Abstract
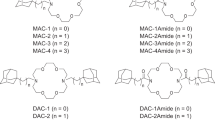
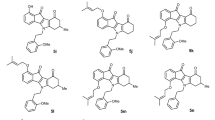
Overexpression of ABC transporters, such as ABCB1 and ABCG2, plays an important role in mediating multidrug resistance (MDR) in cancer. This feature is also attributed to a subpopulation of cancer stem cells (CSCs), having enhanced tumourigenic potential. ABCG2 is specifically associated with the CSC phenotype, making it a valuable target for eliminating aggressive and resistant cells. Several natural and synthetic ionophores have been discovered as CSC-selective drugs that may also have MDR-reversing ability, whereas their interaction with ABCG2 has not yet been explored. We previously reported the biological activities, including ABCB1 inhibition, of a group of adamantane-substituted diaza-18-crown-6 (DAC) compounds that possess ionophore capabilities. In this study, we investigated the mechanism of ABCG2-inhibitory activity of DAC compounds and the natural ionophores salinomycin, monensin and nigericin. We used a series of functional assays, including real-time microscopic analysis of ABCG2-mediated fluorescent substrate transport in cells, and docking studies to provide comparative aspects for the transporter–compound interactions and their role in restoring chemosensitivity. We found that natural ionophores did not inhibit ABCG2, suggesting that their CSC selectivity is likely mediated by other mechanisms. In contrast, DACs with amide linkage in the side arms demonstrated noteworthy ABCG2-inhibitory activity, with DAC-3Amide proving to be the most potent. This compound induced conformational changes of the transporter and likely binds to both Cavity 1 and the NBD–TMD interface. DAC-3Amide reversed ABCG2-mediated MDR in model cells, without affecting ABCG2 expression or localization. These results pave the way for the development of new crown ether compounds with improved ABCG2-inhibitory properties.








Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article (and its supplementary information files).
References
Alfonso I, Quesada R (2013) Biological activity of synthetic ionophores: ion transporters as prospective drugs? Chem Sci 4:3009–3019. https://doi.org/10.1039/c3sc50882j
An Y, Ongkeko WM (2009) ABCG2: the key to chemoresistance in cancer stem cells? Expert Opin Drug Metab Toxicol 5:1529–1542
Antoszczak M (2019) A medicinal chemistry perspective on salinomycin as a potent anticancer and anti-CSCs agent. Eur J Med Chem 164:366–377. https://doi.org/10.1016/j.ejmech.2018.12.057
Basok SS, Schepetkin IA, Khlebnikov AI et al (2021) Synthesis, biological evaluation, and molecular modeling of aza-crown ethers. Molecules 26:2225. https://doi.org/10.3390/molecules26082225
Begicevic RR, Falasca M (2017) ABC transporters in cancer stem cells: beyond chemoresistance. Int J Mol Sci. https://doi.org/10.3390/ijms18112362
Boesch M, Zeimet AG, Rumpold H et al (2015) Drug transporter-mediated protection of cancer stem cells from ionophore antibiotics. Stem Cells Transl Med 4:1028–1032. https://doi.org/10.5966/sctm.2015-0054
Boesch M, Sopper S, Wolf D (2016) Ionophore antibiotics as cancer stem cell-selective drugs: open questions. Oncologist 21:1291–1293. https://doi.org/10.1634/theoncologist.2016-0159
Borrel MN, Fiallo M, Veress I, Garnier-Suillerot A (1995) The effect of crown ethers, tetraalkylammonium salts, and polyoxyethylene amphiphiles on pirarubicin incorporation in K562 resistant cells. Biochem Pharm 50:2069–2076. https://doi.org/10.1016/0006-2952(95)02112-4
Dewangan J, Srivastava S, Rath SK (2017) Salinomycin: a new paradigm in cancer therapy. Tumor Biol. https://doi.org/10.1177/1010428317695035
Ding XW, Wu JH, Jiang CP (2010) ABCG2: a potential marker of stem cells and novel target in stem cell and cancer therapy. Life Sci 86:631–637. https://doi.org/10.1016/j.lfs.2010.02.012
