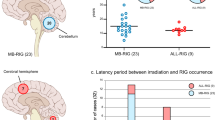
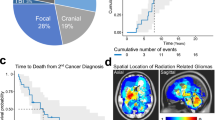
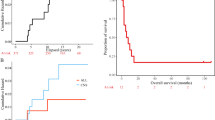
Abstract
Radiotherapy improves survival for common childhood cancers such as medulloblastoma, leukemia, and germ cell tumors. Unfortunately, long-term survivors suffer sequelae that can include secondary neoplasia. Gliomas are common secondary neoplasms after cranial or craniospinal radiation, most often manifesting as high-grade astrocytomas with poor clinical outcomes. Here, we performed genetic profiling on a cohort of 12 gliomas arising after therapeutic radiation to determine their molecular pathogenesis and assess for differences in genomic signature compared to their spontaneous counterparts. We identified a high frequency of TP53 mutations, CDK4 amplification or CDKN2A homozygous deletion, and amplifications or rearrangements involving receptor tyrosine kinase and Ras–Raf–MAP kinase pathway genes including PDGFRA, MET, BRAF, and RRAS2. Notably, all tumors lacked alterations in IDH1, IDH2, H3F3A, HIST1H3B, HIST1H3C, TERT (including promoter region), and PTEN, which genetically define the major subtypes of diffuse gliomas in children and adults. All gliomas in this cohort had very low somatic mutation burden (less than three somatic single nucleotide variants or small indels per Mb). The ten high-grade gliomas demonstrated markedly aneuploid genomes, with significantly increased quantity of intrachromosomal copy number breakpoints and focal amplifications/homozygous deletions compared to spontaneous high-grade gliomas, likely as a result of DNA double-strand breaks induced by gamma radiation. Together, these findings demonstrate a distinct molecular pathogenesis of secondary gliomas arising after radiation therapy and identify a genomic signature that may aid in differentiating these tumors from their spontaneous counterparts.



Similar content being viewed by others
References
Agnihotri S, Suppiah S, Tonge PD et al (2017) Therapeutic radiation for childhood cancer drives structural aberrations of NF2 in meningiomas. Nat Commun 8:186
Alexandrov LB, Nik-Zainal S, Wedge DC et al (2013) Signatures of mutational processes in human cancer. Nature 500:415–421
Behjati S, Gundem G, Wedge DC et al (2016) Mutational signatures of ionizing radiation in second malignancies. Nat Commun 7:12605
Brachman D, Hallahan D, Beckett M, Yandell D, Weichselbaum R (1991) p53 gene mutations and abnormal retinoblastoma protein in radiation-induced human sarcomas. Cancer Res 51:6393–6396
Brat DJ, James CD, Jedlicka AE et al (1999) Molecular genetic alterations in radiation-induced astrocytomas. Am J Pathol 154:1431–1438
Cahan W, Woodard H, Higinbotham N, Stewart F, Coley B (1948) Sarcoma arising in irradiated bone: report of eleven cases. Cancer 82:8–34
Camacho CV, Todorova PK, Hardebeck MC et al (2015) DNA double-strand breaks cooperate with loss of Ink4 and Arf tumor suppressors to generate glioblastomas with frequent Met amplification. Oncogene 34:1064–1072
Cancer Genome Atlas Research Network (2008) Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature 455:1061–1068
Cancer Genome Atlas Research Network (2013) The somatic genomic landscape of glioblastoma. Cell 155:462–477
Cancer Genome Atlas Research Network (2015) Comprehensive, integrative genomic analysis of diffuse lower-grade gliomas. N Engl J Med 372:2481–2498
Chowdhary A, Spence AM, Sales L, Rostomily RC, Rockhill JK, Silbergeld DL (2012) Radiation associated tumors following therapeutic cranial radiation. Surg Neurol Int 3:48
Donson AM, Erwin NS, Kleinschmidt-DeMasters BK, Madden JR, Addo-Yobo SO, Foreman NK (2007) Unique molecular characteristics of radiation-induced glioblastoma. J Neuropathol Exp Neurol 66:740–749
Eckel-Passow JE, Lachance DH, Molinaro AM et al (2015) Glioma groups based on 1p/19q, IDH, and TERT promoter mutations in tumors. N Engl J Med 372:2499–2508
Fontebasso AM, Schwartzentruber J, Khuong-Quang DA et al (2013) Mutations in SETD2 and genes affecting histone H3K36 methylation target hemispheric high-grade gliomas. Acta Neuropathol 125:659–669
Foray N (2016) Victor Despeignes, the forgotten pioneer of radiation oncology. Int J Radiat Oncol Biol Phys 96:717–721
Henson JW, Hobbs W, Chakravarti A, Louis DN (2005) Alterations in p53, p21, and MIB-1 labeling index in primary human astrocytomas following radiation therapy. J Neurooncol 74:151–154
