Abstract
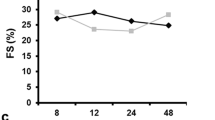
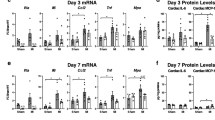
We sought to delineate mechanisms through which the lack of plasminogen activator inhibitor (PAI)-1 in the heart affects remodeling of the heart early after myocardial infarction (MI). MI was induced by coronary occlusion in 10-weeks old PAI-1 knockout (KO) and control mice. Three days after MI, systolic and diastolic function was assessed with high-resolution echocardiography, infarct size was determined biochemically and histologically and accumulation of acute inflammatory cells in zones of infarction was characterized by immunocytochemistry. PAI-1 KO mice exhibited markedly thickened diastolic left ventricular anterior walls (1.38 ± 0.38 mm vs. 0.77 ± 0.13 SD), more profound depression of global and regional cardiac function (19 vs. 22% fractional shortening), and greater evidence of diastolic dysfunction (average E wave amplitude = 568 vs. 675 mm/s) all of which were significant. Markedly greater extent of infarction was demonstrated biochemically and histologically in knockout mice compared with controls (76 vs. 29% of the left ventricle, P < 0.05) associated with striking hemorrhage and intense inflammation. Fibrosis normalized for infarct size was markedly reduced (0.006 vs. 0.022 μg hydroxyproline/mg dry weight). Thus, lack of PAI-1 in the heart exerted deleterious effects mediated, at least in part by increased inflammation and hemorrhage and attenuating of fibrosis.






Similar content being viewed by others
References
Anscher MS, Peters WP, Reisenbichler H, Petros WP, Jirtle RL (1993) Transforming growth factor beta as a predictor of liver and lung fibrosis after autologous bone marrow transplantation for advanced breast cancer. N Engl J Med 328:1592–1598
Askari AT, Brennan M-L, Zhou X, Drinki J, Morehead A, Thomas JD, Topol EJ, Hazen SL, Penn MS (2003) Myeloperoxidase and plasminogen activator inhibitor 1 play a central role in ventricular remodeling after myocardial infarction. J Exp Med 197:615–624
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Carmeliet P, Kieckens L, Schoonjans L, Ream B, van Nuffelen A, Prendergast G, Cole M, Bronson R, Collen D, Mullilgan RC (1993) Plasminogen activator inhibitor-1 gene-deficient mice. I. Generation by homologous recombination and characterization. J Clin Invest 92:2746–2755
Chen Y, Kelm RJ Jr, Budd RC, Sobel BE, Schneider DJ (2004) Inhibition of apoptosis and caspase-3 in vascular smooth muscle cells by plasminogen activator inhibitor type-1. J Cell Biochem 92:178–188
Chen Y, Budd RC, Kelm RJ Jr, Sobel BE, Schneider DJ. (2006) Augmentation of proliferation of vascular smooth muscle cells by plasminogen activator inhibitor type 1. Arterioscler Thromb Vasc Biol 26:1777–1783
Clough MH, Schneider DJ, Sobel BE, White MF, Wadsworth MP, Taatjes DJ (2005) Attenuation of accumulation of neointimal lipid by pioglitazone in mice genetically deficient in insulin receptor substrate-2 and apolipoprotein E. J Histochem Cytochem 53:603–610
Clowes AW, Clowes MM, Au YP, Reidy MA, Belin D (1990) Smooth muscle cells express urokinase during mitogenesis and tissue-type plasminogen activator during migration in injured rat carotid artery. Circ Res 67:61–67
Connors KF, Lamas GA (1995) Postmyocardial infarction patients: experience from the SAVE trial. Am J Crit Care 4:23–28
