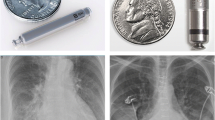
Abstract
Nanostim™ (St. Jude Medical Inc., Saint Paul, MN, USA; now Abbott Medical Inc. Abbott Park, IL, USA) was the first self-contained intracardiac pacemaker to be implanted in a human patient. A total of 1423 Nanostim devices were implanted worldwide between 2013 and 2016 and three clinical trials were initiated. Although the device was recalled in 2016 owing to rare but serious battery failures, the concept of leadless pacing has gained widespread acceptance and is expanding beyond the initial single-chamber devices to dual-chamber systems, biventricular pacing, and combinations with defibrillators. This review describes the design, results from initial clinical trials, and long-term experiences with Nanostim. It discusses the lessons learned from the pioneering device’s successes and shortcomings, many of which are valid for leadless pacemakers in general. This article also considers the Nanostim experience in comparison with the early years of clinical use for other pioneering device therapies. Important questions include how to minimize the risk for short-term complications by appropriate operator training and evaluation of suitable patients, what the long-term performance tells us about safety, as well as the necessity and feasibility of device explantation.
Zusammenfassung
Nanostim™ (St. Jude Medical Inc., Saint Paul/MN, USA; jetzt Abbott Medical Inc. Abbott Park, IL, USA) war der erste sondenlose intrakardiale Herzschrittmacher, der 2013 einem Patienten implantiert wurde. Insgesamt wurden zwischen 2013 und 2016 weltweit 1423 Nanostim™-Geräte im Rahmen von 3 klinischen Studien implantiert. Obwohl die Implantationen 2016 aufgrund seltener, aber schwerwiegender Batterieausfälle gestoppt wurden, fand das Konzept der sondenlosen Stimulation breite Akzeptanz und wird bereits in ersten Studien bei 2‑Kammer-Herzschrittmachern, der linksventrikulären Stimulation und in der Kombination mit subkutanen Defibrillatoren angewendet. In der vorliegenden Arbeit werden das Design des Systems und die Ergebnisse der ersten klinischen Studien kritisch dargestellt. Viele Ergebnisse daraus sind für sondenlose Schrittmacher i. Allg. zutreffend. Die Erfahrungen mit dem Nanostim™-Gerät werden hier mit den ersten Jahren der klinischen Anwendung anderer neuartiger Therapiesysteme verglichen. Wichtige Fragen sind, wie das Risiko für periprozedurale Komplikationen durch entsprechende Schulung der Anwender und die Auswahl der geeigneten Patienten minimiert werden kann und was die Langzeitergebnisse über die Systemsicherheit aussagen können, einschließlich der Möglichkeit der Systemexplantation.



Similar content being viewed by others
References
Acha MR, Keaney JJ, Lubitz SA et al (2015) Increased perforation risk with an MRI-conditional pacing lead: a single-center study. Pacing Clin Electrophysiol PACE 38:334–342. https://doi.org/10.1111/pace.12550
Beurskens NEG, Tjong FVY, Quast AFBE, Knops RE (2018) Successful replacement of the longest worldwide in situ Nanostim leadless cardiac pacemaker for a Micra Transcatheter Pacing System. J Interv Card Electrophysiol 51:161–162. https://doi.org/10.1007/s10840-017-0310-2
Boveda S, Lenarczyk R, Haugaa KH et al (2018) Use of leadless pacemakers in Europe: results of the European Heart Rhythm Association survey. Europace 20:555–559. https://doi.org/10.1093/europace/eux381
Brignole M, Auricchio A, Baron-Esquivias G et al (2013) 2013 ESC Guidelines on cardiac pacing and cardiac resynchronization therapy. Eur Heart J 34:2281–2329. https://doi.org/10.1093/eurheartj/eht150
Defaye P, Klug D, Anselme F et al (2018) Recommendations for the implantation of leadless pacemakers from the French Working Group on Cardiac Pacing and Electrophysiology of the French Society of Cardiology. Arch Cardiovasc Dis 111:53–58. https://doi.org/10.1016/j.acvd.2017.10.002
DeSimone CV, Friedman PA, Noheria A et al (2013) Stroke or transient ischemic attack in patients with transvenous pacemaker or defibrillator and echocardiographically detected patent foramen ovale. Circulation 128:1433–1441. https://doi.org/10.1161/CIRCULATIONAHA.113.003540
Epstein AE, DiMarco JP, Ellenbogen KA et al (2013) 2012 ACCF/AHA/HRS focused update incorporated into the ACCF/AHA/HRS 2008 guidelines for device-based therapy of cardiac rhythm abnormalities. Circulation 127:e283–352. https://doi.org/10.1161/CIR.0b013e318276ce9b (A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society)
Healey JS (2006) Cardiovascular outcomes with atrial-based pacing compared with ventricular pacing: Meta-analysis of randomized trials, using individual patient data. Circulation 114:11–17. https://doi.org/10.1161/CIRCULATIONAHA.105.610303
