Abstract
Purpose
To evaluate the correlation between contrast sensitivity (CS) and retinal nerve fiber layer (RNFL) in different areas of vision.
Methods
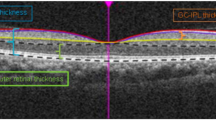
This report is a sub-analysis of a prospective, observational cohort study investigating changes in performance-based assessment, vision-related quality of life, and clinical measures in patients with moderate to advanced glaucoma. The study included 161 participants with at least a 2-year history of glaucoma who underwent annual testing for 4 years. Contrast sensitivity was measured using the Spaeth/Richman contrast sensitivity (SPARCS) test, while RNFL thickness (RNFLT) was measured using Cirrus optical coherence tomography (OCT). Statistical analyses were performed to determine correlations between CS and RNFLT; the correlations were calculated for each annual visit, totaling four correlation coefficients for each patient over the course of 4 years.
Results
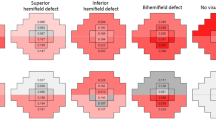
The SPARCS score in the left upper area of vision correlated the most strongly with the RNFLT of the inferior quadrant for both eyes at each annual visit, specifically in the seven o’clock sector for the left eye and the six o’clock sector for the right eye (p < 0.05). There were no discernible trends for the correlations between the other areas of CS and RNFL quadrants or clock hours over the 4 years of the study. Linear regression between the SPARCS total score and average RNFLT showed a significant direct correlation at each visit (p < 0.01).
Conclusions
Contrast sensitivity in the left upper area of vision for both eyes correlated most strongly with the thickness of the inferior quadrant of the RNFL. These fibers project to the temporal portion of the right occipital lobe, implying a potential center for contrast perception in this area. The longitudinal nature of the study suggests that CS may be a predictive tool for changes in RNFL in patients with glaucoma. Despite this finding, retinal damage and its relationship to CS was diffuse. In addition, SPARCS was shown to predict RNFLT. Further research is warranted to understand how CS can be used as a tool in the clinical setting.



Similar content being viewed by others
References
Garway-Heath DF, Poinoosawmy D, Fitzke FW, Hitchings RA (2000) Mapping the visual field to the optic disc in normal tension glaucoma eyes. Ophthalmology 107:1809–1815. https://doi.org/10.1016/S0161-6420(00)00284-0
Wollstein G, Kagemann L, Bilonick RA et al (2012) Retinal nerve fibre layer and visual function loss in glaucoma: the tipping point. Br J Ophthalmol 96:47–52. https://doi.org/10.1136/bjo.2010.196907
Malik R, Swanson WH, Garway-Heath DF (2012) “structure–function relationship” in glaucoma: past thinking and current concepts. Clin Experiment Ophthalmol 40:369–380. https://doi.org/10.1111/j.1442-9071.2012.02770.x
Harwerth RS, Wheat JL, Fredette MJ, Anderson DR (2010) Linking structure and function in glaucoma. Prog Retin Eye Res 29:249–271. https://doi.org/10.1016/j.preteyeres.2010.02.001
Ekici F, Loh R, Waisbourd M et al (2015) Relationships between measures of the ability to perform vision-related activities, vision-related quality of life, and clinical findings in patients with glaucoma. JAMA Ophthalmol 133:1–10. https://doi.org/10.1001/jamaophthalmol.2015.3426
Waisbourd M, Parker S, Ekici F et al (2015) A prospective, longitudinal, observational cohort study examining how glaucoma affects quality of life and visually-related function over 4 years: design and methodology. BMC Ophthalmol 15:91. https://doi.org/10.1186/s12886-015-0088-x
Hu CX, Zangalli C, Hsieh M et al (2014) What do patients with glaucoma see? Visual symptoms reported by patients with glaucoma. Am J Med Sci 348:403–409. https://doi.org/10.1097/MAJ.0000000000000319
Crabb DP, Smith ND, Glen FC et al (2013) How does glaucoma look?: patient perception of visual field loss. Ophthalmology 120:1120–1126. https://doi.org/10.1016/j.ophtha.2012.11.043
Hawkins AS, Szlyk JP, Ardickas Z et al (2003) Comparison of contrast sensitivity, visual acuity, and Humphrey visual field testing in patients with glaucoma. J Glaucoma 12:134–138
Breton ME, Wilson TW, Wilson R et al (1991) Temporal contrast sensitivity loss in primary open-angle glaucoma and glaucoma suspects. Invest Ophthalmol Vis Sci 32:2931–2941
Richman J, Lorenzana LL, Lankaranian D et al (2010) Importance of visual acuity and contrast sensitivity in patients with glaucoma. Arch Ophthalmol 128:1576–1582. https://doi.org/10.1001/archophthalmol.2010.275