Donnan PT, Boyle DIR, Broomhall J, Hunter K, MacDonald TM, Newton RW, Morris AD (2002) Prognosis following first acute myocardial infarction in type 2 diabetes: a comparative population study. Diabetic Med 19:448–455
Eitzman DT, McCoy RD, Zheng X, Fay WP, Shen T, Ginsburg D, Simon RH (1996) Bleomycin-induced pulmonary fibrosis in transgenic mice that either lack or overexpress the murine plasminogen activator inhibitor-1 gene. J Clin Invest 98:232–237
Feener EP, Northrup JM, Aiello LP, King GL (1995) Angiotensin II induces plasminogen activator inhibitor-1 and -2 expression in vascular endothelial and smooth muscle cells. J Clin Invest 95:1353–1362
Fogo AB (2003) Renal fibrosis: not just PAI-1 in the sky. J Clin Invest 112:326–328
Folkes A, Brown SD, Canne LE, Chan J, Engelhardt E, Epshteyn S, Faint R, Golec J, Hanel A, Kearney P, Leahy JW, Mac M, Matthews D, Prisbylla MP, Sanderson J, Simon RJ, Tesfai Z, Vicker N, Wang S, Webb RR, Charlton P (2002) Design, synthesis and in vitro evaluation of potent, novel, small molecular inhibitors of plasminogen activator inhibitor-1. Bioorg Med Chem Lett 12:1063–1066
Gwilt DJ, Petri M, Lewis PW, Nattrass M, Pentecost BL (1985) Myocardial infarct size and mortality in diabetic patients. Br Heart J 54:466–472
Haider B, Yeh CK, Thomas G, Oldewurtel HA, Lyons MM, Regan TJ (1978) Altered myocardial function and collagen in diabetic rhesus monkeys on atherogenic diet. Trans Assoc Am Physicians 91:197–203
Hayashidani S, Tsutsui H, Shiomi T, Ikeuchi M, Matsusaka H, Suematsu N, Wen J, Egashira K, Takeshita A (2003) Anti-monocyte chemoattractant protein-1 gene therapy attenuates left ventricular remodeling and failure after experimental myocardial infarction. Circulation 108:2134–2140
Horrow JC, Van Riper DF, Strong MD, Brodsky I, Parmet JL (1991) Hemostatic effects of tranexamic acid and desmopressin during cardiac surgery. Circulation 84:2063–2070
Juhan-Vague I, Alessi MC, Mavri A, Morange PE (2003) Plasminogen activator inhibitor-1, inflammation, obesity, insulin resistance and vascular risk. J Thromb Haemost 1:1575–1579
Kaikita K, Fogo AB, Ma L, Schoenhard JA, Brown NJ, Vaughan DE (2001) Plasminogen activator inhibitor-1 deficiency prevents hypertension and vascular fibrosis in response to long-term nitric oxide synthase inhibition. Circulation 104:839–844
Keeton MR, Curriden SA, van Zonneveld A-J, Loskutoff DJ (1991) Identification of regulatory sequences in the type 1 plasminogen activator inhibitor gene responsive to transforming growth factor beta. J Biol Chem 266:23048–23052
Kenagy RD, Vesti BR, Clowes AW (2002) The urokinase receptor mediates basic fibroblast growth factor-dependent smooth muscle cell migration through baboon aortic explants. Atherosclerosis 162:63–67
Kivirikko KI, Laitinen O, Prockop DJ (1967) Modifications of a specific assay for hydroxyproline in urine. Anal Biochem 19:249–255
Kjekshus JK , Sobel BE (1970) Depressed myocardial creatine phosphokinase activity following experimental myocardial infarction in rabbit. Circ Res 27:403–414
Maroko PR, Kjekshus JK, Sobel BE, Watanabe T, Covell JW, Ross J Jr, Braunwald E (1971) Factors influencing infarct size following experimental coronary artery occlusions. Circulation 43:67–82
