153:11–19
Flake GP, Andersen J, Dixon D (2003) Etiology and pathogenesis of uterine leiomyomas: a review. Environ Health Perspect 111:1037–1054
Gabriel SB, Schaffner SF, Nguyen H, Moore JM, Roy J, Blumenstiel B, Higgins J, DeFelice M, Lochner A, Faggart M, Liu-Cordero SN, Rotimi C, Adeyemo A, Cooper R, Ward R, Lander ES, Daly MJ, Altshuler D (2002) The structure of haplotype blocks in the human genome. Science 296:2225–2229
Giralt A, Villarroya F (2012) SIRT3, a pivotal actor in mitochondrial functions: metabolism, cell death and aging. Biochem J 444:1–10
Green MF, Hirschey MD (2012) SIRT3 weighs heavily in the metabolic balance: a new role for SIRT3 in metabolic syndrome. J Gerontol A Biol Sci Med Sci 68(2):105–107
Guarente L (2011) Sirtuins, aging, and metabolism. Cold Spring Harb Symp Quant Biol 76:81–90
Guo Y, Shen H, Xiao P, Xiong DH, Yang TL, Guo YF, Long JR, Recker RR, Deng HW (2006) Genomewide linkage scan for quantitative trait loci underlying variation in age at menarche. J Clin Endocrinol Metab 91:1009–1014
Hartmann KE, Birnbaum H, Ben-Hamadi R, Wu EQ, Farrell MH, Spalding J, Stang P (2006) Annual costs associated with diagnosis of uterine leiomyomata. Obstet Gynecol 108:930–937
Je EM, Kim MR, Min KO, Yoo NJ, Lee SH (2012) Mutational analysis of MED12 exon 2 in uterine leiomyoma and other common tumors. Int J Cancer 131:E1044–E1047
Kurbanova MK, Koroleva AG, Sergeev AS (1989) Genetic-epidemiologic analysis of uterine myoma: assessment of repeated risk. Genetika 25:1896–1898
Laughlin SK, Baird DD, Savitz DA, Herring AH, Hartmann KE (2009) Prevalence of uterine leiomyomas in the first trimester of pregnancy: an ultrasound-screening study. Obstet Gynecol 113:630–635
Laughlin SK, Herring AH, Savitz DA, Olshan AF, Fielding JR, Hartmann KE, Baird DD (2010a) Pregnancy-related fibroid reduction. Fertil Steril 94:2421–2423
Laughlin SK, Schroeder JC, Baird DD (2010b) New directions in the epidemiology of uterine fibroids. Semin Reprod Med 28:204–217
Laughlin SK, Hartmann KE, Baird DD (2011) Postpartum factors and natural fibroid regression. Am J Obstet Gynecol 204:496
Liu H, Wang B, Han C (2011) Meta-analysis of genome-wide and replication association studies on prostate cancer. Prostate 71:209–224
Low SK, Takahashi A, Cha PC, Zembutsu H, Kamatani N, Kubo M, Nakamura Y (2012) Genome-wide association study for intracranial aneurysm in the Japanese population identifies three candidate susceptible loci and a functional genetic variant at EDNRA. Hum Mol Genet 21:2102–2110
Lumbiganon P, Rugpao S, Phandhu-fung S, Laopaiboon M, Vudhikamraksa N, Werawatakul Y (1996) Protective effect of depot-medroxyprogesterone acetate on surgically treated uterine leiomyomas: a multicentre case–control study. Br J Obstet Gynaecol 103:909–914
Luo X, Chegini N (2008) The expression and potential regulatory function of microRNAs in the pathogenesis of leiomyoma. Semin Reprod Med 26:500–514
Luoto R, Kaprio J, Rutanen EM, Taipale P, Perola M, Koskenvuo M (2000) Heritability and risk factors of uterine fibroids—the Finnish Twin Cohort study. Maturitas 37:15–26
Makinen N, Heinonen HR, Moore S, Tomlinson IP, van der Spuy ZM, Aaltonen LA (2011) MED12 exon 2 mutations are common in uterine leiomyomas from South African patients. Oncotarget 2:966–969
Malik M, Norian J, McCarthy-Keith D, Britten J, Catherino WH (2010) Why leiomyomas are called fibroids: the central role of extracellular matrix in symptomatic women. Semin Reprod Med 28:169–179
Markowski DN, Bartnitzke S, Loning T, Drieschner N, Helmke BM, Bullerdiek J (2012) MED12 mutations in uterine fibroids-their relationship to cytogenetic subgroups. Int J Cancer 131:1528–1536
Marsh EE, Lin Z, Yin P, Milad M, Chakravarti D, Bulun SE (2008) Differential expression of microRNA species in human uterine leiomyoma versus normal myometrium. Fertil Steril 89:1771–1776
Marshall LM, Spiegelman D, Barbieri RL, Goldman MB, Manson JE, Colditz GA, Willett WC, Hunter DJ (1997) Variation in the incidence of uterine leiomyoma among premenopausal women by age and race. Obstet Gynecol 90:967–973
