Abstract
Purpose
Yerba maté is widely consumed in South America as different beverages, such as maté tea (roasted leaves) and chimarrão (green dried leaves), and linked to health benefits, mainly attributed to chlorogenic acids (CGAs). Health effects of CGAs depend on their bioavailability, but such data are scarce. The aim of this study was to investigate the distribution of CGAs and metabolites in tissues, hepatic and plasmatic kinetic profile and urinary excretion after ingestion of maté tea or 5-caffeoylquinic acid (5-CQA).
Methods
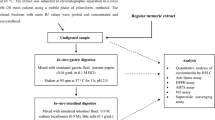
Wistar rats ingested maté tea (MT) or 5-CQA (ST) and were killed after 1.5 h for tissue distribution analysis (pilot study) or at 0.5, 1, 2, 4 and 8 h for liver and plasma kinetics (main experiment). Urine was collected in metabolic cages. Biological samples were analyzed by UPLC-DAD-MS with and without incubation with β-glucuronidase and sulfatase.
Results
CGAs and metabolites were detected in all tissues. Caffeic acid was the main compound in plasma up to 2 h after ingestion of maté tea, while 5-CQA predominated in ST group. Concentration of microbial metabolites increased 4 h after gavage and reached higher amounts in MT plasma and liver, when compared to ST group. Approximately 4.0 % of compounds ingested by MT and 3.3 % by ST were recovered in urine up to 8 h after the gavage.
Conclusion
The study confirms that not only absorption, but also metabolization of CGAs begins in stomach. There were differences in compounds formed from maté tea or isolated 5-CQA, showing that CGAs profile in food may influence qualitatively and quantitatively the metabolites formed in the body.






Similar content being viewed by others
References
Bastos DHM, Oliveira DM, Matsumoto RLT, Carvalho PO, Ribeiro ML (2007) Yerba maté: pharmacological properties, research and biotechnology. Med Aromat Plant Sci Biotechnol 1:37–46
Gambero A, Ribeiro ML (2015) The positive effects of yerba maté (Ilex paraguariensis) in obesity. Nutrients 7:730–750
Crozier A, Jaganath IB, Clifford MN (2009) Dietary phenolics: chemistry, bioavailability and effects on health. Nat Prod Rep 26:1001–1043
Clifford MN (1999) Chlorogenic acids and other cinnamates—nature, occurrence and dietary burden. J Sci Food Agric 1999(79):362–372
Lafay S, Gil-izquierdo A (2008) Bioavailability of phenolic acids. Phytochem Rev 7:301–311
Zhao Z, Moghadasian MH (2010) Bioavailability of hydroxycinnamates: a brief review of in vivo and in vitro studies. Phytochem Rev 9:133–145
Ludwig IA, Pena MP, Cid C, Crozier A (2013) Catabolism of coffee chlorogenic acids by human colonic microbiota. BioFactors 39:623–632
Crozier A, Del Rio D, Clifford MN (2010) Bioavailability of dietary flavonoids and phenolic compounds. Mol Asp Med 31:446–467
Serra A, Macia A, Rubio L, Angle’s N, Ortega N, Morello JR, Romero MP, Motilva MJ (2013) Distribution of procyanidins and their metabolites in rat plasma and tissues in relation to ingestion of procyanidin-enriched or procyanidin-rich cocoa cream. Eur J Nutr 52:1029–1038
Porrini M, Riso P (2008) Factors influencing the bioavailability of antioxidants in foods: a critical appraisal. Nutr Metab Cardiovasc Dis 18:647–650
Stalmach A, Williamson G, Crozier A (2014) Impact of dose on the bioavailability of coffee chlorogenic acids in humans. Food Funct 5:1727–1737
