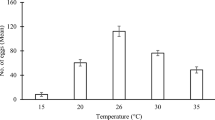
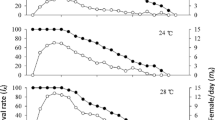
Abstract
The influence of temperature on the development and survival of Toxocara canis larvae was investigated under laboratory conditions, in water at 15, 20, 25, 30 and 35°C and at room temperature 22°C ± 1°C. T. canis eggs were able to develop to the larvated stage at all the tested temperatures. Development rate increased with temperature. Linear regression of development rate against temperature predicted a lower development threshold of 11.8°C. Eggs survived cooling to 1 and −2°C for 6 weeks, and could develop to the infective, larvated stage when transferred to higher temperatures, but their development rates were then retarded compared with non-chilled eggs. Larvated eggs remained viable after 7 weeks of incubation across the tested temperature range, with the highest percentage viability (47%) obtained at 25°C. Development of eggs to the infective larval stage required, on average, 121 degree days between 20°C and 30°C. Results provide a basis for predicting variation in the infectivity of eggs in the environment over time in different climates.



Similar content being viewed by others
References
Al-Tae ARA, Al-Bashir NM (1988) The viability and infectivity of Toxocara canis infective larvae after a prolonged period of storage at different temperatures. Mircen J Appl Microb 4:349–356
Basualdo JA, Ciarmela ML, Sarimento PL, Minivielle MC (2000) Biological activity of Paecilomyces genus against Toxocara canis eggs. Parasitol Res 86:854–859
Brunaska MP, Dubinsky KT (1995) Toxocara canis: ultrasturctual aspects of larval moulting in the maturing eggs. Int J Parasitol 25:683–690
Colli CM, Rubinsky-Elefant G, Paludo ML, Falavigna DLM, Guilherem EV, Mattia S, Araujo SM, Ferreira EC, Previdelli ITS, Falavigna-Guilherme AL (2010) Serological, clinical and epidemiological evaluation of toxocariasis in urban areas of South Brazil. Rev Inst Med Trop S Paolo 52:69–74
Crowcroft NS, Gillespie SH (1991) Hatching of second-stage larvae of Toxocara canis: a rapid method for processing large numbers of worms. J Helminthol 65:311–312
Cuellar del Hoyo C, Fenoy-Rodriquez S, Aguila de la Puente C, Guillen-Liera JL (1986) Nuevos datos sobre la fotodependencia del desarrollo embrionario de Toxocara canis. Rev Iber Parasitol 46:409–417
Eriksen L (1990) Ascaris suum: influence of egg density on in vitro development from embryonated egg to infective stage. Acta Vet Scand 31:489–491
Gamboa MI (2005) Effects of temperature and humidity on the development of eggs of Toxocara canis under laboratory conditions. J Helminthol 79:327–331
Glickman LT, Schantz PM (1981) Epidemiology and pathogenesis of zoonotic toxocariasis. Epidemiol Rev 3:230–250
Hill IR, Denham DA, Scholtz CL (1985) Toxocara canis larvae in the brain of a British child. Trans R Soc Trop Med Hyg 79:351–354
Huang SP, Pereira AC (1994) Influence of inoculum density, host, and low temperature period on delayed hatching of Meloidogyne javanica eggs. J Nematol 26:72–75
Jenkins EJ, Kutz SJ, Hoberg EP, Polley L (2006) Bionomics of larvae of Parelaphostrongylus odocoilei (Nematoda: Protostrongylidae) in experimentally infected gastropod intermediate hosts. J Parasitol 92:298–305
Kasai T (1995) Chemotherapy of larval toxocarosis: progress and problems. Overview from veterinary aspects. Helminthologia 32:133–141
Levine ND (1968) Nematode parasites of domestic animals and man, 2nd edn. Minneapolis, Burgess, pp 317–372
McSorley R (2003) Adaptations of nematodes to environmental extremes. Florida Entomologist 86:138–142
Mizgajska H (2001) Eggs of Toxocara spp. in the environment and their public health implications. J Helminthol 75:147–151