Marshall LM, Spiegelman D, Goldman MB, Manson JE, Colditz GA, Barbieri RL, Stampfer MJ, Hunter DJ (1998) A prospective study of reproductive factors and oral contraceptive use in relation to the risk of uterine leiomyomata. Fertil Steril 70:432–439
Meister G, Landthaler M, Peters L, Chen PY, Urlaub H, Luhrmann R, Tuschl T (2005) Identification of novel argonaute-associated proteins. Curr Biol 15:2149–2155
Mestre-Alfaro A, Ferrer MD, Banquells M, Riera J, Drobnic F, Sureda A, Tur JA, Pons A (2012) Body temperature modulates the antioxidant and acute immune responses to exercise. Free Radic Res 46:799–808
Moore AB, Flake GP, Swartz CD, Heartwell G, Cousins D, Haseman JK, Kissling GE, Sidawy MK, Dixon D (2008) Association of race, age and body mass index with gross pathology of uterine fibroids. J Reprod Med 53:90–96
Myers SL, Baird DD, Olshan AF, Herring AH, Schroeder JC, Nylander-French LA, Hartmann KE (2012) Self-report versus ultrasound measurement of uterine fibroid status. J Womens Health (Larchmt) 21:285–293
Ojeda VJ (1979) The pathology of hysterectomy specimens. N Z Med J 89:169–171
Owerbach D, Johansen K, Billesbolle P, Poulsen S, Schroll M, Nerup J (1982) Possible association between DNA sequences flanking the insulin gene and atherosclerosis. Lancet 2:1291–1293
Parazzini F, Negri E, La VC, Chatenoud L, Ricci E, Guarnerio P (1996) Reproductive factors and risk of uterine fibroids. Epidemiology 7:440–442
Promislow JH, Makarushka CM, Gorman JR, Howards PP, Savitz DA, Hartmann KE (2004) Recruitment for a community-based study of early pregnancy: the Right From The Start study. Paediatr Perinat Epidemiol 18:143–152
Pulley J, Clayton E, Bernard GR, Roden DM, Masys DR (2010) Principles of human subjects protections applied in an opt-out, de-identified biobank. Clin Transl Sci 3:42–48
Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, Bender D, Maller J, Sklar P, de Bakker PI, Daly MJ, Sham PC (2007) PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 81:559–575
Rotwein P, Chyn R, Chirgwin J, Cordell B, Goodman HM, Permut MA (1981) Polymorphism in the 5′-flanking region of the human insulin gene and its possible relation to type 2 diabetes. Science 213:1117–1120
Samadi AR, Lee NC, Flanders WD, Boring JR III, Parris EB (1996) Risk factors for self-reported uterine fibroids: a case–control study. Am J Public Health 86:858–862
Snieder H, MacGregor AJ, Spector TD (1998) Genes control the cessation of a woman’s reproductive life: a twin study of hysterectomy and age at menopause. J Clin Endocrinol Metab 83:1875–1880
Sozen I, Arici A (2002) Interactions of cytokines, growth factors, and the extracellular matrix in the cellular biology of uterine leiomyomata. Fertil Steril 78:1–12
Sun J, Zheng SL, Wiklund F, Isaacs SD, Li G, Wiley KE, Kim ST, Zhu Y, Zhang Z, Hsu FC, Turner AR, Stattin P, Liu W, Kim JW, Duggan D, Carpten J, Isaacs W, Gronberg H, Xu J, Chang BL (2009) Sequence variants at 22q13 are associated with prostate cancer risk. Cancer Res 69:10–15
Takeda T, Sakata M, Isobe A, Miyake A, Nishimoto F, Ota Y, Kamiura S, Kimura T (2008) Relationship between metabolic syndrome and uterine leiomyomas: a case–control study. Gynecol Obstet Invest 66:14–17
Tao S, Wang Z, Feng J, Hsu FC, Jin G, Kim ST, Zhang Z, Gronberg H, Zheng LS, Isaacs WB, Xu J, Sun J (2012) A genome-wide search for loci interacting with known prostate cancer risk-associated genetic variants. Carcinogenesis 33:598–603
Terry KL, De Vivo I, Hankinson SE, Spiegelman D, Wise LA, Missmer SA (2007) Anthropometric characteristics and risk of uterine leiomyoma. Epidemiology 18:758–763
Thorisson GA, Smith AV, Krishnan L, Stein LD (2005) The International HapMap Project Web site. Genome Res 15:1592–1593
Treloar SA, Martin NG, Dennerstein L, Raphael B, Heath AC (1992) Pathways to hysterectomy: insights from longitudinal twin research. Am J Obstet Gynecol 167:82–88
Valdecantos MP, Perez-Matute P, Gonzalez-Muniesa P, Prieto-Hontoria PL, Moreno-Aliaga MJ, Martinez JA (2012) Lipoic acid improves mitochondrial function in nonalcoholic steatosis through the stimulation of sirtuin 1 and sirtuin 3. Obesity (Silver Spring) 20(10):1974–1983
