Abstract
Objective
Transverse aortic constriction (TAC) results in a transient increase of proinflammatory cytokines, which return to baseline levels within 3 d. In contrast to cytokine baseline levels, the myocardium remains capable to respond even stronger to a new stimulus. As the molecular mechanisms for this phenomenon are unknown, we tested whether TAC modulates the innate immune system in mice and changes the inflammatory reaction to a new stimulus.
Methods
Following 3 d of TAC or sham-operation procedure (SOP), LPS (20 mg/kg) or PBS (control) were administered intraperitoneal for 10 min as well as for 6 h. Hemodynamic parameters were recorded to measure the effects of TAC and LPS. After TAC/SOP alone CD14 expression was monitored and after additional 6 h of LPS/PBS the expression of CD14, TLR4 and proinflammatory cytokines were determined by western-blot, ELISA and RNase protection assay, respectively. Following TAC/SOP and 10 min of LPS/PBS, NFκB activation was investigated by EMSA.
Results
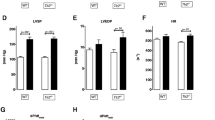
TAC induced cardiac hypertrophy and elevated blood pressure. LPS application led to hypotension and other symptoms of sepsis. CD14 expression increased after TAC alone and even further after additional LPS challenge. However, we did not detect changes of TLR4 expression. Also NFκB activation increased after LPS challenge higher in the TAC than in the SOP group. LPS-stimulation induced also higher cytokine expression in the TAC than in the SOP group.
Conclusion
TAC modulates innate immunity by regulating the expression of CD14 and changes the myocardial tissue to respond more powerful to LPS.
Similar content being viewed by others
Reference
Asehnoune K, Strassheim D, Mitra S, Yeol KJ, Abraham E (2005) Involvement of PKCalpha/beta in TLR4 and TLR2 dependent activation of NF-kappaB. Cell Signal 17:385–394
Baumgarten G, Knuefermann P, Kalra D, Gao F, Taffet GE, Michael L, Blackshear PJ, Carballo E, Sivasubramanian N, Mann DL (2002) Load-dependent and -independent regulation of proinflammatory cytokine and cytokine receptor gene expression in the adult mammalian heart. Circulation 105:2192–2197
Baumgarten G, Knuefermann P, Nozaki N, Sivasubramanian N, Mann DL, Vallejo JG (2001) In vivo expression of proinflammatory mediators in the adult heart after endotoxin administration: the role of toll-like receptor-4. J Infect Dis 183:1617–1624
Baumgarten G, Knuefermann P, Schuhmacher G, Vervolgyi V, von RJ, Dreiner U, Fink K, Djoufack C, Hoeft A, Grohe C, Knowlton AA, Meyer R (2006) Toll-like receptor 4, nitric oxide, and myocardial depression in endotoxemia. Shock 25:43–49
Bozkurt B, Kribbs SB, Clubb FJ Jr, Michael LH, Didenko VV, Hornsby PJ, Seta Y, Oral H, Spinale FG, Mann DL (1998) Pathophysiologically relevant concentrations of tumor necrosis factor- alpha promote progressive left ventricular dysfunction and remodeling in rats. Circulation 97:1382–1391
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate- phenol-chloroform extraction. Anal Biochem 162:156–159
Cowan DB, Poutias DN, Del Nido PJ, McGowan FX Jr (2000) CD14-independent activation of cardiomyocyte signal transduction by bacterial endotoxin. Am J Physiol Heart 279:H619–H629
Fearns C, Kravchenko VV, Ulevitch RJ, Loskutoff DJ (1995) Murine CD14 gene expression in vivo: extramyeloid synthesis and regulation by lipopolysaccharide. J Exp Med 181:857–866
Frankenberger M, Pechumer H, Ziegler-Heitbrock HW (1995) Interleukin-10 is upregulated in LPS tolerance. J Inflamm 45:56–63
Frantz S, Kobzik L, Kim YD, Fukazawa R, Medzhitov R, Lee RT, Kelly RA (1999) Toll4 (TLR4) expression in cardiac myocytes in normal and failing myocardium. J Clin Invest 104:271–280
Frey EA, Miller DS, Jahr TG, Sundan A, Bazil V, Espevik T, Finlay BB, Wright SD (1992) Soluble CD14 participates in the response of cells to lipopolysaccharide. J Exp Med 176:1665–1671