Mizgajska-Wiktor H, Uga S (2006) Exposure and environmental contamination. In: Holland CV, Smith HV (eds) Toxocara, the enigmatic parasite, CABI Publishing, pp 211–227
Morrondo P, Diez-Morrondo C, Pedreira J, Diez-Banos N, Sanchez-Andrade R, Paz-Silva A, Diez-Banos P (2006) Toxocara canis larvae viability after disinfectant-exposure. Parasitol Res 99:558–561
O'Donnell CJ, Meyer KB, Jones JV, Benton T, Kaneshiro ES, Nichols JS, Schaefer FW (1984) Survival of parasite eggs upon storage in sludge. Appl Environ Microbiol 48:618–625
Okoshi S, Usui M (1968) Experimental studies on Toxascaris leonina. 4. Development of eggs of three ascarids, T. leonina Toxocara canis and Toxocara cati, in dogs and cats. Jpn J Vet Sci 30:29–38
Oksanen A, Eriksen L, Roepstorff A, Ilsoe B, Nansen P, Lind P (1990) Embryonation and infectivity of Ascaris suum eggs. A comparison of eggs collected from worm uteri with eggs isolated from pig faeces. Acta Vet Scand 31:393–398
O'Lorcain P (1995a) The effects of freezing on the viability of Toxocara canis and T. cati embryonated eggs. J Helminthol 69:169–171
O'Lorcain P (1995b) The identification of the optimum conditions for the embryonation of Toxocara canis ova. Int J Environ Health Res 5:143–148
Onorato AR (1932) The effects of temperature and humidity on the ova of Toxocara canis and Trichuris vulpis. Am J Hyg 16:266–287
Overgaauw PAM (1997) Aspects of Toxocara epidemiology: toxocarosis in dogs and cats. Crit Rev Microbiol 23:233–251
Parsons JC (1987) Ascarid infections of cats and dogs. Vet Clin Am Small Pract 17:1307–1339
Rodriguez-Caballero A, Ponce-Macotela M, Peralta-Abarca GE, Martinez-Gordillo MN (2007) A simple and inexpensive in vitro method for retrieving fertilized Toxocara canis eggs. Parasitol Res 101:829–832
Romano N, Mon A, Rahmah N, Yal L, Rohela M (2010) Seroprevalence of toxocariasis among Orang Asli (Indigenous people) in Malaysia using two immunoassays. Trop Biomed 27:585–594
Rubinsky-Elefant G, Hirata CE, Yamamoto JH, Ferreira MU (2010) Human toxocariasis: diagnosis, worldwide seroprevalences and clinical expression of the systemic and ocular forms. Ann Trop Med Parasitol 104:3–23
Saunders LM, Tompkins DM, Hudson PJ (2000) The role of oxygen availability in the embryonation of Heterakis gallinarum eggs. Int J Parasitol 30:1481–1485
Schantz PM, Stehr-Green JK (1988) Toxocaral larva migrans. J Am Vet Med Assoc 192:28–32
Schjetlein J, Skorping A (1995) The temperature threshold for development of Elaphostrongylus rangiferi in the intermediate host: an adaptation to winter survival. Parasitology 111:103–110
Smith H, Holland C, Taylor M, Magnaval JF, Schantz P, Maizels R (2009) How common is human toxocariasis? Towards standardizing our knowledge. Trends Parasitol 25:182–188
Sprent JFA (1958) Observations on the development of Toxocara canis (Werner, 1782) in the dog. Parasitology 48:184–209
van Dijk J, Morgan ER (2008) The influence of temperature on the development, hatching and survival of Nematodirus battus larvae. Parasitology 135:269–283
van Dijk J, Morgan ER (2010) Variation in the hatching behaviour of Nematodirus battus: polymorphic bet hedging? Int J Parasitol 40:675–681
Won KY, Kruszon-Moran D, Schantz PM, Jones JL (2008) National seroprevalence and risk factors for zoonotic Toxocara spp. Infection. Am J Trop Med Hyg 79:552–557
Acknowledgements
DA is grateful for the support from Mansoura University and the Government of the Arab Republic of Egypt through the Channel Scheme.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Azam, D., Ukpai, O.M., Said, A. et al. Temperature and the development and survival of infective Toxocara canis larvae. Parasitol Res 110, 649–656 (2012). https://doi.org/10.1007/s00436-011-2536-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-011-2536-8