Abstract
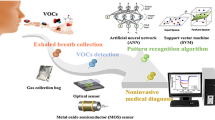
Electronic noses (e-noses) are based on arrays of different sensor types that respond to specific features of an odorant molecule, mostly volatile organic compounds (VOCs). Differently from gas chromatography and mass spectrometry, e-noses can distinguish VOCs spectrum by pattern recognition. E-nose technology has successfully been used in commercial applications, including military, environmental, and food industry. Human-exhaled breath contains a mixture of over 3000 VOCs, which offers the postulate that e-nose technology can have medical applications. Based on the above hypothesis, an increasing number of studies have shown that breath profiling by e-nose could play a role in the diagnosis and/or screening of various respiratory and systemic diseases. The aim of the present study was to review the principal literature on the application of e-nose technology in respiratory diseases.



Similar content being viewed by others
References
Pauling L, Robinson AB, Teranishi R, Cary P (1971) Quantitative analysis of urine vapor and breath by gas-liquid partition chromatography. Proc Natl Acad Sci USA 68:2374–2376
Moser B, Bodrogi F, Eibl G et al (2005) Mass spectrometric profile of exhaled breath. Field study by PTR-MS. Respir Physiol Neurobiol 145:295–300
Phillips M, Gleeson K, Hughes JM et al (1999) Volatile organic compounds in breath as markers of lung cancer: a cross-sectional study. Lancet 353:1930–1933
Phillips M, Altorki N, Austin JHM et al (2007) Prediction of lung cancer using volatile biomarkers in breath. Cancer Biomark 3:95–109
Poli D, Carbognani P, Corrdai M et al (2005) Exhaled volatile organic compounds in patients with non-small cell lung cancer: cross sectional and nested short-term follow-up study. Respir Res 6:71
Song G, Qin T, Liu H et al (2010) Quantitative breath analysis of volatile organic compounds of lung cancer patients. Lung Cancer 67:227–231
Dragonieri S, Schot R, Mertens JA et al (2007) An electronic nose in the discrimination of patients with asthma and controls. J Allergy Clin Immunol 120:856–862
Montuschi P, Santonico M, Penazza G et al (2010) Diagnostic performance of an electronic nose, fractional exhaled nitric oxide and lung function testing in asthma. Chest 137:790–796
Bennet L, Ciaffoni L, Denzer W et al (2009) A chemometric study on human breath mass spectra for biomarker identification in cystic fibrosis. J Breath Res 3:046002
Van Berkel JJ, Dallinga JW, Moller GM et al (2010) A profile of volatile organic compounds in breath discriminates COPD patients from controls. Respir Med 104:557–563
Persaud K, Dodd G (1982) Analysis of discrimination mechanisms in the mammalian olfactory system using a model nose. Nature 299:352
Buck L, Axel R (1991) A novel multigene family may encode odorant receptors: a molecular basis for odor recognition. Cell 65:175–187
Röck F, Barsan N, Weimar U (2008) Electronic nose: current status and future trends. Chem Rev 108:705–725
Wilson AD, Baietto M (2011). Advances in electronic-nose technologies developed for biomedical applications. Sensors 11:1105–1176.
