Abstract
Introduction
There is evidence of a probable key role of the activator protein-2 γ (AP-2γ) in placental development. It is still an open question whether AP-2γ expression may be influenced by preeclampsia, which is a serious pregnancy complication, or by smoking, which has deleterious effects on trophoblastic development.
Material and Methods
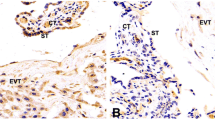
Thus, the expression of AP-2γ was studied in trophoblastic epithelium and endothelium of placentas from patients with preeclampsia (n = 43) and smokers (n = 45) as well as placentas of healthy pregnant women (control group, n = 26) between gestational ages 23 and 43 weeks. To allow differential expression in primary, secondary and tertiary villi, AP-2γ expression (arbitrary units) was determined immunohistologically.
Results
In preeclamptic placentas trophoblastic as well as endothelial cells AP-2γ expression was significantly higher compared to that in control placentas. Endothelial AP-2γ expression in placentas from smokers was similar to that of healthy women while trophoblastic AP-2γ expression in smokers’ placenta was insignificantly higher compared to that of control placentas. In all three groups expression rates of AP-2γ did not differ between primary, secondary and tertiary villi.
Conclusion
A correlation between increased trophoblastic and endothelial AP-2γ expression in patients with preeclampsia and reduced trophoblastic invasion and migration in preeclampsia has to be discussed. Furthermore, increased AP-2γ expression may play a protective role in preeclampsia, protecting from raised blood pressure. The tendency of an enhanced trophoblastic AP-2γ expression in smokers may indicate a compensatory response to the disturbed balance between proliferation and differentiation of villi induced by smoking.





Similar content being viewed by others
References
Williams T, Tjian R (1991) Characterization of a dimerization motif in AP-2 and its function in heterologous DNA-binding proteins. Science 251:1067–1071
Wankhade S, Yu Y, Weinberg J, Tainsky MA, Kannan P (2000) Characterization of the activation domains of AP-2 family transcription factors. J Biol Chem 275(38):29701–29708
Eckert D, Buhl S, Weber S et al (2005) The AP-2 family of transcription factors. Genome Biol 6:246
Hilger-Eversheim K, Moser M, Schorle H, Buettner R (2000) Regulatory roles of AP-2 transcription factors in vertebrate development, apoptosis and cell-cycle control. Gene 260:1–12
Auman HJ, Nottoli T, Lakiza O et al (2002) Transcription factor AP-2 gamma is essential in the extraembryonic lineages for early postimplantation development. Development 129:2733–2747
Werling U, Schorle H (2002) Transcription factor gene AP-2γ essential for early murine development. Mol Cell Biol 22(9):3149–3156
Li M, Kellems RE (2003) Sp1 and Sp3 are important regulators of AP-2γ gene transcription. Biol Repro 69(4):1220–1230
Richardson BD, Cheng Y-H, Langland RA, Handwerger S (2001) Differential expression of AP-2γ and AP-2α during human trophoblast differentiation. Life Sci 69(18):2157–2165
Biadasiewicz K, Sonderegger S, Haslinger P et al (2011) Transcription Factor AP-2γ promotes EGF-dependent invasion of human trophoblast. Endocrinology 152:1458–1469
Sibai B, Dekker G, Kupferminc M (2005) Pre-eclampsia. Lancet 365:785–799
Kotani T, Iwase A, Ino K et al (2009) Activator protein-2 impairs the invasion of a human extravillous trophoblast cell line. Endocrinol 150:4276–4285
Einarson A, Riordan S (2009) Smoking in pregnancy and lactation: a review of risks and cessation strategies. Eur J Clin Pharmacol 65:325–330
Rodriguez-Thompson D (2012) Smoking and pregnancy. Up To Date® 19.3 Jan 2012
Genbacev O, Bass KE, Joslin RJ, Fisher SJ (1995) Maternal smoking inhibits early human cytotrophoblast differentiation. Reprod Toxicol 9:245–255
Zdravkovic T, Genbacev O, McMaster MT, Fisher SJ (2005) The adverse effects of maternal smoking on the human placenta: a review. Placenta 26(Suppl A):S81–S86
Janiaux E, Burton GJ (2007) Morphological and biological effects of maternal exposure to tobacco smoke on the feto-placental unit. Early Hum Dev 83(11):699–706
Rappolee DA, Awonuga AO, Puscheck EE (2010) Benzopyrene and experimental stressors cause compensatory differentiation in placental trophoblast stem cells. Syst Biol Repro Med 56:168–183
Karumanchi SA, Levine RJ (2010) How does smoking reduce the risk of preeclampsia? Hypertension 55(5):1100–1101
Lockwood CJ, Yen CF, Basar M, Kayisli UA, Martel M, Buhimschi M, Buhimschi C, Huang SJ, Krikun G, Schatz F (2008) Preeclampsia-related inflammatory cytokines regulate interleukin-6 expression in human decidual cells. Am J Pathol 172:1571–1579
Xie C, Yao MZ, Liu JB, Xiong LK (2011) A meta-analysis of tumor necrosis factor alpha, interleukin-6, and interleukin-10 in preeclampsia. Cytokine 56(3):550–559
Zhou P, Luo X, Qi HB et al (2012) The expression of pentraxin 3 and tumor necrosis factor-alpha is increased in preeclamptic placental tissue and maternal serum. Inflamm Res 61(9):1005–1012
Nomura S, Ito T, Yamamoto E et al (2005) Gene regulation and physiological function of placental leucine aminopeptidase/oxytocinase during pregnancy. Biochim Biophys Acta 1751(1):19–25
Collet M, Beillard C (2005) Consequences of smoking on fetal development and risk of intra-uterine growth retardation or in utero fetal death. J Gynecol Obstet Biol Reprod 34(1):3S135–3S145
Shea AK, Steiner M (2008) Cigarette smoking during pregnancy. Nicotine Tob Res 10(2):267–278
Lorquet S, Pequeux C, Munaut C, Foidart JM (2010) Aetiology and physiopathology of preeclampsia and related forms. Acta Clin Belg 65(4):237–241
George EM, Granger JP (2011) Mechanisms and potential therapies for preeclampsia. Curr Hypertens Rep 13(4):269–275
Petrescu F, Voican SC, Silosi S (2010) Tumor necrosis factor-a serum levels in healthy smokers and nonsmokers. Int J Chron Obstruct Pul Dis 5:217–222
Nunes SO, Vargas HO, Brum J et al (2011) A comparison of inflammatory markers in depressed and nondepressed smokers. Nicotine Tob Res 14(5):540–546
Conflict of interest
The authors declare that they have no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schneider, H.A., Gembruch, U., Fimmers, R. et al. Expression of AP-2γ in placentas of patients with preeclampsia and of smokers. Arch Gynecol Obstet 291, 1015–1021 (2015). https://doi.org/10.1007/s00404-014-3473-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-014-3473-4