Iorgulescu JB, Van Ziffle J, Stevers M et al (2018) Deep sequencing of WNT-activated medulloblastomas reveals secondary SHH pathway activation. Acta Neuropathol 135:635–638
Kline CN, Joseph NM, Grenert JP et al (2017) Targeted next-generation sequencing of pediatric neuro-oncology patients improves diagnosis, identifies pathogenic germline mutations, and directs targeted therapy. Neuro-Oncol 19:699–709
Mackay A, Burford A, Carvalho D et al (2017) Integrated molecular meta-analysis of 1000 pediatric high-grade and diffuse intrinsic pontine glioma. Cancer Cell 32:520–537
Nakao T, Sasagawa Y, Nobusawa S et al (2017) Radiation-induced gliomas: a report of four cases and analysis of molecular biomarkers. Brain Tumor Pathol 34:149–154
Paugh BS, Qu C, Jones C et al (2010) Integrated molecular genetic profiling of pediatric high-grade gliomas reveals key differences with the adult disease. J Clin Oncol 28:3061–3068
Pekmezci M, Stevers M, Phillips JJ et al (2018) Multinodular and vacuolating neuronal tumor of the cerebrum is a clonal neoplasm defined by genetic alterations that activate the MAP kinase signaling pathway. Acta Neuropathol 135:485–488
Pekmezci M, Villanueva-Meyer JE, Goode B et al (2018) The genetic landscape of ganglioglioma. Acta Neuropathol Commun 6:47
Phi JH, Park AK, Lee S et al (2018) Genomic analysis reveals secondary glioblastoma after radiotherapy in a subset of recurrent medulloblastomas. Acta Neuropathol 135:939–953
Phillips JJ, Gong H, Chen K et al (2016) Activating NRF1-BRAF and ATG7-RAF1 fusions in anaplastic pleomorphic xanthoastrocytoma without BRAF p. V600E mutation. Acta Neuropathol 132:757–760
Phillips JJ, Gong H, Chen K, Joseph NM, van Ziffle J, Bastian BC, Grenert JP, Kline CN, Mueller S, Banerjee A, Nicolaides T, Gupta N, Berger MS, Lee HS, Pekmezci M, Tihan T, Bollen AW, Perry A, Shieh JTC, Solomon DA (2018) The genetic landscape of anaplastic pleomorphic xanthoastrocytoma. Brain Pathol. https://doi.org/10.1111/bpa.12639
Rodriguez FJ, Schniederjan MJ, Nicolaides T, Tihan T, Burger PC, Perry A et al (2015) High rate of concurrent BRAF-KIAA1549 gene fusion and 1p deletion in disseminated oligodendroglioma-like leptomeningeal neoplasms (DOLN). Acta Neuropathol 129:609–610
Sahm F, Toprak UH, Hübschmann D et al (2017) Meningiomas induced by low-dose radiation carry structural variants of NF2 and a distinct mutational signature. Acta Neuropathol 134:155–158
Salvati M, D’Elia A, Melone GA et al (2008) Radio-induced gliomas: 20-year experience and critical review of the pathology. J Neurooncol 89:169–177
Solomon DA, Wood MD, Tihan T et al (2016) Diffuse midline gliomas with histone H3-K27M mutation: a series of 47 cases assessing the spectrum of morphologic variation and associated genetic alterations. Brain Pathol 26:569–580
Walter AW, Hancock ML, Pui CH et al (1998) Secondary brain tumors in children treated for acute lymphoblastic leukemia at St Jude Children’s Research Hospital. J Clin Oncol 16:3761–3767
Wu G, Diaz AK, Paugh BS et al (2014) The genomic landscape of diffuse intrinsic pontine glioma and pediatric non-brainstem high-grade glioma. Nat Genet 46:444–450
Yamanaka R, Hayano A, Kanayama T (2018) Radiation-induced gliomas: a comprehensive review and meta-analysis. Neurosurg Rev 41:719–731
Acknowledgements
G.Y.L is supported by the National Cancer Institute Training Program in Translational Brain Tumor Research (T32 CA151022). B.C.B. is supported by an NCI Outstanding Investigator Award (R35 CA220481). D.A.S. is supported by the National Institutes of Health Director’s Early Independence Award (DP5 OD021403) and the UCSF Physician-Scientist Scholar Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This study was approved by the Committee on Human Research of the University of California, San Francisco, with a waiver of patient consent.
Conflict of interest
The authors declare that they have no competing interests related to this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
López, G.Y., Van Ziffle, J., Onodera, C. et al. The genetic landscape of gliomas arising after therapeutic radiation. Acta Neuropathol 137, 139–150 (2019). https://doi.org/10.1007/s00401-018-1906-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-018-1906-z