Abstract
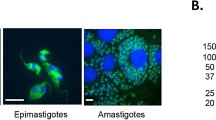
Animal African trypanosomosis is a serious constraint to livestock sector development in sub-Saharan Africa. The disease, mainly caused by Trypanosoma congolense, has a limitation in its diagnosis and treatment. There is urgent need for a simple, rapid detection technique to replace the few available serological tests that are of variable sensitivity and specificity. Currently, there is a promising use of recombinant proteins to improve on the trypanosome lysate to detect antibodies. In this respect, we have identified a stage-specific gene that is relatively highly expressed in metacyclic and blood trypomastigotes of T. congolense. According to previously obtained differential protein expression data, the gene TcIL3000.0.38630 (1,236 bp) is by 8.5 times more expressed in metacyclic and blood trypomastigotes than in procyclic trypomastigotes and epimastigotes. The same stage specific expression pattern was shown in Western blot analysis. In addition, in confocal laser scanning microscopy the Tc38630 protein was present in the cytosol and on the cell surface of metacyclic and blood trypomastigotes. Through bioinformatics, the Tc38630 had N-terminal signal sequence, hydrophilic extracellular domain, single transmembrane alpha-helix and short cytoplasmic domain, which is characteristic of the Trypanosoma brucei invariant surface glycoprotein. However, unlike T. brucei invariant surface glycoprotein, the Tc38630 existed as a single copy gene with a probable allelic polymorphism at the Nar I restriction site. The recombinant Tc38630-based ELISA detected antibodies against Tc38630 as early as 7 days post infection in experimentally infected mouse model. Taken together, our results suggest that the Tc38630 is a novel potential diagnostic antigen of Animal African trypanosomosis.





Similar content being viewed by others
References
Alsford S, Eckert S, Baker N, Glover L, Sanchez-Flores A, Leung KF, Turner DJ, Field MC, Berriman M, Horn D (2012) High-throughput decoding of antitrypanosomal drug efficacy and resistance. Nature 482(7384):232–237
Donelson JE (2003) Antigenic variation and the African trypanosome genome. Acta Trop 85(3):391–404
Donelson JE, Hill KL, El-Sayed NM (1998) Multiple mechanisms of immune evasion by African trypanosomes. Mol Biochem Parasitol 91(1):51–66
Eyford BA, Sakurai T, Smith D, Loveless B, Hertz-Fowler C, Donelson JE, Inoue N, Pearson TW (2011) Differential protein expression throughout the life cycle of Trypanosoma congolense, a major parasite of cattle in Africa. Mol Biochem Parasitol 177(2):116–125
Geerts S, Holmes PH, Eisler MC, Diall O (2001) African bovine trypanosomiasis: the problem of drug resistance. Trends Parasitol 17(1):25–28
Goto Y, Duthie MS, Kawazu S, Inoue N, Carter D (2011) Biased cellular locations of tandem repeat antigens in African trypanosomes. Biochem Biophys Res Commun 405(3):434–438
Grebaut P, Chuchana P, Brizard J-P, Demettre E, Seveno M, Bossard G, Jouin P, Vincendeau P, Bengaly Z, Boulange A, Cuny G, Holmuller P (2009) Identification of total and differentially expressed excreted-secreted proteins from Trypanosoma congolense strains exhibiting different virulence and pathogenicity. Int J Parasitol 39(10):1137–1150
Herbert WJ, Lumsden WHR (1976) Trypanosoma brucei: A rapid matching method for estimating the hosts parasitemia. Exp Parasitol 40(3):427–431
Hirumi H, Hirumi K (1991) In vitro cultivation of Trypanosoma congolense bloodstream forms in the absence of feeder cell layers. Parasitology 102(Pt 2):225–236
Jackson AP et al (2012) Antigenic diversity is generated by distinct evolutionary mechanisms in African trypanosome species. Proc Natl Acad Sci U S A 109(9):3416–3421