Broadhurst DI, Kjell DB (2006) Statistical strategies for avoiding false discoveries in metabolomics and related experiments. Metabolomics 2:171–196
Fens N, Zwinderman AH, van der Schee MP et al (2009) Exhaled breath profiling enables discrimination of chronic obstructive pulmonary disease and asthma. Am J Respir Crit Care Med 180:1076–1082
Fens N, Roldaan AC, van der Schee MP et al (2011) External validation of exhaled breath profiling using an electronic nose in the discrimination of asthma with fixed airways obstruction and chronic obstructive pulmonary disease. Clin Exp Allergy 41:1371–1378
Wagener A, Brinkman P, Zwinderman A, et al (2013). Exhaled breath profiling and eosinophilic airway inflammation in asthma—results of a pilot study. Am J Respir Crit Care Med. 187:A2392
Plaza V, Crespo A, Giner J et al (2015) Inflammatory asthma phenotype discrimination using an electronic nose breath analyzer. J Investig Allergol Clin Immunol 25:431–437
van der Schee MP, Palmay R, Cowan JO et al (2013) Predicting steroid responsiveness in patients with asthma using exhaled breath profiling. Clin Exp Allergy 43:1217–1225
de Vries R, Brinkman P, van der Schee MP et al (2015) Integration of electronic nose technology with spirometry: validation of a new approach for exhaled breath analysis. J Breath Res 9:046001
Cosio MG, Saetta M, Agusti A (2009) Immunologic aspects of chronic obstructive pulmonary disease. N Engl J Med 360:2445–2454
Sibila O, Garcia-Bellmunt L, Giner J et al (2014) Identification of airway bacterial colonization by an electronic nose in chronic obstructive pulmonary disease. Respir Med 108:1608–1614
Hattesohl AD, Jörres RA, Dressel H et al (2011) Discrimination between COPD patients with and without alpha 1-antitrypsin deficiency using an electronic nose. Respirology 16:1258–1264
Dragonieri S, Annema JT, Schot R et al (2009) An electronic nose in the discrimination of patients with lung cancer and COPD. Lung Cancer 64:166–170
Fens N, de Nijs SB, Peters S et al (2011) Exhaled air molecular profiling in relation to inflammatory subtype and activity in COPD. Eur Respir J 38:1301–1309
Fens N, van Rossum AG, Zanen P et al (2013) Subphenotypes of mild-to-moderate COPD by factor and cluster analysis of pulmonary function, CT imaging and breathomics in a population-based survey. COPD 10:277–285
Shafiek H, Fiorentino F, Merino JL et al (2015) Using the electronic nose to identify airway infection during COPD exacerbations. PLoS ONE 10:e0135199
van Geffen WH, Bruins M, Kerstjens HA (2016) Diagnosing viral and bacterial respiratory infections in acute COPD exacerbations by an electronic nose: a pilot study. J Breath Res 10:036001
Marciniuk DD, Hernandez P, Balter M et al (2012) Alpha-1 antitrypsin deficiency targeted testing and augmentation therapy: a Canadian Thoracic Society clinical practice guideline. Can Respir J 19:109–116
Pavlou A, Turner AP, Magan N (2002) Recognition of anaerobic bacterial isolates in vitro using electronic nose technology. Lett Appl Microbiol 35:366–369
Pavlou AK, Magan N, Jones JM et al (2004) Detection of Mycobacterium tuberculosis (TB) in vitro and in situ using an electronic nose in combination with a neural network system. Biosens Bioelectron 20:538–544
Lai SY, Deffenderfer OF, Hanson W et al (2002) Identification of upper respiratory bacterial pathogens with the electronic nose. Laryngoscope 112:975–979
Dutta R, Hines EL, Gardner JW et al (2002) Bacteria classification using Cyranose 320 electronic nose. Biomed Eng Online 16:1–4
van der Schee MP, Fens N, Buijze H et al (2012) Diagnostic value of exhaled breath analysis in tuberculosis. Am J Respir Crit Care Med 185:a6510
Nakhleh MK, Jeries R, Gharra A et al (2014) Detecting active pulmonary tuberculosis with a breath test using nanomaterial-based sensors. Eur Respir J 43:1522–1525
Thaler ER, Hanson CW (2005). Medical applications of electronic nose technology. Exert Rev Med Devices 2:559–566
Hanson CW, Thaler ER (2005) Electronic nose prediction of a clinical pneumonia score: biosensors and microbes. Anesthesiology 102:63–68