Burton R, Crabb DP, Smith ND et al (2012) Glaucoma and reading: exploring the effects of contrast lowering of text. Optom Vis Sci 89:1282–1287. https://doi.org/10.1097/OPX.0b013e3182686165
Richman J, Spaeth GL, Wirostko B (2013) Contrast sensitivity basics and a critique of currently available tests. J Cataract Refract Surg 39:1100–1106. https://doi.org/10.1016/j.jcrs.2013.05.001
Kingsnorth A, Drew T, Grewal B, Wolffsohn JS (2016) Mobile app Aston contrast sensitivity test. Clin Exp Optom 99(4):350–355. https://doi.org/10.1111/cxo.12362
Rodriguez-Vallejo M, Remon L, Monsoriu JA, Furlan WD (2015) Designing a new test for contrast sensitivity function measurement with iPad. J Optom 8:101–108. https://doi.org/10.1016/j.optom.2014.06.003
Richman J, Zangalli C, Lu L et al (2014) The Spaeth/Richman contrast sensitivity test (SPARCS): design, reproducibility and ability to identify patients with glaucoma. Br J Ophthalmol 99(1):16–20. https://doi.org/10.1136/bjophthalmol-2014-305223
Sun Y, Erdem E, Lyu A, et al (2016) The SPARCS: a novel assessment of contrast sensitivity and its reliability in patients with corrected refractive error. Br J Ophthalmol 100(10):1421–1426. doi: https://doi.org/10.1136/bjophthalmol-2015-307378
Mwanza JC, Oakley JD, Budenz DL, Anderson DR (2011) Ability of cirrus HD-OCT optic nerve head parameters to discriminate normal from glaucomatous eyes. Ophthalmology 118:241–248. https://doi.org/10.1016/j.ophtha.2010.06.036
Shin HY, Park HYL, Jung Y et al (2014) Glaucoma diagnostic accuracy of optical coherence tomography parameters in early glaucoma with different types of optic disc damage. Ophthalmology 121:1990–1997. https://doi.org/10.1016/j.ophtha.2014.04.030
Hood DC, De Cuir N, Blumberg DM et al (2016) A single wide-field OCT protocol can provide compelling information for the diagnosis of early glaucoma. Transl Vis Sci Technol 5:4. https://doi.org/10.1167/tvst.5.6.4
Harris PA, Taylor R, Thielke R et al (2009) Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform 42:377–381. https://doi.org/10.1016/j.jbi.2008.08.010
R Development Core Team R (2011) R: a language and environment for statistical computing. R Found Stat Comput. https://doi.org/10.1007/978-3-540-74686-7
Law SK, Choe R, Caprioli J (2001) Optic disk characteristics before the occurrence of disk hemorrhage in glaucoma patients. Am J Ophthalmol 132:411–413. https://doi.org/10.1016/S0002-9394(01)01009-1
Lloyd MJ, Mansberger SL, Fortune BA et al (2013) Features of optic disc progression in patients with ocular hypertension and early glaucoma. J Glaucoma 22:343–348. https://doi.org/10.1097/IJG.0b013e31824c9251
Ou Y, Jo RE, Ullian EM et al (2016) Selective vulnerability of specific retinal ganglion cell types and synapses after transient ocular hypertension. J Neurosci 36:9240–9252. https://doi.org/10.1523/JNEUROSCI.0940-16.2016
Mwanza JC, Durbin MK, Budenz DL et al (2012) Glaucoma diagnostic accuracy of ganglion cell-inner plexiform layer thickness: comparison with nerve fiber layer and optic nerve head. Ophthalmology 119:1151–1158. https://doi.org/10.1016/j.ophtha.2011.12.014
Lenoble Q, Lek JJ, McKendrick AM (2016) Visual object categorisation in people with glaucoma. Br J Ophthalmol 100(11):1585-1590. doi: https://doi.org/10.1136/bjophthalmol-2015-308289
Tatham AJ, Boer ER, Rosen PN et al (2014) Glaucomatous retinal nerve fiber layer thickness loss is associated with slower reaction times under a divided attention task. Am J Ophthalmol 158:1008–1017. https://doi.org/10.1016/j.ajo.2014.07.028
Acknowledgements
We are grateful to both David Garway-Heath, MD (Moorsfields Eye Hospital, London, UK), who served as a scientific adviser and created the image we used for Fig. 3, and Michael Waisbourd, MD (Tel Aviv Sourasky Medical Center, Tel Aviv, Israel), who also served as a scientific advisor.
Funding
Merck Sharp & Dohme provided financial support in the form of a grant (IISP # 39138). The sponsor had no role in the design or conduct of this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The primary investigator, George L. Spaeth, has developed and patented the Spaeth/Richman Contrast Sensitivity test. The remaining authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge, or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants in the study.
Rights and permissions
About this article
Cite this article
Amanullah, S., Okudolo, J., Rahmatnejad, K. et al. The relationship between contrast sensitivity and retinal nerve fiber layer thickness in patients with glaucoma. Graefes Arch Clin Exp Ophthalmol 255, 2415–2422 (2017). https://doi.org/10.1007/s00417-017-3789-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-017-3789-4