Abstract
Introduction
The distribution of lymph node metastases in locally advanced Siewert type I and type II AEG (adenocarcinoma of the esophagogastric junction) remains unclear. The diversity of data in the literature reflects the non-uniformity of tumor stages and surgical procedures in previous studies.
Materials and methods
Based on a retrospective analysis from our single-center database, we examined distributions of lymph node metastases in types I and II cT2-4 AEG. The dataset comprised 44 patients; 19 and 25 patients had type I and type II, respectively. All patients underwent subtotal esophagectomy and total mediastinal lymphadenectomy, which included dissection of the upper mediastinal lymph nodes. The histological data of the surgical specimens were analyzed to evaluate metastasis rates in each lymph node station according to the Japanese Esophageal Society (JES) and American Joint Committee on Cancer (AJCC) guidelines.
Results
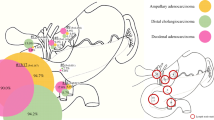
Lymph node metastases were observed in 75.0% cases (n = 33/44). There was no significant difference in the total lymph node metastasis rate between the two groups (type I 73.7% versus type II 76.0%). On comparing each lymph node region separately, no statistically significant differences were noted between the groups: upper mediastinal (type I 31.6% versus type II 24.0%), middle and lower mediastinal (type I 31.6% versus type II 44.0%), paragastric (type I 61.1% versus type II 76.0%), and celiac lymph nodes (type I 16.7% versus type II 25.0%).
Conclusion
In advanced clinical stages, the metastasis rate is high at all mediastinal lymph node regions in both type I and type II AEGs.



Similar content being viewed by others
Abbreviations
- AEG:
-
Adenocarcinoma of esophagogastric junction
- AJCC:
-
American Joint Committee on Cancer
- CT:
-
Chemotherapy
- CRT:
-
Chemoradiation
- DFS:
-
Disease-free survival
- JES:
-
Japan Esophageal Society
- LN:
-
Lymph node
- OS:
-
Overall survival
- UICC:
-
Union for International Cancer Control
References
Brown LM, Devesa SS, Chow WH (2008) Incidence of adenocarcinoma of the esophagus among white Americans by sex, stage, and age. J Natl Cancer Inst 100:1184–1187. https://doi.org/10.1093/jnci/djn211
Hur C, Y C, Miller M, Kong ECD, Nattinger KJ, Dunn M, Feuer EJ Trends in esophageal adenocarcinoma incidence and mortality. Cancer 119(2012):1149–1158. https://doi.org/10.1002/cncr.27834
Castro C, Bosetti C, Malvezzi M, Bertuccio P, Levi F, Negri E, La Vecchia C, Lunet N (2014) Patterns and trends in esophageal cancer mortality and incidence in Europe (1980-2011) and predictions to 2015. Ann Oncol 25:283–290. https://doi.org/10.1093/annonc/mdt486
Hatta W, Tong D, Lee YY, Ichihara S, Uedo N, Gotoda T (2017) Different time trend and management of esophagogastric junction adenocarcinoma in three Asian countries. Dig Endosc 29(Suppl 2):18–25. https://doi.org/10.1111/den.12808
Imamura Y, Watanabe M, Toihata T, Takamatsu M, Kawachi H, Haraguchi I, Ogata Y, Yoshida N, Saeki H, Oki E, Taguchi K, Yamamoto M, Morita M, Mine S, Hiki N, Baba H, Sano T (2019) Recent incidence trend of surgically resected esophagogastric junction adenocarcinoma and microsatellite instability status in Japanese patients. Digestion. 99:6–13. https://doi.org/10.1159/000494406
Siewert JR, Stein HJ (1998) Classification of adenocarcinoma of the oesophagogastric junction. Br J Surg 85:1457–1459
Haverkamp L, Seesing MF, Ruurda JP, Boone J, van Hillegersberg R (2017) Worldwide trends in surgical techniques in the treatment of esophageal and gastroesophageal junction cancer. Dis Esophagus 30:1–7. https://doi.org/10.1111/dote.12480