Kristjanson PM, Swallow BM, Rowlands GJ, Kruska RL, de Leeuw PN (1999) Measuring the costs of African animal trypanosomosis, the potential benefits of control and returns to research. Agr Syst 59(1):79–98
Magez S, Radwanska M (2009) African trypanosomiasis and antibodies: implications for vaccination, therapy and diagnosis. Future Microbiol 4(8):1075–1087
Marcotty T, Simukoko H, Berkvens D, Vercruysse J, Praet N, Van den Bossche P (2008) Evaluating the use of packed cell volume as an indicator of trypanosomal infections in cattle in eastern Zambia. Prev Vet Med 87(3–4):288–300
McDermott JJ, Coleman PG (2001) Comparing apples and oranges - model-based assessment of different tsetse-transmitted trypanosomosis control strategies. Int J Parasitol 31(5–6):603–609
Nguyen Thu T, Goto Y, Lun Z-R, Kawazu S-I, Inoue N (2012) Tandem repeat protein as potential diagnostic antigen for Trypanosoma evansi infection. Parasitol Res 110(2):733–739
Njiru ZK, Mikosza AS, Matovu E, Enyaru JC, Ouma JO, Kibona SN, Thompson RC, Ndung’u JM (2008) African trypanosomiasis: sensitive and rapid detection of the sub-genus Trypanozoon by loop-mediated isothermal amplification (LAMP) of parasite DNA. Int J Parasitol 38(5):589–599
OIE (2012) Manual of Diagnostic Tests and Vaccines for Terrestrial Animals, 7th edn. OIE, Paris
Omamo SW, D’Ieteren GDM (2003) Managing animal trypanosomosis in Africa: issues and options. Rev Sci Tech OIE 22(3):989–1002
Picozzi K, Tilley A, Fevre EM, Coleman PG, Magona JW, Odiit M, Eisler MC, Welburn SC (2002) The diagnosis of trypanosome infections: Applications of novel technology for reducing disease risk. Afr J Biotechnol 1(2):39–45
Sakurai T, Sugimoto C, Inoue N (2008) Identification and molecular characterization of a novel stage-specific surface protein of Trypanosoma congolense epimastigotes. Mol Biochem Parasitol 161(1):1–11
Sambrook J, Russell DW (2001) Preparation and analysis of eukaryotic genomic DNA Molecular Cloning, 3rd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, pp 6.1–6.64
Thekisoe OM, Kuboki N, Nambota A, Fujisaki K, Sugimoto C, Igarashi I, Yasuda J, Inoue N (2007) Species-specific loop-mediated isothermal amplification (LAMP) for diagnosis of trypanosomosis. Acta Trop 102(3):182–189
Tran T, Claes F, Verloo D, De Greve H, Buscher P (2009) Towards a new reference test for surra in camels. Clin Vaccine Immunol 16(7):999–1002
Ziegelbauer K, Overath P (1992) Identification of invariant surface glycoproteins in the blood-stream stage of Trypanosoma brucei. J Biol Chem 267(15):10791–10796
Ziegelbauer K, Multhaup G, Overath P (1992) Molecular characterization of two invariant surface glycoproteins specific for the bloodstream stage of Trypanosoma brucei. J Biol Chem 267(15):10797–10803
Ziegelbauer K, Rudenko G, Kieft R, Overath P (1995) Genomic organization of an invariant surface glycoprotein gene family of Trypanosoma brucei. Mol Biochem Parasitol 69(1):53–63
Acknowledgements
The authors are grateful to Dusit Laohasinarong, Jose Angeles, Hassan Hakimi, Nguyen Thu Thuy and Victor Zulu for their technical assistances. This study was financially supported by the Ministry of Education, Culture, Sports, Science and Technology (MEXT) Japan and Japan Society for the Promotion of Science (JSPS). This work was in part supported by SATREPS from Japan Science and Technology Agency (JST) and Japan International Cooperation Agency (JICA) to YS.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mochabo, K.M., Zhou, M., Suganuma, K. et al. Expression, immunolocalization and serodiagnostic value of Tc38630 protein from Trypanosoma congolense . Parasitol Res 112, 3357–3363 (2013). https://doi.org/10.1007/s00436-013-3515-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-013-3515-z