Matsushima S, Ide T, Yamato M, Matsusaka H, Hattori F, Ikeuchi M, Kubota T, Sunagawa K, Hasegawa Y, Kurihara T, Oikawa S, Kinugawa S, Tsutsui H (2006) Overexpression of Mitochondrial Peroxiredoxin-3 prevents left ventricular remodeling and failure after myocardial infarction in mice. Circulation 113:1779–1786
McGill JB, Schneider DJ, Arfken CL, Lucore CL, Sobel BE (1994) Factors responsible for impaired fibrinolysis in obese subjects and NIDDM patients. Diabetes 43:104–109
Mizushige K, Yao L, Noma T, Kiyomoto H, Yu Y, Hosomi N, Ohmori K, Matsuo H (2000) Alteration in left ventricular diastolic filling and accumulation of myocardial collagen at insulin-resistant prediabetic stage of a type II diabetic rat model. Circulation 101:899–907
Nagai N, DeMol M, Lijnen HR, Carmeliet P, Collen D (1999) Role of plasminogen system components in focal cerebral ischemic infarction. A gene targeting and gene transfer study in mice. Circulation 99:2440–2444
Nordt TK, Sawa H, Fujii S, Sobel BE (1995) Induction of plasminogen activator inhibitor type-1 (PAI-1) by proinsulin and insulin in vivo. Circulation 91:764–770
Oikawa T, Freeman M, Lo W, Vaughan DE, Fogo A (1997) Modulation of plasminogen activator inhibitor-1 in vivo: a new mechanism for the anti-fibrotic effect of rennin-angiotensin inhibition. Kidney Int 51:164–172
Olman MA, Mackman N, Gladson CL, Moser KM, Loskutoff DJ (1995) Changes in procoagulant and fibrinolytic gene expression during bleomycin-induced lung injury in the mouse. J Clin Invest 96:1621–1630
Perez JE, McGill JB, Santiago JV, Schechtman KB, Waggoner AD, Miller JG, Sobel BE (1992) Abnormal myocardial acoustic properties in diabetic patients and their correlation with the severity of disease. J Am Coll Cardiol 19:1154–1162
Renckens R, Roelofs JJTH, DeWaard V, Florquin S, Lijnen HR, Carmeliet P, van der Poll T (2005) The role of plasminogen activator inhibitor type 1 in the inflammatory response to local tissue injury. J Thromb Haemost 3:1018–1025
Roberts R, Henry PD, Sobel BE (1975) An improved basis for enzymatic estimation of infarct size. Circulation 52:743–754
Romer MU, Kirkebjerg Due A, Knud Larsen J, Hofland KF, Christensen IJ, Buhl-Jensen P, Almholt K, Lerberg Neilson O, Brunner N, Lademann U (2005) Indication of a role of plasminogen activator inhibitor type 1 in protecting murine fibrosarcoma cells against apoptosis. Thromb Haemost 94:859–866
Rosalki SB (1967) An improved procedure for serum creatine phosphokinase determination. J Lab Clin Med 69:696–705
Rossignol P, Angeles-Cano E, Lijnen HR (2006a) Plasminogen activator inhibitor-1 impairs plasminogen activation-mediated vascular smooth muscle cell apoptosis. Thromb Haemost 96:665–670
Rossignol P, Luttun A, Martin-Ventura JL, Lupu F, Carmeliet P, Collen D, Angles-Cano E, Lijnen HR (2006b) Plasminogen activation: a mediator of vascular smooth muscle cell apoptosis in atherosclerotic plaques. J Thromb Haemost 4:664–670
Schneider DJ, Hayes M, Wadsworth M, Taatjes H, Rincon M, Taatjes DJ, Sobel BE (2004) Attenuation of neointimal vascular smooth muscle cellularity in atheroma by plasminogen activator inhibitor type-1 (PAI-1). J Histochem Cytochem 52:1091–1099
Shell WE, Kjekshus JK, Sobel BE (1971) Quantitative assessment of the extent of myocardial infarction in the conscious dog by means of analysis of serial changes in serum creatine phosphokinase activity. J Clin Invest 50:2614–2626