Lordick F, Mariette C, Haustermans K, Obermannová R, Arnold D ESMO Guidelines Committee. Oesophageal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 27(2016, suppl 5):v50–v57
Hölscher AH, Stahl M, Messmann H, Stuschke M, Meyer HJ, Porschen R (2016) New S3 guideline for esophageal cancer: important surgical aspects. Chirurg. 87:865–872. https://doi.org/10.1007/s00104-016-0214-1
Omloo JM, Lagarde SM, Hulscher JB, Reitsma JB, Fockens P, van Dekken H, Ten Kate FJ, Obertop H, Tilanus HW, van Lanschot JJ (2007) Extended transthoracic resection compared with limited transhiatal resection for adenocarcinoma of the mid/distal esophagus: five-year survival of a randomized clinical trial. Ann Surg 246:992–1000
Davies AR, Sandhu H, Pillai A, Sinha P, Mattsson F, Forshaw MJ, Gossage JA, Lagergren J, Allum WH, Mason RC (2014) Surgical resection strategy and the influence of radicality on outcomes in oesophageal cancer. Br J Surg 101:511–517. https://doi.org/10.1002/bjs.9456
Blank S, Schmidt T, Heger P, Strowitzki MJ, Sisic L, Heger U, Nienhueser H, Haag GM, Bruckner T, Mihaljevic AL, Ott K, Büchler MW, Ulrich A (2018) Surgical strategies in true adenocarcinoma of the esophagogastric junction (AEG II): thoracoabdominal or abdominal approach? Gastric Cancer 21:303–314. https://doi.org/10.1007/s10120-017-0746-1
Tosolini C, Reim D, Schirren R, Feith M, Friess H, Novotny AR (2019) Influence of the surgical technique on survival in the treatment of carcinomas of the true cardia (Siewert Type II) - right thoracoabdominal vs. transhiatal-abdominal approach. Eur J Surg Oncol 45:416–424. https://doi.org/10.1016/j.ejso.2018.09.017
Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines 2018 (ver. 5). Kanehara ISBN 978-4-307-20381-4 in Japanese
Japanese Gastric Cancer Association (2017) Japanese gastric cancer treatment guidelines 2014 (ver. 4). Gastric Cancer 20:1–19. https://doi.org/10.1007/s10120-016-0622-4
Yamashita H, Seto Y, Sano T, Makuuchi H, Ando N, Sasako M (2017) Japanese Gastric Cancer Association and the Japan Esophageal Society. Results of a nation-wide retrospective study of lymphadenectomy for esophagogastric junction carcinoma. Gastric Cancer 20(Suppl 1):69–83. https://doi.org/10.1007/s10120-016-0663-8
Kurokawa Y, Hiki N, Yoshikawa T, Kishi K, Ito Y, Ohi M, Wada N, Takiguchi S, Mine S, Hasegawa S, Matsuda T, Takeuchi H (2015) Mediastinal lymph node metastasis and recurrence in adenocarcinoma of the esophagogastric junction. Surgery. 157:551–555. https://doi.org/10.1016/j.surg.2014.08.099
Mine S, Sano T, Hiki N, Yamada K, Kosuga T, Nunobe S, Shigaki H, Yamaguchi T (2014) Thoracic lymph node involvement in adenocarcinoma of the esophagogastric junction and lower esophageal squamous cell carcinoma relative to the location of the proximal end of the tumor. Ann Surg Oncol 21:1596–1601. https://doi.org/10.1245/s10434-014-3548-2
Matsuda T, Takeuchi H, Tsuwano S, Nakamura R, Takahashi T, Wada N, Kawakubo H, Saikawa Y, Omori T, Kitagawa Y (2014) Optimal surgical management for esophagogastric junction carcinoma. Gen Thorac Cardiovasc Surg 62:560–566. https://doi.org/10.1007/s11748-014-0381-2
Lagarde SM, Phillips AW, Navidi M, Disep B, Griffin SM (2017) Clinical outcomes and benefits for staging of surgical lymph node mapping after esophagectomy. Dis Esophagus 30:1–7. https://doi.org/10.1093/dote/dox086
Feith M, Stein HJ, Siewert JR (2006) Adenocarcinoma of the esophagogastric junction: surgical therapy based on 1602 consecutive resected patients. Surg Oncol Clin N Am 15:751–764