Andreasen MF, Kroon PA, Williamson G, Garcia-Conesa MT (2001) Esterase activity able to hydrolyze dietary antioxidant hydroxycinnamates is distributed along the intestine of mammals. J Agric Food Chem 49:5679–5684
Kern SM, Bennett RN, Needs PW, Mellon FA, Kroon PA, Garcia-Conesa MT (2003) Characterization of metabolites of hydroxycinnamates in the in vitro model of human small intestinal epithelium caco-2 cells. J Agric Food Chem 51:7884–7891
Rechner AR, Kuhnle G, Bremner P, Hubbard GP, Moore KP, Rice-Evans C (2002) The metabolic fate of dietary polyphenols in humans. Free Radic Biol Med 33:220–235
Stalmach A, Mullen W, Barron D, Uchida K, Yokota T, Cavin C, Steiling H, Williamson G, Crozier A (2009) Metabolite profiling of hydroxycinnamate derivatives in plasma and urine after the ingestion of coffee by humans: identification of biomarkers of coffee consumption. Drug Metab Dispos 37:1749–1758
Stalmach A, Steiling H, Williamson G, Crozier A (2010) Bioavailability of chlorogenic acids following acute ingestion of coffee by humans with an ileostomy. Arch Biochem Biophys 501:98–105
Monteiro M, Farah A, Perrone D, Trugo LC, Donangelo C (2007) Chlorogenic acid compounds from coffee are differentially absorbed and metabolized in humans. J Nutr 137:2196–2201
Duarte GS, Farah A (2011) Effect of simultaneous consumption of milk and coffee on chlorogenic acid’s bioavailability in humans. J Agric Food Chem 59:7925–7931
Renouf M, Guy PA, Marmet C, Fraering AL, Longet K, Moulin J, Enslen M, Barron D, Dionisi F et al (2010) Measurement of caffeic and ferulic acid equivalents in plasma after coffee consumption: small intestine and colon are key sites for coffee metabolism. Mol Nutr Food Res 54:1–7
Gonthier MP, Verny MA, Besson C, Remesy C, Scalbert A (2003) Chlorogenic acid bioavailability largely depends on its metabolism by the gut microflora in rats. J Nutr 133:1853–1859
Rechner AR, Smith MA, Kuhnle G, Gibson GR, Debnam ES, Srai SK, Moore KP, Rice-Evans CA (2004) Colonic metabolism of dietary polyphenols: influence of structure on microbial fermentation products Free Radic. Biol Metab 36(2):212–225
Williamson G, Dionisi F, Renouf M (2011) Flavanols from green tea and phenolic acids from coffee: critical quantitative evaluation of the pharmacokinetic data in humans after consumption of single doses of beverages. Mol Nutr Food Res 55:864–873
Bastos DHM, Saldanha LA, Catharino RR, Sawaya HF, Cunha IBS, Carvalho PO, Eberlin MN (2007) Phenolic antioxidants identified by ESI-MS from yerba maté (Ilex paraguariensis) and green tea (Camelia sinensis) extracts. Molecules 12:423–432
Jaiswal R, Sovdat T, Vivan F, Kuhnert N (2010) Profiling and characterization by LC-MSn of the chlorogenic acids and hydroxycinnamoylshikimate esters in maté (Ilex paraguariensis). J Agric Food Chem 58:5471–5484
Clifford MN, Johnston KL, Knight S, Kuhnert N (2003) Hierarchical Scheme for LC-MSn Identification of Chlorogenic Acids. J Agric Food Chem 51:2900–2911
Miranda DDC, Arçari DP, Pedrazzoli J Jr, Carvalho PO, Cerutti SM, Bastos DHM, Ribeiro ML (2008) Protective effects of maté tea (Ilex paraguariensis) on H2O2-induced DNA damage and DNA repair in mice. Mutagenesis 23:261–265
Oliveira DM, Pinto CB, Sampaio GR, Yonekura L, Catharino RR (2013) Development and validation of methods for the extraction of phenolic acids from plasma, urine, and liver and analysis by UPLC-MS. J Agric Food Chem 61:6113–6121
