Abstract
Purpose
To compare the effectiveness of intravenous carbetocin to that of intravenous oxytocin for prevention of atonic postpartum hemorrhage (PPH) after vaginal delivery in high-risk singleton pregnancies.
Methods
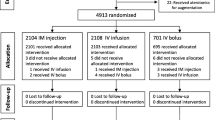
This triple-blind randomized controlled trial included singleton pregnant women who delivered at Siriraj Hospital between August 2016 and January 2017 and who were 20 years or older, had a gestational age of at least 34 weeks, had a vaginal delivery, and had at least one risk factor for atonic postpartum hemorrhage. Immediately after vaginal delivery, participants were randomly assigned to receive either 5 U of oxytocin or 100 mcg of carbetocin intravenously. Postpartum blood loss was measured objectively in mL using a postpartum drape with a calibrated bag.
Results
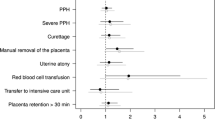
A total of 174 and 176 participants constituted the oxytocin and carbetocin groups, respectively. The baseline characteristics were comparable between the groups. The carbetocin group had less postpartum blood loss (146.7 ± 90.4 vs. 195.1 ± 146.2 mL; p < 0.01), a lower incidence of atonic PPH (0 vs. 6.3%; p < 0.01), less usage of additional uterotonic drugs (9.1 vs. 27.6%; p < 0.01), and a lower incidence of postpartum anemia (Hb ≤ 10 g/dL) (9.1 vs. 18.4%; p < 0.05) than the oxytocin group. No significant differences regarding side effects were evident between the groups.
Conclusions
Intravenous carbetocin is more effective than intravenous oxytocin for the prevention of atonic PPH among singleton pregnancies with at least one risk factor for PPH.
Clinical trial registration
TCTR20160715004.


Similar content being viewed by others
References
Say L, Chou D, Gemmill A, Tuncalp O, Moller AB, Daniels J, Gulmezoglu AM, Temmerman M, Alkema L (2014) Global causes of maternal death: a WHO systematic analysis. Lancet Glob Health 2(6):e323–e333
World Health Organization (2012) WHO recommendations for the prevention and treatment of postpartum haemorrhage. WHO Guidelines Approved by the Guidelines Review Committee, Geneva
Cordovani D, Carvalho JCA, Boucher M, Farine D (2012) Carbetocin for the prevention of postpartum hemorrhage. In: Arulkumaran S, Karoshi M, Keith LG, Lalonde AB, B-Lynch C (eds) A Comprehensive textbook of postpartum hemorrhage: an essential clinical reference for effective management, 2nd edn. Sapiens Publishing, London, pp 361–368
Westhoff G, Cotter AM, Tolosa JE (2013) Prophylactic oxytocin for the third stage of labour to prevent postpartum haemorrhage. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD001808.pub2
Meshykhi LS, Nel MR, Lucas DN (2016) The role of carbetocin in the prevention and management of postpartum haemorrhage. Int J Obstet Anesth 28:61–69
Samimi M, Imani-Harsini A, Abedzadeh-Kalahroudi M (2013) Carbetocin vs. syntometrine in prevention of postpartum hemorrhage: a double blind randomized control trial. Iran Red Crescent Med J 15(9):817–822
Bruyere M, Ait Hamou N, Benhamou D, Chousterman B, Boulard V, Charbit B (2014) QT interval prolongation following carbetocin in prevention of post-cesarean delivery hemorrhage. Int J Obstet Anesth 23(1):88–89
Jacquenod P, Cattenoz M, Canu G, Bois E, Lieutaud T (2015) Acute coronary syndrome following a 100 microg carbetocin injection during an emergency Cesarean delivery. Can J Anaesth 62(5):513–517
Su LL, Chong YS, Samuel M (2012) Carbetocin for preventing postpartum haemorrhage. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD005457.pub4
Leduc D, Senikas V, Lalonde AB, Ballerman C, Biringer A, Delaney M, Clinical Practice Obstetrics Committee; Society of Obstetricians and Gynaecologists of Canada et al (2009) Active management of the third stage of labour: prevention and treatment of postpartum hemorrhage. J Obstet Gynaecol Can 31(10):980–993
Maged AM, Hassan AM, Shehata NA (2016) Carbetocin versus oxytocin for prevention of postpartum hemorrhage after vaginal delivery in high risk women. J Matern Fetal Neonatal Med 29(4):532–536
Gulmezoglu AM, Lumbiganon P, Landoulsi S, Widmer M, Abdel-Aleem H, Festin M, Carroli G, Qureshi Z, Souza JP, Bergel E, Piaggio G, Goudar SS, Yeh J, Armbruster D, Singata M, Pelaez-Crisologo C, Althabe F, Sekweyama P, Hofmeyr J, Stanton ME, Derman R, Elbourne D (2012) Active management of the third stage of labour with and without controlled cord traction: a randomised, controlled, non-inferiority trial. Lancet 379(9827):1721–1727
Hunter DJ, Schulz P, Wassenaar W (1992) Effect of carbetocin, a long-acting oxytocin analog on the postpartum uterus. Clin Pharmacol Ther 52(1):60–67
Amsalem H, Aldrich CJ, Oskamp M, Windrim R, Farine D (2014) Postpartum uterine response to oxytocin and carbetocin. J Reprod Med 59(3–4):167–173
Silcox J, Schulz P, Horbay GL, Wassenaar W (1993) Transfer of carbetocin into human breast milk. Obstet Gynecol 82(3):456–459