Johnson BA, Geha M, Blackwell TK (2000) Similar but distinct effects of the tristetraprolin/TIS11 immediate-early proteins on cell survival. Oncogene 19:1657–1664
Johnson GB, Brunn GJ, Platt JL (2004) Cutting edge: an endogenous pathway to systemic inflammatory response syndrome (SIRS)-like reactions through Toll-like receptor 4. J Immunol 172:20–24
Kapadia S, Lee J, Torre-Amione G, Birdsall HH, Ma TS, Mann DL (1995) Tumor necrosis factor-alpha gene and protein expression in adult feline myocardium after endotoxin administration. J Clin Invest 96:1042–1052
Knuefermann P, Chen P, Misra A, Shi SP, Abdellatif M, Sivasubramanian N (2002) Myotrophin/V-1, a protein up-regulated in the failing human heart and in postnatal cerebellum, converts NFkappa B p50-p65 heterodimers to p50-p50 and p65-p65 homodimers. J Biol Chem 277:23888–23897
Knuefermann P, Nemoto S, Misra A, Nozaki N, Defreitas G, Goyert SM, Carabello BA, Mann DL, Vallejo JG (2002) CD14-deficient mice are protected against lipopolysaccharide-induced cardiac inflammation and left ventricular dysfunction. Circulation 106:2608–2615
Kopp EB, Medzhitov R (1999) The toll-receptor family and control of innate immunity. Curr Opin Immunol 11:13–18
Mann DL (2002) Inflammatory mediators and the failing heart: past, present, and the foreseeable future. Circ Res 91:988–998
Muller-Werdan U, Engelmann H, Werdan K (1998) Cardiodepression by tumor necrosis factor-alpha. Eur Cytokine Netw 9:689–691
Nemoto S, Vallejo JG, Knuefermann P, Misra A, Defreitas G, Carabello BA, Mann DL (2002) Escherichia coli LPS-induced LV dysfunction: role of toll-like receptor-4 in the adult heart. Am J Physiol Heart 282:H2316–H2323
Ohashi K, Burkart V, Flohe S, Kolb H (2000) Cutting edge: heat shock protein 60 is a putative endogenous ligand of the toll-like receptor-4 complex. J Immunol 164:558–561
Okamura Y, Watari M, Jerud ES, Young DW, Ishizaka ST, Rose J, Chow JC, Strauss JF (2001) The extra domain A of fibronectin activates toll-like receptor 4. J Biol Chem 276:10229–10233
Poltorak A, He X, Smirnova I, Liu MY, Huffel CV, Du X, Birdwell D, Alejos E, Silva M, Galanos C, Freudenberg M, Ricciardi-Castagnoli P, Layton B, Beutler B (1998) Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science 282:2085–2088
Poltorak A, Smirnova I, He X, Liu MY, Van Huffel C, McNally O, Birdwell D, Alejos E, Silva M, Du X, Thompson P, Chan EK, Ledesma J, Roe B, Clifton S, Vogel SN, Beutler B (1998) Genetic and physical mapping of the Lps locus: identification of the toll-4 receptor as a candidate gene in the critical region. Blood Cells Mol Dis 24:340–355
Qureshi ST, Lariviere L, Leveque G, Clermont S, Moore KJ, Gros P, Malo D (1999) Endotoxin-tolerant mice have mutations in Toll-like receptor 4 (Tlr4). J Exp Med 189:615–625
Smiley ST, King JA, Hancock WW (2001) Fibrinogen stimulates macrophage chemokine secretion through toll-like receptor 4. J Immunol 167:2887–2894
Tavener SA, Long EM, Robbins SM, McRae KM, Van Remmen H, Kubes P (2004) Immune cell Toll-like receptor 4 is required for cardiac myocyte impairment during endotoxemia. Circ Res 95:700–707
Wright SD, Ramos RA, Tobias PS, Ulevitch RJ, Mathison JC (1990) CD14, a receptor for complexes of lipopolysaccharide (LPS) and LPS binding protein. Science 249:1431–1433
Ziegler-Heitbrock HW (1995) Molecular mechanism in tolerance to lipopolysaccharide. J Inflamm 45:13–26
Author information
Authors and Affiliations
Corresponding author
Additional information
Returned for 1st revision: 30 November 2005 1st revision received: 28 March 2006
Rights and permissions
About this article
Cite this article
Baumgarten, G., Kim, SC., Stapel, H. et al. Myocardial injury modulates the innate immune system and changes myocardial sensitivity. Basic Res Cardiol 101, 427–435 (2006). https://doi.org/10.1007/s00395-006-0597-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00395-006-0597-0