Abstract
Main conclusion
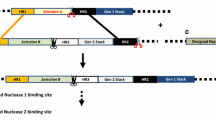
A gene targeting method has been developed, which allows the conversion of ‘breeding stacks’, containing unlinked transgenes into a ‘molecular stack’ and thereby circumventing the breeding challenges associated with transgene segregation.
A gene targeting method has been developed for converting two unlinked trait loci into a single locus transgene stack. The method utilizes intra-genomic homologous recombination (IGHR) between stably integrated target and donor loci which share sequence homology and nuclease cleavage sites whereby the donor contains a promoterless herbicide resistance transgene. Upon crossing with a zinc finger nuclease (ZFN)-expressing plant, double-strand breaks (DSB) are created in both the stably integrated target and donor loci. DSBs flanking the donor locus result in intra-genomic mobilization of a promoterless selectable marker-containing donor sequence, which can be utilized as a template for homology-directed repair of a concomitant DSB at the target locus resulting in a functional selectable marker via nuclease-mediated cassette exchange (NMCE). The method was successfully demonstrated in maize using a glyphosate tolerance gene as a donor whereby up to 3.3 % of the resulting progeny embryos cultured on selection medium regenerated plants with the donor sequence integrated into the target locus. The process could be extended to multiple cycles of trait stacking by virtue of a unique intron sequence homology for NMCE between the target and the donor loci. This is the first report that describes NMCE via IGHR, thereby enabling trait stacking using conventional crossing.





Similar content being viewed by others
References
Ainley WM, Sastry-Dent L, Welter ME, Murray MG, Zeitler B, Amora R, Corbin DR, Miles RR, Arnold NL, Strange TL, Simpson MA, Cao Z, Carroll C, Pawelczak KS, Blue R, West K, Rowland LM, Perkins D, Samuel P, Dewes CM, Shen L, Sriram S, Evans SL, Rebar EJ, Zhang L, Gregory PD, Urnov FD, Webb SR, Petolino JF (2013) Trait stacking via targeted genome editing. Plant Biotechnol J 11(9):1126–1134. doi:10.1111/pbi.12107
Allen GC, Flores-Vergara MA, Krasynanski S, Kumar S, Thompson WF (2006) A modified protocol for rapid DNA isolation from plant tissues using cetyltrimethylammonium bromide. Nat Protoc 1(5):2320–2325. doi:10.1038/nprot.2006.384
Ayar A, Wehrkamp-Richter S, Laffaire JB, Le Goff S, Levy J, Chaignon S, Salmi H, Lepicard A, Sallaud C, Gallego ME, White CI, Paul W (2013) Gene targeting in maize by somatic ectopic recombination. Plant Biotechnol J 11(3):305–314. doi:10.1111/Pbi.12014
Christian M, Cermak T, Doyle EL, Schmidt C, Zhang F, Hummel A, Bogdanove AJ, Voytas DF (2010) Targeting DNA double-strand breaks with TAL effector nucleases. Genetics 186(2):757–761. doi:10.1534/genetics.110.120717
Endo M, Osakabe K, Ichikawa H, Toki S (2006) Molecular characterization of true and ectopic gene targeting events at the acetolactate synthase gene in Arabidopsis. Plant Cell Physiol 47(3):372–379. doi:10.1093/pcp/pcj003
Epinat JC, Arnould S, Chames P, Rochaix P, Desfontaines D, Puzin C, Patin A, Zanghellini A, Paques F, Lacroix E (2003) A novel engineered meganuclease induces homologous recombination in yeast and mammalian cells. Nucleic Acids Res 31(11):2952–2962
Even-Faitelson L, Samach A, Melamed-Bessudo C, Avivi-Ragolsky N, Levy AA (2011) Localized egg-cell expression of effector proteins for targeted modification of the Arabidopsis genome. Plant J 68(5):929–937. doi:10.1111/j.1365-313X.2011.04741.x
Fauser F, Roth N, Pacher M, Ilg G, Sanchez-Fernandez R, Biesgen C, Puchta H (2012) In planta gene targeting. Proc Natl Acad Sci USA 109(19):7535–7540. doi:10.1073/pnas.1202191109
Gao HR, Smith J, Yang MZ, Jones S, Djukanovic V, Nicholson MG, West A, Bidney D, Falco SC, Jantz D, Lyznik LA (2010) Heritable targeted mutagenesis in maize using a designed endonuclease. Plant J 61(1):176–187. doi:10.1111/j.1365-313X.2009.04041.x
Gao J, Wang G, Ma S, Xie X, Wu X, Zhang X, Wu Y, Zhao P, Xia Q (2015) CRISPR/Cas9-mediated targeted mutagenesis in Nicotiana tabacum. Plant Mol Biol 87(1–2):99–110. doi:10.1007/s11103-014-0263-0
Hallauer AR, Lamkey KR, White PR (1997) Registration of five inbred lines of maize: B102, B103, B104, B105, and B106. Crop Sci 37(4):1405–1406
Hanin M, Volrath S, Bogucki A, Briker M, Ward E, Paszkowski J (2001) Gene targeting in Arabidopsis. Plant J 28(6):671–677 (pii 1183)
Kim YG, Cha J, Chandrasegaran S (1996) Hybrid restriction enzymes: zinc finger fusions to Fok I cleavage domain. Proc Natl Acad Sci USA 93(3):1156–1160
Kumar S, Fladung M (2001) Controlling transgene integration in plants. Trends Plant Sci 6(4):155–159 (pii S1360-1385(01)01890-8)
Kumar S, AlAbed D, Whitteck JT, Chen W, Bennett S, Asberry A, Wang X, DeSloover D, Rangasamy M, Wright TR, Gupta M (2015a) A combinatorial bidirectional and bicistronic approach for coordinated multi-gene expression in corn. Plant Mol Biol 87(4–5):341–353. doi:10.1007/s11103-015-0281-6