Parry K, Haverkamp L, Bruijnen RC, Siersema PD, Ruurda JP, van Hillegersberg R (2015) Surgical treatment of adenocarcinomas of the gastro-esophageal junction. Ann Surg Oncol 22:597–603. https://doi.org/10.1245/s10434-014-4047-1
Leers JM, DeMeester SR, Chan N, Ayazi S, Oezcelik A, Abate E, Banki F, Lipham JC, Hagen JA, DeMeester TR (2009) Clinical characteristics, biologic behavior, and survival after esophagectomy are similar for adenocarcinoma of the gastroesophageal junction and the distal esophagus. J Thorac Cardiovasc Surg 138:594–602; discussion 601-602. https://doi.org/10.1016/j.jtcvs.2009.05.039
Yoshikawa T, Takeuchi H, Hasegawa S, Nozaki I, Kishi K, Ito S, Ohi M, Mine S, Hara J, Matsuda T, Hiki N, Kurokawa Y (2016) Theoretical therapeutic impact of lymph node dissection on adenocarcinoma and squamous cell carcinoma of the esophagogastric junction. Gastric Cancer 19:143–149. https://doi.org/10.1007/s10120-014-0439-y
Koyanagi K, Kato F, Kanamori J, Daiko H, Ozawa S, Tachimori Y (2018) Clinical significance of esophageal invasion length for the prediction of mediastinal lymph node metastasis in Siewert type II adenocarcinoma: a retrospective single-institution study. Ann Gastroenterol Surg 2:187–196. https://doi.org/10.1002/ags3.12069
Yura M, Takeuchi H, Fukuda K, Nakamura R, Suda K, Wada N, Kawakubo H, Kitagawa Y (2018) High-risk group of upper and middle mediastinal lymph node metastasis in patients with esophagogastric junction carcinoma. Ann Gastroenterol Surg 2:419–427. https://doi.org/10.1002/ags3.12215
Heger P, Blank S, Gooßen K, Nienhüser H, Diener MK, Ulrich A, Mihaljevic AL, Schmidt T (2019 Feb) Thoracoabdominal versus transhiatal surgical approaches for adenocarcinoma of the esophagogastric junction-a systematic review and meta-analysis. Langenbeck's Arch Surg 404(1):103–113. https://doi.org/10.1007/s00423-018-1745-3
Japan Esophageal Society (2017) Japanese Classification of Esophageal Cancer, 11th Edition: part I. Esophagus 14:1–36. https://doi.org/10.1007/s10388-016-0551-7
Tachimori Y, Ozawa S, Numasaki H, Matsubara H, Shinoda M, Toh Y, Udagawa H, Fujishiro M, Oyama T, Uno T (2016) Registration Committee for Esophageal Cancer of the Japan Esophageal Society. Efficacy of lymph node dissection by node zones according to tumor location for esophageal squamous cell carcinoma. Esophagus. 13:1–7
Rice TW, Apperson-Hansen C, Di Paola LM, Semple ME, Lerut TE, Orringer MB, Chen LQ, Hofstetter WL, Smithers BM, Rusch VW, Wijnhoven BP, Chen KN, Davies AR, D’Journo XB, Kesler KA, Luketich JD, Ferguson MK, Räsänen JV, van Hillegersberg R, Fang W, Durand L, Allum WH, Cecconello I, Cerfolio RJ, Pera M, Griffin SM, Burger R, Liu JF, Allen MS, Law S, Watson TJ, Darling GE, Scott WJ, Duranceau A, Denlinger CE, Schipper PH, Ishwaran H, Blackstone EH (2016) Worldwide Esophageal Cancer Collaboration: clinical staging data. Dis Esophagus 29:707–714. https://doi.org/10.1111/dote.12493
Rice TW, Ishwaran H, Ferguson MK, Blackstone EH, Goldstraw P (2017) Cancer of the esophagus and esophagogastric junction: an eighth edition staging primer. J Thorac Oncol 12:36–42. https://doi.org/10.1016/j.jtho.2016.10.016
Sisic L, Blank S, Nienhüser H, Dorr S, Haag GM, Jäger D, Bruckner T, Müller-Stich BP, Ott K, Büchler MW, Ulrich A, Schmidt T (2018) Prognostic differences in 8th edition TNM staging of esophagogastric adenocarcinoma after neoadjuvant treatment. Eur J Surg Oncol 44:1646–1656. https://doi.org/10.1016/j.ejso.2018.06.030