Vasan RS, Larson MG, Aragam J, Wang TJ, Mitchell GF, Kathiresan S, Newton-Cheh C, Vita JA, Keyes MJ, O’Donnell CJ, Levy D, Benjamin EJ (2007) Genome-wide association of echocardiographic dimensions, brachial artery endothelial function and treadmill exercise responses in the Framingham Heart Study. BMC Med Genet 8(Suppl 1):S2
Vollenhoven B (1998) Introduction: the epidemiology of uterine leiomyomas. Baillieres Clin Obstet Gynaecol 12:169–176
Willer CJ, Li Y, Abecasis GR (2010) METAL: fast and efficient meta-analysis of genomewide association scans. Bioinformatics 26:2190–2191
Wise LA, Palmer JR, Harlow BL, Spiegelman D, Stewart EA, Adams-Campbell LL, Rosenberg L (2004) Reproductive factors, hormonal contraception, and risk of uterine leiomyomata in African-American women: a prospective study. Am J Epidemiol 159:113–123
Wise LA, Palmer JR, Spiegelman D, Harlow BL, Stewart EA, Adams-Campbell LL, Rosenberg L (2005) Influence of body size and body fat distribution on risk of uterine leiomyomata in US black women. Epidemiology 16:346–354
Wise LA, Ruiz-Narvaez EA, Palmer JR, Cozier YC, Tandon A, Patterson N, Radin RG, Rosenberg L, Reich D (2012) African ancestry and genetic risk for uterine leiomyomata. Am J Epidemiol 176:1159–1168
Xu Z, Kaplan NL, Taylor JA (2007) TAGster: efficient selection of LD tag SNPs in single or multiple populations. Bioinformatics 23:3254–3255
Acknowledgments
The field research for Right from the Start was supported by grants from the National Institute of Child and Human Development (R01HD043883 and R01HD049675) and the American Water Works Association Research Foundation (2579). Additional funds were provided by the Building Interdisciplinary Research Careers in Women’s Health career development program (2K12HD043483-11) to DRVE, the Vanderbilt Clinical and Translational Research Scholar Award 5KL2RR024977 to TLE from the National Center for Advancing Translational Sciences, the Vanderbilt CTSA award UL1TR000445 from the National Center for Advancing Translational Sciences, and the BioVU dataset used for the analyses described was obtained from Vanderbilt University Medical Center’s BioVU which is supported by institutional funding and by the Vanderbilt CTSA grant ULTR000445 from NCATS/NIH. The content of this manuscript are solely the responsibility of the authors and do not necessarily represent official views of the National Center for Advancing Translational Sciences or the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Additional information
T. L. Edwards and K. A. Michels are joint first authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
439_2013_1306_MOESM1_ESM.docx
Supplemental Figure 1. Candidate and haplotype tagging SNPs near BET1L. BET1L is oriented 5′ to 3′ and labeled in center. In bold are labeled GWAS index SNPs (DOCX 38 kb)
439_2013_1306_MOESM2_ESM.docx
Supplemental Figure 2. Candidate and haplotype tagging SNPs near SLK. SLK is oriented 5′ to 3′. In bold are labeled GWAS index SNPs (DOCX 35 kb)
439_2013_1306_MOESM3_ESM.docx
Supplemental Figure 3. Candidate and haplotype tagging SNPs near TNRC6B. TNRC6B is oriented 5′ to 3′ and labeled in center. In bold are labeled GWAS index SNPs (DOCX 137 kb)
439_2013_1306_MOESM4_ESM.docx
Supplemental Figure 4. Right from the Start linkage disequilibrium structure among European Americans. LD plots are presented for EAs cases and controls. All figures are oriented 5′ to 3′, right to left, relative to the minus strand. r 2 (shades of black) are indicated in percentages within squares in the LD plots, with solid blocks with numbers indicating r 2 = 1. Strong LD is indicated by dark gray and like gray and white indicate weak LD and/or low confidence values (DOCX 460 kb)
439_2013_1306_MOESM5_ESM.docx
Supplemental Table 1. Summary of adjusted index SNP and strongest (p ≤ 0.05) single locus associations with UFs among the Right from the Start and BioVU cohorts (DOCX 20 kb)
Rights and permissions
About this article
Cite this article
Edwards, T.L., Michels, K.A., Hartmann, K.E. et al. BET1L and TNRC6B associate with uterine fibroid risk among European Americans. Hum Genet 132, 943–953 (2013). https://doi.org/10.1007/s00439-013-1306-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-013-1306-3