Hockstein NG, Thaler ER, Torigian D et al (2004) Diagnosis of pneumonia with an electronic nose: correlation of vapor signature with chest computed tomography scan findings. Laryngoscope 114:1701–1705
Bos LD, Martin-Loeches I, Kastelijn JB et al (2014) The volatile metabolic fingerprint of ventilator-associated pneumonia. Intensive Care Med 40:761–762
Schnabel RM, Boumans ML, Smolinska A et al (2015) Electronic nose analysis of exhaled breath to diagnose ventilator-associated pneumonia. Respir Med 109:1454–1459
de Heer K, Kok MG, Fens N et al (2016) Detection of airway colonization by Aspergillus fumigatus by use of electronic nose technology in patients with cystic fibrosis. J Clin Microbiol 54:569–575
de Heer K, van der Schee MP, Zwinderman K et al (2013) Electronic nose technology for detection of invasive pulmonary aspergillosis in prolonged chemotherapy-induced neutropenia: a proof-of-principle study. J Clin Microbiol 5:1490–1495
Di Natale C, Macagnano A, Martinelli E et al (2003) Lung cancer identification by the analysis of breath by means of an array of non-selective gas sensors. Biosens Bioelectron 18:1209–1218
Santonico M, Lucantoni G, Pennazza G et al (2012) In situ detection of lung cancer volatile fingerprints using bronchoscopic air-sampling. Lung Cancer 77:46–50
D’Amico A, Pennazza G, Santonico M et al (2010) An investigation on electronic nose diagnosis of lung cancer. Lung Cancer 68:170–176
Gasparri R, Santonico M, Valentini C et al (2016) Volatile signature for the early diagnosis of lung cancer. J Breath Res 10:016007
Mazzone PJ, Hammel J, Dweik R et al (2007) Diagnosis of lung cancer by the analysis of exhaled breath with a colorimetric sensor array. Thorax 62:565–568
Machado RF, Laskowski O, Deffenderfer O et al (2005) Detection of lung cancer by sensor array analysis of exhaled breath. Am J Respir Crit Care Med 171:1286–1291
Bikov A, Hernadi M, Korosi BZ et al (2014) Expiratory flow rate, breath hold and anatomic dead space influence electronic nose ability to detect lung cancer. BMC Pulm Med 14:202
Hubers AJ, Brinkman P, Boksem RJ et al (2014) Combined sputum hypermethylation and eNose analysis for lung cancer diagnosis. J Clin Pathol 67:707–711
Chen X, Cao M, Hao Y et al (2005) A Non-invasive detection of lung cancer combined virtual gas sensors array with imaging recognition technique. Conf Proc IEEE Eng Med Biol Soc 6:5873–5876
Peng G, Tisch U, Adams O et al (2009) Diagnosing lung cancer in exhaled breath using gold nanoparticles. Nat Nanotechnol 4:669–673
Broza YY, Kremer R, Tisch U et al (2013) A nanomaterial-based breath test for short-term follow-up after lung tumor resection. Nanomedicine 9:15–21
Dragonieri S, van der Schee MP, Massaro T et al (2012) An electronic nose distinguishes exhaled breath of patients with malignant pleural mesothelioma from controls. Lung Cancer 75:326–331
Chapman EA, Thomas PS, Stone E et al (2012) A breath test for malignant mesothelioma using an electronic nose. Eur Resp J 40:448–454
Greulich T, Hattesohl A, Grabisch A et al (2013) Detection of obstructive sleep apnoea by an electronic nose. Eur Respir J 42:145–155
Benedek P, Lázár Z, Bikov A et al (2013) Exhaled biomarker pattern is altered in children with obstructive sleep apnoea syndrome. Int J Pediatr Otorhinolaryngol 77:1244–1247
Kunos L, Bikov A, Lazar Z et al (2015) Evening and morning exhaled volatile compound patterns are different in obstructive sleep apnoea assessed with electronic nose. Sleep Breath 19:247–253
Dragonieri S, Porcelli F, Longobardi F et al (2015) An electronic nose in the discrimination of obese patients with and without obstructive sleep apnoea. J Breath Res 9:026005
Dragonieri S, Quaranta V, Carratu P et al (2016) Exhaled breath profiling in patients with COPD and OSA overlap syndrome: a pilot study. J Breath Res 3(10):041001
Antonelli Incalzi R, Pennazza G, Scarlata S et al (2015) Comorbidity modulates non invasive ventilation-induced changes in breath print of obstructive sleep apnea syndrome patients. Sleep Breath 19:623–630