Kumar S, AlAbed D, Worden A, Novak S, Wu H, Ausmus C, Beck M, Robinson H, Minnicks T, Hemingway D, Lee R, Skaggs N, Wang L, Marri P, Gupta M (2015b) A modular gene targeting system for sequential transgene stacking in plants. J Biotechnol 207:12–20. doi:10.1016/j.jbiotec.2015.04.006
Kumar S, Barone P, Smith M (2016) Gene targeting and transgene stacking using intra genomic homologous recombination in plants. Plant Methods 12:11. doi:10.1186/s13007-016-0111-0
Liang Z, Zhang K, Chen K, Gao C (2014) Targeted mutagenesis in Zea mays using TALENs and the CRISPR/Cas system. J Genet Genom 41(2):63–68. doi:10.1016/j.jgg.2013.12.001
Lira JM, Cicchillo R, Yerkes CN, Robinson AE (2013) Glyphosate resistant plants and associated methods. US20130219552A1
Miller PD (2013) Method for improved transformation using agrobacterium. US Patent 0157369 A1
Moehle EA, Rock JM, Lee YL, Jouvenot Y, DeKelver RC, Gregory PD, Urnov FD, Holmes MC (2007) Targeted gene addition into a specified location in the human genome using designed zinc finger nucleases. Proc Natl Acad Sci USA 104(9):3055–3060. doi:10.1073/pnas.0611478104
Mueller D, Pope R (2009) Corn field guide. Iowa State University, Ames
Murray MG, Cuellar RE, Thompson WF (1978) DNA sequence organization in the pea genome. Biochemistry 17(26):5781–5790
Petolino JF, Kumar S (2015) Transgenic trait deployment using designed nucleases. Plant Biotechnol J. doi:10.1111/pbi.12457
Que Q, Chilton MD, de Fontes CM, He C, Nuccio M, Zhu T, Wu Y, Chen JS, Shi L (2010) Trait stacking in transgenic crops: challenges and opportunities. GM Crops 1(4):220–229. doi:10.4161/gmcr.1.4.13439
Rong YS, Golic KG (2000) Gene targeting by homologous recombination in Drosophila. Science 288(5473):2013–2018 (pii 8589)
Schiml S, Fauser F, Puchta H (2014) The CRISPR/Cas system can be used as nuclease for in planta gene targeting and as paired nickases for directed mutagenesis in Arabidopsis resulting in heritable progeny. Plant J 80(6):1139–1150. doi:10.1111/tpj.12704
Shukla VK, Doyon Y, Miller JC, DeKelver RC, Moehle EA, Worden SE, Mitchell JC, Arnold NL, Gopalan S, Meng XD, Choi VM, Rock JM, Wu YY, Katibah GE, Zhifang G, McCaskill D, Simpson MA, Blakeslee B, Greenwalt SA, Butler HJ, Hinkley SJ, Zhang L, Rebar EJ, Gregory PD, Urnov FD (2009) Precise genome modification in the crop species Zea mays using zinc-finger nucleases. Nature 459(7245):437–441. doi:10.1038/Nature07992
Siebert MW, Nolting SP, Hendrix W, Dhavala S, Craig C, Leonard BR, Stewart SD, All J, Musser FR, Buntin GD, Samuel L (2012) Evaluation of corn hybrids expressing Cry1F, Cry1A.105, Cry2Ab2, Cry34Ab1/Cry35Ab1, and Cry3Bb1 against Southern United States insect pests. J Econ Entomol 105(5):1825–1834. doi:10.1603/Ec12155
Storer NP, Thompson GD, Head GP (2012) Application of pyramided traits against Lepidoptera in insect resistance management for Bt crops. GM Crops Food 3(3):154–162. doi:10.4161/gmcr.20945
Wehrmann A, Van Vliet A, Opsomer C, Botterman J, Schulz A (1996) The similarities of bar and pat gene products make them equally applicable for plant engineers. Nat Biotechnol 14(10):1274–1278. doi:10.1038/nbt1096-1274
Wiedenheft B, Sternberg SH, Doudna JA (2012) RNA-guided genetic silencing systems in bacteria and archaea. Nature 482(7385):331–338. doi:10.1038/nature10886
Zhang F, Maeder ML, Unger-Wallace E, Hoshaw JP, Reyon D, Christian M, Li X, Pierick CJ, Dobbs D, Peterson T, Joung JK, Voytas DF (2010) High frequency targeted mutagenesis in Arabidopsis thaliana using zinc finger nucleases. Proc Natl Acad Sci USA 107(26):12028–12033. doi:10.1073/pnas.0914991107
Acknowledgments
We are grateful to Diaa AlAbed, Stephen Foulk, Huixia Wu, Carla Ausmus, Margaret Beck, Heather Robinson, Tatyana Minnicks, and Daren Hemingway for plant transformation and regeneration work, Jamie Torrence for greenhouse support, and Nicole Skaggs for molecular analysis. The authors would also like to thank Manju Gupta, Michelle Smith, and Rodrigo Sarria for their support. We also thank anonymous reviewers for their critical and constructive review of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing financial interests
The authors in this publication certify that they are current or former employees of Dow AgroSciences LLC, a subsidiary of The Dow Chemical Company. As employees, they authors may have some company stock (The Dow Chemical Company), but no other personal financial interest in the subject matter of the materials discussed in this manuscript.
Additional information
S. Kumar and A. Worden contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kumar, S., Worden, A., Novak, S. et al. A trait stacking system via intra-genomic homologous recombination. Planta 244, 1157–1166 (2016). https://doi.org/10.1007/s00425-016-2595-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-016-2595-2