Lafay S, Gil-Izquierdo A, Manach C, Morand C, Besson C, Scalbert A (2006) Chlorogenic acid is absorbed in its intact form in the stomach of rats. J Nutr 136:1192–1197
Erk T, Renouf M, Williamson G, Melcher R, Steiling H, Richling E (2014) Absorption and isomerization of caffeoylquinic acids from different foods using ileostomist volunteers. Eur J Nutr 53:159–166
Clifford MN (2004) Diet-derived phenols in plasma and tissues and their implications for health. Planta Med 70:1103–1114
Farah A, Monteiro M, Donangelo CM, Lafay S (2008) Chlorogenic acids from green coffee extract are highly bioavailable in humans. J Nutr 138:2309–2315
Farrell TL, Dew TP, Poquet L, Hanson P, Williamson G (2011) Absorption and metabolism of chlorogenic acids in cultured gastric epithelial monolayers. Drug Metab Dispos 39:2338–2346
Zhao Z, Egashira Y, Sanada H (2004) Ferulic acid is quickly absorbed from rat stomach as the free form and then conjugated mainly in liver. J Nutr 134:3083–3088
Karhunen T, Tilgmann C, Ulmanen I, Julkunen I, Panula P (1994) Distribution of catechol-O-methyltransferase enzyme in rat tissues. J Histochem Cytochem 42:1079
Tahara T, Shibata T, Arisawa T, Nakamura M, Yamashita H, Yoshioka D, Okubo M, Maruyama N, Kamano T, Kamiya Y, Fujita H, Nagasaka M, Iwata M, Takahama K, Watanabe M, Hirata I (2009) Impact of catechol-O-methyltransferase (COMT) gene polymorphism on promoter methylation status in gastric mucosa. Anticancer Res 29:2857–2862
Konishi Y, Kobayashi S (2004) Transepithelial transport of chlorogenic acid, caffeic acid, and their colonic metabolites in intestinal Caco-2 cell monolayers. J Agric Food Chem 52:2518–2526
Kahle K, Kempf M, Schreier P, Scheppach W, Schrenk D, Kautenburger T, Hecker D, Huemmer W, Ackermann M, Richling E (2011) Intestinal transit and systemic metabolism of apple polyphenols. Eur J Nutr 50:507–522
Olthof MR, Hollman PCH, Buijsman MCNP, Amelsvoort JMM, Katan MB (2003) Chlorogenic acid, quercetin-3-Rutinoside and black tea phenols are extensively metabolized in humans. J Nutr 133:1806–1814
Rechner AR, Spencer JPE, Kuhnle G, Hahn U, Rice-Evans CA (2001) Novel biomarkers of the bioavailability and metabolism of caffeic acid derivatives in humans. Free Radic Biol Med 30:1213–1222
Olthof MR, Hollman PC, Katan MB (2001) Chlorogenic acid and caffeic acid are absorbed in humans. J Nutr 131:66–71
Renouf M, Marmet C, Giuffrida F, Lepage M, Barron D, Beaumont M, Williamson G, Dionisi F (2014) Dose–response plasma appearance of coffee chlorogenic and phenolic acids in adults. Mol Nutr Food Res 58:301–309
Azuma K, Ipposushi K, Nakayama M, Ito H, Higashio H, Terao J (2000) Absorption of chlorogenic acid and caffeic acid in rats after oral administration. J Agric Food Chem 48:5496–5500
Lafay S, Morand C, Manach C, Besson C, Scalbert A (2006) Absorption and metabolism of caffeic acid and chlorogenic acid in the small intestine of rats. Br J Nutr 96(1):39–46
Acknowledgments
Authors thank Dr Lina Yonekura and Rosana Manólio Soares, MSc, for their technical assistance. This research was supported by the São Paulo Research Foundation (FAPESP).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
de Oliveira, D.M., Sampaio, G.R., Pinto, C.B. et al. Bioavailability of chlorogenic acids in rats after acute ingestion of maté tea (Ilex paraguariensis) or 5-caffeoylquinic acid. Eur J Nutr 56, 2541–2556 (2017). https://doi.org/10.1007/s00394-016-1290-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-016-1290-1