Paff T, van der Schee MP, Daniels JM et al (2013) Exhaled molecular profiles in the assessment of cystic fibrosis and primary ciliary dyskinesia. J Cyst Fibros 12:454–460
Joensen O, Paff T, Haarman EG et al (2014) Exhaled breath analysis using electronic nose in cystic fibrosis and primary ciliary dyskinesia patients with chronic pulmonary infections. PLoS ONE 9:e115584
Kovacs D, Bikov A, Losonczy G et al (2013) Follow up of lung transplant recipients using an electronic nose. J Breath Res 7:017117
Fens N, Douma RA, Sterk PJ et al (2010) Breathomics as a diagnostic tool for pulmonary embolism. J Thromb Haemost 8:2831–2833
Cohen-Kaminsky S, Nakhleh M, Perros F et al (2013) A proof of concept for the detection and classification of pulmonary arterial hypertension through breath analysis with a sensor array. Am J Respir Crit Care Med 188:756–759
Dragonieri S, Brinkman P, Mouw E et al (2013) An electronic nose discriminates exhaled breath of patients with untreated pulmonary sarcoidosis from controls. Respir Med 107:1073–1078
Bos LD, Schultz MJ, Sterk PJ (2014) Exhaled breath profiling for diagnosing acute respiratory distress syndrome. BMC Pulm Med 14:72
van der Schee MP, Paff T, Brinkman P et al (2015) Breathomics in lung disease. Chest 147:224–231
Bikov A, Lázár Z, Horvath I (2015) Established methodological issues in electronic nose research: how far are we from using these instruments in clinical settings of breath analysis? J Breath Res 9:034001
Scarlata S, Pennazza G, Santonico M et al (2015) Exhaled breath analysis by electronic nose in respiratory diseases. Expert Rev Mol Diagn 15:933–956
van der Schee MP, Fens N, Brinkman P et al (2013) Effect of transportation and storage using sorbent tubes of exhaled breath samples on diagnostic accuracy of electronic nose analysis. J Breath Res 7:016002
Incalzi RA, Pennazza G, Scarlata S et al (2012) Reproducibility and respiratory function correlates of exhaled breath fingerprint in chronic obstructive pulmonary disease. PLoS ONE 7:e45396
Nake A, Dubreuil B, Raynaud C, et al (2005). Outdoor in situ monitoring of volatile emissions from wastewater treatment plants with two portable technologies of electronic noses. Sens Actuators 106:36–39
Meka VV, Lutz BJ, Melker RJ et al (2007) Prototype of a breath-based analysis system for medication compliance monitoring. J Breath Res 1:026006
Lazar Z, Fens N, van der Maten J et al (2010) Electronic nose breathprints are independent of acute changes in airway caliber in asthma. Sensors (Basel) 10:9127–9138
Dragonieri S, Quaranta VN, Carratu P et al (2016) Influence of age and gender on the profile of exhaled volatile organic compounds analyzed by an electronic nose. J Bras Pneumol 42:143–145
Bikov A, Lazar Z, Schandl K et al (2011) Exercise changes volatiles in exhaled breath assessed by an electronic nose. Acta Physiol Hung 98:321–328
Bikov A, Pako J, Kovacs D et al (2011) Exhaled breath volatile alterations in pregnancy assessed with electronic nose. Biomarkers 16:476–484
Kischkel S, Miekisch W, Sawacki A et al (2010) Breath biomarkers for lung cancer detection and assessment of smoking related effects-confounding variables, influence of normalization and statistical algorithms. Clin Chim Acta 411:1637–1644
Bossuyt PM, Reitsma JB, Bruns DE et al (2003) Towards complete and accurate reporting of studies of diagnostic accuracy: the STARD initiative. Ann Clin Biochem 40:357–363
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Ethical Approval
All procedures performed in studies involving humans were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Informed Consent
An informed consent was signed by all the participants of the studies cited in the present review.
Rights and permissions
About this article
Cite this article
Dragonieri, S., Pennazza, G., Carratu, P. et al. Electronic Nose Technology in Respiratory Diseases. Lung 195, 157–165 (2017). https://doi.org/10.1007/s00408-017-9987-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-017-9987-3