Matsuda T, Kurokawa Y, Yoshikawa T, Kishi K, Misawa K, Ohi M, Mine S, Hiki N, Takeuchi H (2016) Clinicopathological characteristics and prognostic factors of patients with Siewert type II esophagogastric junction carcinoma: a retrospective multicenter study. World J Surg 40:1672–1679. https://doi.org/10.1007/s00268-016-3451-z
Butter R, Lagarde SM, van Oijen MGH, Anderegg MCJ, Gisbertz SS, Meijer SL, Hulshof MCCM, Bergman JJGHM, van Berge Henegouwen MI, van Laarhoven HWM (2017) Treatment strategies in recurrent esophageal or junctional cancer. Dis Esophagus 30:1–9. https://doi.org/10.1093/dote/dox082
Depypere L, Lerut T, Moons J, Coosemans W, Decker G, Van Veer H, De Leyn P, Nafteux P (2017) Isolated local recurrence or solitary solid organ metastasis after esophagectomy for cancer is not the end of the road. Dis Esophags 30:1–8. https://doi.org/10.1111/dote.12508
Dresner SM, Griffin SM (2000) Pattern of recurrence following radical oesophagectomy with two-field lymphadenectomy. Br J Surg 87:1426–1433
van Hagen P, Wijnhoven BP, Nafteux P, Moons J, Haustermans K, De Hertogh G, van Lanschot JJ, Lerut T (2013) Recurrence pattern in patients with a pathologically complete response after neoadjuvant chemoradiotherapy and surgery for oesophageal cancer. Br J Surg 100:267–273. https://doi.org/10.1002/bjs.8968
Hulscher JB, van Sandick JW, Tijssen JG, Obertop H, van Lanschot JJ (2000) The recurrence pattern of esophageal carcinoma after transhiatal resection. J Am Coll Surg 191:143–148
Knight WRC, Zylstra J, Van Hemelrijck M, Griffin N, Jacques AET, Maisey N, Baker CR, Gossage JA, Largergren J, Davies AR (2018) Patterns of recurrence in oesophageal cancer following oesophagectomy in the era of neoadjuvant chemotherapy. BJS Open 1:182–190. https://doi.org/10.1002/bjs5.30
Oppedijk V, van der Gaast A, van Lanschot JJ, van Hagen P, van Os R, van Rij CM, van der Sangen MJ, Beukema JC, Rütten H, Spruit PH, Reinders JG, Richel DJ, van Berge Henegouwen MI, Hulshof MC (2014) Patterns of recurrence after surgery alone versus preoperative chemoradiotherapy and surgery in the CROSS trials. J Clin Oncol 32:385–391. https://doi.org/10.1200/JCO.2013.51.2186
Yamashita K, Watanabe M, Mine S, Kurogochi T, Okamura A, Hayami M, Imamura Y (2017) Patterns and outcomes of recurrent esophageal cancer after curative esophagectomy. World J Surg 41:2337–2344. https://doi.org/10.1007/s00268-017-4024-5
Author information
Authors and Affiliations
Contributions
H. Daiko and A. Sakaki performed the study conception and design. A. Sakaki, K. Ishiyama, and D. Kurita acquired the data for this study from the prospectively maintained institutional database. A. Sakaki, J. Kanamori, and H. Daiko analyzed and interpreted the gathered data. This manuscript was drafted by A. Sakaki. H. Daiko, J. Kanamori, and J. Oguma critically revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics and consent
Written informed consent was obtained from all patients in this study, which was approved by the Institutional Review Board of the National Cancer Center Hospital. All treatment was performed according to the ethical standards laid down in the Declaration of Helsinki.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 27 kb)
Rights and permissions
About this article
Cite this article
Sakaki, A., Kanamori, J., Ishiyama, K. et al. Distribution of lymph node metastases in locally advanced adenocarcinomas of the esophagogastric junction (cT2-4): comparison between Siewert type I and selected Siewert type II tumors. Langenbecks Arch Surg 405, 509–519 (2020). https://doi.org/10.1007/s00423-020-01894-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-020-01894-z