Febles M, Montalvão S, Crespín GD et al (2016) Synthesis and biological evaluation of crown ether acyl derivatives. Bioorganic Med Chem Lett 26:5591–5593. https://doi.org/10.1016/j.bmcl.2016.09.066
Fuchs D, Daniel V, Sadeghi M et al (2010) Salinomycin overcomes ABC transporter-mediated multidrug and apoptosis resistance in human leukemia stem cell-like KG-1a cells. Biochem Biophys Res Commun 394:1098–1104. https://doi.org/10.1016/j.bbrc.2010.03.138
Gokel MR, McKeever M, Meisel JW et al (2021) Crown ethers having side arms: a diverse and versatile supramolecular chemistry. J Coord Chem 74:14–39
Gose T, Shafi T, Fukuda Y et al (2020) ABCG2 requires a single aromatic amino acid to “clamp” substrates and inhibitors into the binding pocket. FASEB J 34:4890–4903. https://doi.org/10.1096/fj.201902338RR
Guberović I, Marjanović M, Mioč M et al (2018) Crown ethers reverse P-glycoprotein-mediated multidrug resistance in cancer cells. Sci Rep. https://doi.org/10.1038/s41598-018-32770-y
Gupta PB, Onder TT, Jiang G et al (2009) Identification of selective inhibitors of cancer stem cells by high-throughput screening. Cell 138:645–659. https://doi.org/10.1016/j.cell.2009.06.034
Hasanabady MH, Kalalinia F (2016) ABCG2 inhibition as a therapeutic approach for overcoming multidrug resistance in cancer. J Biosci 41:313–324. https://doi.org/10.1007/s12038-016-9601-5
Homolya L, Orbán TI, Csanády L, Sarkadi B (2011) Mitoxantrone is expelled by the ABCG2 multidrug transporter directly from the plasma membrane. Biochim Biophys Acta Biomembr 1808:154–163. https://doi.org/10.1016/j.bbamem.2010.07.031
Humphrey W, Dalke A, Schulten K (1996) VMD: visual molecular dynamics. J Mol Graph 14:33–38. https://doi.org/10.1016/0263-7855(96)00018-5
Jackson SM, Manolaridis I, Kowal J et al (2018) Structural basis of small-molecule inhibition of human multidrug transporter ABCG2. Nat Struct Mol Biol 25:333–340. https://doi.org/10.1038/s41594-018-0049-1
Jiang ZS, Sun YZ, Wang SM, Ruan JS (2017) Epithelial–mesenchymal transition: potential regulator of ABC transporters in tumor progression. J Cancer 8:2319–2327. https://doi.org/10.7150/jca.19079
Kaushik V, Yakisich JS, Kumar A et al (2018) Ionophores: potential use as anticancer drugs and chemosensitizers. Cancers (Basel) 10:1–21. https://doi.org/10.3390/cancers10100360
Khunweeraphong N, Stockner T, Kuchler K (2017) The structure of the human ABC transporter ABCG2 reveals a novel mechanism for drug extrusion. Sci Rep 7:1–15. https://doi.org/10.1038/s41598-017-11794-w
Kralj M, Tušek-Božić L, Frkanec L (2008) Biomedical potentials of crown ethers: prospective antitumor agents. ChemMedChem. https://doi.org/10.1002/cmdc.200800118
Li W, Zhang H, Assaraf YG et al (2016) Overcoming ABC transporter-mediated multidrug resistance: molecular mechanisms and novel therapeutic drug strategies. Drug Resist Updat 27:14–29
Manolaridis I, Jackson SM, Taylor NMI et al (2018) Cryo-EM structures of a human ABCG2 mutant trapped in ATP-bound and substrate-bound states. Nature 563:426–430. https://doi.org/10.1038/s41586-018-0680-3
Marjanović M, Kralj M, Supek F et al (2007) Antitumor potential of crown ethers: structure–activity relationships, cell cycle disturbances, and cell death studies of a series of ionophores. J Med Chem. https://doi.org/10.1021/jm061162u
Markowska A, Kaysiewicz J, Markowska J, Huczyński A (2019) Doxycycline, salinomycin, monensin and ivermectin repositioned as cancer drugs. Bioorganic Med Chem Lett 29:1549–1554. https://doi.org/10.1016/j.bmcl.2019.04.045
Mlinarić-Majerski K, Šumanovac Ramljak T (2002) Synthesis and alkali metal binding properties of novel N-adamantylaza-crown ethers. Tetrahedron 58:4893–4898. https://doi.org/10.1016/S0040-4020(02)00444-1
Morris GM, Huey R, Lindstrom W et al (2009) AutoDock4 and autodocktools4: automated docking with selective receptor flexibility. J Comput Chem 30:2785–2791. https://doi.org/10.1002/jcc.21256