Rosseland LA, Hauge TH, Grindheim G, Stubhaug A, Langesaeter E (2013) Changes in blood pressure and cardiac output during cesarean delivery: the effects of oxytocin and carbetocin compared with placebo. Anesthesiology 119(3):541–551
Boucher M, Nimrod CA, Tawagi GF, Meeker TA, Rennicks White RE, Varin J (2004) Comparison of carbetocin and oxytocin for the prevention of postpartum hemorrhage following vaginal delivery: a double-blind randomized trial. J Obstet Gynaecol Can 26(5):481–488
Askar AA, Ismail MT, El-Ezz AA, Rabie NH (2011) Carbetocin versus syntometrine in the management of third stage of labor following vaginal delivery. Arch Gynecol Obstet 284:1359–1365
Jin B, Du Y, Zhang F, Zhang K, Wang L, Cui L (2016) Carbetocin for the prevention of postpartum hemorrhage: a systematic review and meta-analysis of randomized controlled trials. J Matern Fetal Neonatal Med 29(3):400–407
Widmer M, Piaggio G, Abdel-Aleem H, Carroli G, Chong YS, Coomarasamy A, Fawole B, Goudar S, Hofmeyr GJ, Lumbiganon P, Mugerwa K, Nguyen TM, Qureshi Z, Souza JP, Gulmezoglu AM (2016) Room temperature stable carbetocin for the prevention of postpartum haemorrhage during the third stage of labour in women delivering vaginally: study protocol for a randomized controlled trial. Trials 17(1):143
(2017) Prevention and management of postpartum haemorrhage: green-top guideline no. 52. BJOG 124(5):e106–e149. https://doi.org/10.1111/1471-0528.14178
Snelgrove JW (2009) Postpartum haemorrhage in the developing world a review of clinical management strategies. Mcgill J Med 12(2):61
Su LL, Rauff M, Chan YH, Mohamad Suphan N, Lau TP, Biswas A, Chong YS (2009) Carbetocin versus syntometrine for the third stage of labour following vaginal delivery—a double-blind randomised controlled trial. BJOG 116(11):1461–1466
Chen CY, Su YN, Lin TH, Chang Y, Horng HC, Wang PH, Yeh CC, Chang WH, Huang HY (2016) Carbetocin in prevention of postpartum hemorrhage: experience in a tertiary medical center of Taiwan. Taiwan J Obstet Gynecol 55(6):804–809
Lertbunnaphong T, Lapthanapat N, Leetheeragul J, Hakularb P, Ownon A (2016) Postpartum blood loss: visual estimation versus objective quantification with a novel birthing drape. Singap Med J 57(6):325–328
Luni Y, Borakati A, Matah A, Skeats K, Eedarapalli P (2017) A prospective cohort study evaluating the cost-effectiveness of carbetocin for prevention of postpartum haemorrhage in caesarean sections. J Obstet Gynaecol 37(5):601–604
van der Nelson HA, Draycott T, Siassakos D, Yau CW, Hatswell AJ (2017) Carbetocin versus oxytocin for prevention of post-partum haemorrhage at caesarean section in the United Kingdom: an economic impact analysis. Eur J Obstet Gynecol Reprod Biol 210:286–291
Acknowledgements
The authors gratefully acknowledge Mr. Suthipol Udompunthurak for assistance with the statistical analysis. We are also grateful to a grant obtained from the Siriraj Research Development Fund, which is managed by the Routine to Research Project (R2R) and to 100 doses of carbetocin used in this study which were supported unconditionally by Ferring Pharmaceutical Ltd., Thailand.
Funding
This study was funded by the Siriraj Research Development Fund, which is managed by the Routine to Research Project (R2R; Grant number 16OG00021/044/16). The one hundred doses of carbetocin used in this study were provided unconditionally by Ferring Pharmaceutical Ltd., Thailand. The funders had no role in the processes of the study design, data collection, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
TL: protocol/project development, data analysis and interpretation, manuscript writing/editing, final form of manuscript. PA: data collection and management, manuscript writing, funding management. DB: protocol/project development, data analysis and interpretation. JL: data collection and management. RS: data collection and management. RJ: data collection and management, funding management.
Corresponding author
Ethics declarations
Conflict of interest
Assistant professor Tripop Lertbunnaphong has received a speaker honorarium from Ferring Pharmaceutical Ltd., Thailand. Paweena Amornpetchakul, Dittakarn Boriboonhiransarn, Jarunee Leetheeragul, Ratree Sirisomboon and Ratchada Jiraprasertwong declare that they have no conflict of interest.
Research involving human participants and/or animals
Ethics approval: All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Amornpetchakul, P., Lertbunnaphong, T., Boriboonhiransarn, D. et al. Intravenous carbetocin versus intravenous oxytocin for preventing atonic postpartum hemorrhage after normal vaginal delivery in high-risk singleton pregnancies: a triple-blind randomized controlled trial. Arch Gynecol Obstet 298, 319–327 (2018). https://doi.org/10.1007/s00404-018-4806-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-018-4806-5