Higgins S, Rogers JD (2014) Advances in pacing therapy: examining the potential impact of leadless pacing. Therapy J Innov Card Rhythm Manag 5:1825–1833
Jeffrey K, Parsonnet V (1998) Cardiac pacing, 1960–1985: a quarter century of medical and industrial innovation. Circulation 97:1978–1991
Kilic T, Yilmaz I (2017) Transcatheter aortic valve implantation: a revolution in the therapy of elderly and high-risk patients with severe aortic stenosis. J Geriatr Cardiol Jgc 14:204–217. https://doi.org/10.11909/j.issn.1671-5411.2017.03.002
Kirkfeldt RE, Johansen JB, Nohr EA et al (2014) Complications after cardiac implantable electronic device implantations: an analysis of a complete, nationwide cohort in Denmark. Eur Heart J 35:1186–1194. https://doi.org/10.1093/eurheartj/eht511
Knops RE, Tjong FVY, Neuzil P et al (2015) Chronic performance of a leadless cardiac pacemaker: 1‑year follow-up of the LEADLESS trial. J Am Coll Cardiol 65:1497–1504. https://doi.org/10.1016/j.jacc.2015.02.022
Lakkireddy D, Knops R, Atwater B et al (2017) A worldwide experience of the management of battery failures and chronic device retrieval of the Nanostim leadless pacemaker. Heart Rhythm 14:1756–1763. https://doi.org/10.1016/j.hrthm.2017.07.004
Mahapatra S, Bybee KA, Bunch TJ et al (2005) Incidence and predictors of cardiac perforation after permanent pacemaker placement. Heart Rhythm 2:907–911. https://doi.org/10.1016/j.hrthm.2005.06.011
MHRA Expert Advisory Group (2017) https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/598730/Leadless_cardiac_pacemaker_therapy_-_EAG_initial_recommendations_.pdf
Miller MA, Neuzil P, Dukkipati SR, Reddy VY (2015) Leadless cardiac pacemakers: back to the future. J Am Coll Cardiol 66:1179–1189. https://doi.org/10.1016/j.jacc.2015.06.1081
Omdahl P, Eggen MD, Bonner MD et al (2016) Right ventricular anatomy can accommodate multiple micra transcatheter pacemakers. Pacing Clin Electrophysiol PACE 39:393–397. https://doi.org/10.1111/pace.12804
Reddy VY, Exner DV, Cantillon DJ et al (2015) Percutaneous implantation of an entirely Intracardiac Leadless pacemaker. N Engl J Med 373:1125–1135. https://doi.org/10.1056/NEJMoa1507192
Reddy VY, Knops RE, Sperzel J et al (2014) Permanent leadless cardiac pacing: results of the LEADLESS trial. Circulation 129:1466–1471. https://doi.org/10.1161/CIRCULATIONAHA.113.006987
Reddy VY, Miller MA, Neuzil P et al (2017) Cardiac resynchronization therapy with wireless left ventricular Endocardial pacing: the SELECT-LV study. J Am Coll Cardiol 69:2119–2129. https://doi.org/10.1016/j.jacc.2017.02.059
Richter S, Döring M, Ebert M et al (2018) Battery malfunction of a leadless cardiac pacemaker—A worrisome single-center experience. Circulation. https://doi.org/10.1161/CIRCULATIONAHA.117.033371
Sperzel J, Defaye P, Delnoy P‑P et al (2018) Primary safety results from the LEADLESS observational study. Europace. https://doi.org/10.1093/europace/eux359
Spickler JW, Rasor NS, Kezdi P et al (1970) Totally self-contained intracardiac pacemaker. J Electrocardiol 3:325–331
Tjong FVY, Brouwer TF, Koop B et al (2017) Acute and 3‑month performance of a communicating leadless antitachycardia pacemaker and subcutaneous implantable defibrillator. Jacc Clin Electrophysiol 3:1487–1498. https://doi.org/10.1016/j.jacep.2017.04.002
Tjong FVY, Knops RE, Neuzil P et al (2018) Midterm safety and performance of a leadless cardiac pacemaker: 3‑year follow-up to the LEADLESS trial (Nanostim safety and performance trial for a Leadless cardiac pacemaker system). Circulation 137:633–635. https://doi.org/10.1161/CIRCULATIONAHA.117.030106
Tjong FVY, Reddy VY (2017) Permanent leadless cardiac pacemaker therapy: a comprehensive review. Circulation 135:1458–1470. https://doi.org/10.1161/CIRCULATIONAHA.116.025037
Udo EO, Zuithoff NPA, van Hemel NM et al (2012) Incidence and predictors of short- and long-term complications in pacemaker therapy: the FOLLOWPACE study. Heart Rhythm 9:728–735. https://doi.org/10.1016/j.hrthm.2011.12.014
Wilkoff BL, Bello D, Taborsky M et al (2011) Magnetic resonance imaging in patients with a pacemaker system designed for the magnetic resonance environment. Heart Rhythm 8:65–73. https://doi.org/10.1016/j.hrthm.2010.10.002
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
J. Sperzel, C. Hamm, and A. Hain declare that they have no competing interests.
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Sperzel, J., Hamm, C. & Hain, A. Nanostim—leadless pacemaker. Herzschr Elektrophys 29, 327–333 (2018). https://doi.org/10.1007/s00399-018-0598-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00399-018-0598-3
Keywords
- Leadless pacemaker
- Nanostim™ study results
- Safety aspects
- Implantable defibrillators
- Cardiac resynchronization therapy