Mózner O, Bartos Z, Zámbó B et al (2019) Cellular processing of the ABCG2 transporter—potential effects on gout and drug metabolism. Cells 8:1215
Nicolle E, Boumendjel A, Macalou S et al (2009) QSAR analysis and molecular modeling of ABCG2-specific inhibitors. Adv Drug Deliv Rev 61:34–46. https://doi.org/10.1016/j.addr.2008.10.004
Orbán TI, Seres L, Özvegy-Laczka C et al (2008) Combined localization and real-time functional studies using a GFP-tagged ABCG2 multidrug transporter. Biochem Biophys Res Commun 367:667–673. https://doi.org/10.1016/j.bbrc.2007.12.172
Orlando BJ, Liao M (2020) ABCG2 transports anticancer drugs via a closed-to-open switch. Nat Commun 11:1–11. https://doi.org/10.1038/s41467-020-16155-2
Özvegy C, Litman T, Szakács G et al (2001) Functional characterization of the human multidrug transporter, ABCG2, expressed in insect cells. Biochem Biophys Res Commun 285:111–117. https://doi.org/10.1006/bbrc.2001.5130
Özvegy C, Varadi A, Sarkadi B (2002) Characterization of drug transport, ATP hydrolysis, and nucleotide trapping by the human ABCG2 multidrug transporter modulation of substrate specificity by a point mutation. J Biol Chem 277:47980–47990. https://doi.org/10.1074/jbc.M207857200
Özvegy-Laczka C, Várady G, Köblös G et al (2005) Function-dependent conformational changes of the ABCG2 multidrug transporter modify its interaction with a monoclonal antibody on the cell surface. J Biol Chem 280:4219–4227. https://doi.org/10.1074/jbc.M411338200
Peña-Solórzano D, Stark SA, König B et al (2017) ABCG2/BCRP: specific and nonspecific modulators. Med Res Rev 37:987–1050
Robey RW, Steadman K, Polgar O et al (2004) Pheophorbide A is a specific probe for ABCG2 function and inhibition. Cancer Res 64:1242–1246. https://doi.org/10.1158/0008-5472.CAN-03-3298
Robey RW, Pluchino KM, Hall MD et al (2018) Revisiting the role of ABC transporters in multidrug-resistant cancer. Nat Rev Cancer 18:452–464
Roussel E, Moréno A, Altounian N et al (2020) Chromones bearing amino acid residues: easily accessible and potent inhibitors of the breast cancer resistance protein ABCG2. Eur J Med Chem 202:112503. https://doi.org/10.1016/j.ejmech.2020.112503
Sarkadi B, Özvegy-Laczka C, Német K, Váradi A (2004) ABCG2—a transporter for all seasons. FEBS Lett 567:116–120. https://doi.org/10.1016/j.febslet.2004.03.123
Sarkadi B, Homolya L, Szakács G, Váradi A (2006) Human multidrug resistance ABCB and ABCG transporters: participation in a chemoimmunity defense system. Physiol Rev 86:1179–1236. https://doi.org/10.1152/physrev.00037.2005
Schrödinger Release 2022-1 (2021) Maestro, Schrödinger, LLC, New York, NY
Sehested M, Skovsgaard T, Roed H (1988) The carboxylic ionophore monensin inhibits active drug efflux and modulates in vitro resistance in daunorubicin resistant Ehrlich ascites tumor cells. Biochem Pharm 37:3305–3310. https://doi.org/10.1016/0006-2952(88)90643-0
Shibue T, Weinberg RA (2017) EMT, CSCs, and drug resistance: the mechanistic link and clinical implications. Nat Rev Clin Oncol 14:611–629. https://doi.org/10.1038/nrclinonc.2017.44
Singh A, Settleman J (2010) EMT cancer stem cells and drug resistance. Oncogene 29:4741–4751. https://doi.org/10.1038/onc.2010.215.EMT
Strope JD, Peer CJ, Sissung TM et al (2020) Botryllamide G is an ABCG2 inhibitor that improves lapatinib delivery in mouse brain. Cancer Biol Ther 21:223–230. https://doi.org/10.1080/15384047.2019.1683324
Šumanovac Ramljak T, Mlinarić-Majerski K, Bertoša B (2012) Alkali metal ion complexation of adamantane functionalized diaza-bibracchial lariat ethers. Croat Chem Acta 85:559–568. https://doi.org/10.5562/cca2100
Supek F, Ramljak TS, Marjanović M et al (2011) Could LogP be a principal determinant of biological activity in 18-crown-6 ethers? Synthesis of biologically active adamantane-substituted diaza-crowns. Eur J Med Chem. https://doi.org/10.1016/j.ejmech.2011.05.009