Shell WE, Lavelle JF, Covell JW, Sobel BE (1973) Early estimation of myocardial damage in conscious dogs and patients with evolving acute myocardial infarction. J Clin Invest 52:2579–2590
Shimizu M, Umeda K, Sugihara N, Yoshio H, Ino H, Takeda R, Okada Y, Nakanishi I (1993) Collagen remodeling in myocardium of patients with diabetes. J Clin Pathol 46:32–36
Sobel BE (1999) The potential influence of insulin and plasminogen activator inhibitor type-1 on formulation of vulnerable atherosclerotic plaques associated with type 2 diabetes. Proc Assoc Am Physicians 111:313–318
Sobel BE, Lee Y-H, Prately RE, Schneider DJ (2006a) Increased plasminogen activator inhibitor type-1 (PAI-1) in the heart as a function of age. Life Sci 79:1600–1605
Sobel BE, Schneider DJ, Lee Y-H, Pratley RE (2006b) Insulin resistance increases PAI-1 in the heart. Biochem Biophys Res Commun 346:102–107
Sobel BE, Taatjes DJ, Schneider DJ (2003) Intramural plasminogen activator inhibitor type-1 and coronary atherosclerosis. Arterioscler Thromb Vasc Biol 23:1979–1989
Sobel BE, Woodcock-Mitchell J, Schneider DJ, Holt RE, Marutsuka K, Gold H (1998) Increased plasminogen activator inhibitor type-1 in coronary artery atherectomy specimens from type 2 diabetic compared with nondiabetic patients: a potential factor predisposing to thrombosis and its persistence. Circulation 97:2213–2221
Takeshita K, Hayashi M, Iino S, Kondo T, Inden Y, Iwase M, Kojima T, Hirai M, Ito M, Loskutoff DJ, Saito H, Murohara T, Yamamoto K (2004) Increased expression of plasminogen activator inhibitor-1 in cardiomyocytes contributes to cardiac fibrosis after myocardial infarction. Am J Pathol 164:449–456
Wadsworth MP, Sobel BE, Schneider DJ, Tra W, van Hirtum H, Taatjes DJ (2006) Quantitative analysis of atherosclerotic lesion composition in mice. Methods Mol Biol 319:137–152
Young ME, McNulty P, Taegtmeyer H (2002) Adaptation and maladaptation of the heart in diabetes: Part II: potential mechanisms. Circulation 105:1861–1870
Zaman AKMT, Fujii S, Goto D, Furumoto T, Mishima T, Nakai Y, Dong J, Imagawa S, Sobel E, Kitabatake A (2004) Salutary effects of attenuation of angiotensin II on coronary perivascular fibrosis associated with insulin resistance and obesity. J Mol Cell Cardiol 37:525–535
Zaman AKMT, Fujii S, Sawa H, Goto D, Ishimori N, Watano K, Kaneko T, Furumoto T, Sugawara T, Sakuma I, Kitabatake A, Sobel BE (2001) Angiotensin-converting enzyme inhibition attenuates hypofibrinolysis and reduces cardiac perivascular fibrosis in genetically obese diabetic mice. Circulation 103:3123–3128
Acknowledgments
The work was supported in part by the American Diabetes Association grant No. 1-04-RA-105 entitled “Intramural Plasminogen Activator Inhibitor Type-1 (PAI-1): A Determinant of Plaque Vulnerability Associated with Type 2 Diabetes” and by a grant from the Leducq Foundation (Transatlantic Networks of Excellence for Cardiovascular Research). The technical assistance of Patricia Baumann, Keara McElroy-Yaggy, Dagnija Neimane, and Marilyn Wadsworth and the secretarial assistance of Lori Dales are deeply appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zaman, A.K.M.T., Fujii, S., Schneider, D.J. et al. Deleterious effects of lack of cardiac PAI-1 after coronary occlusion in mice and their pathophysiologic determinants. Histochem Cell Biol 128, 135–145 (2007). https://doi.org/10.1007/s00418-007-0300-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-007-0300-z