Taylor NMI, Manolaridis I, Jackson SM et al (2017) Structure of the human multidrug transporter ABCG2. Nature 546:504–509. https://doi.org/10.1038/nature22345
Telbisz Á, Müller M, Özvegy-Laczka C et al (2007) Membrane cholesterol selectively modulates the activity of the human ABCG2 multidrug transporter. Biochim Biophys Acta Biomembr 1768:2698–2713. https://doi.org/10.1016/j.bbamem.2007.06.026
Telbisz Á, Hegedüs C, Özvegy-Laczka C et al (2012) Antibody binding shift assay for rapid screening of drug interactions with the human ABCG2 multidrug transporter. Eur J Pharm Sci 45:101–109. https://doi.org/10.1016/j.ejps.2011.10.021
Telbisz Á, Ambrus C, Mózner O et al (2021) Interactions of potential anti-COVID-19 compounds with multispecific ABC and OATP drug transporters. Pharmaceutics 13:1–18. https://doi.org/10.3390/pharmaceutics13010081
Trott O, Olson AJ (2009) AutoDock vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J Comput Chem 31:455–461. https://doi.org/10.1002/jcc.21334
Ujhelly O, Özvegy C, Várady G et al (2003) Application of a human multidrug transporter (ABCG2) variant as selectable marker in gene transfer to progenitor cells. Hum Gene Ther 14:403–412. https://doi.org/10.1089/104303403321209005
Uzelac L, Škalamera Đ, Mlinarić-Majerski K et al (2017) Selective photocytotoxicity of anthrols on cancer stem-like cells: the effect of quinone methides or reactive oxygen species. Eur J Med Chem. https://doi.org/10.1016/j.ejmech.2017.05.063
Wang H, Zhang H, Zhu Y et al (2021) Anticancer mechanisms of salinomycin in breast cancer and its clinical applications. Front Oncol 11:2748
Waterhouse A, Bertoni M, Bienert S et al (2018) SWISS-MODEL: homology modelling of protein structures and complexes. Nucleic Acids Res 46:W296–W303. https://doi.org/10.1093/nar/gky427
Wei Y, Ma Y, Zhao Q et al (2012) New use for an old drug: Inhibiting ABCG2 with sorafenib. Mol Cancer Ther 11:1693–1702. https://doi.org/10.1158/1535-7163.MCT-12-0215
Wiese M (2015) BCRP/ABCG2 inhibitors: a patent review (2009–present). Expert Opin Ther Pat 25:1229–1237
Williams CJ, Headd JJ, Moriarty NW et al (2018) MolProbity: more and better reference data for improved all-atom structure validation. Protein Sci 27:293–315
Wu CP, Hsiao SH, Huang YH et al (2020) Sitravatinib sensitizes ABCB1-and ABCG2-overexpressing multidrug-resistant cancer cells to chemotherapeutic drugs. Cancers (Basel). https://doi.org/10.3390/cancers12010195
Yun Z, Lin Q (2014) Hypoxia and regulation of cancer cell stemness. Advances in experimental medicine and biology. Springer, New York LLC, pp 41–53
Acknowledgements
This work was financed by Croatian Science Foundation project number IP-2013-5660 (“MultiCaST”) to MK. The work was also supported by the FP7-REGPOT-2012-2013-1 project, grant agreement number 316289–InnoMol. MM received a financial support from the Ruđer Bošković Institute for a short-term secondment to the ELKH Research Centre for Natural Sciences, Budapest. We sincerely thank Lucija Horvat for wonderful assistance with confocal microscopy, Dr Marijana Popović Hadžija and Lucija Lulić for invaluable experimental help. TŠ is thankful to Prof. K. Majerski for her support and introduction to the field of crown ethers.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file2 (MP4 3051 KB)
Supplementary file3 (MP4 3558 KB)
Supplementary file4 (MP4 3108 KB)
Rights and permissions
About this article
Cite this article
Mioč, M., Telbisz, Á., Radman, K. et al. Interaction of crown ethers with the ABCG2 transporter and their implication for multidrug resistance reversal. Histochem Cell Biol 158, 261–277 (2022). https://doi.org/10.1007/s00418-022-02106-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-022-02106-z