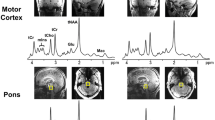
Abstract
Proton magnetic resonance spectroscopy (1H-MRS) has provided evidence for a reduction of N-acetyl-aspartate (NAA) in the medial temporal lobe (MTL) in cerebral disorders such as Alzheimer’s Disease. Within the 1H-MRS study of the German Research Network on Dementia, we determined the multicenter reproducibility of single-voxel 1H-MRS of the MTL. At five sites with 1.5T MR systems, single-voxel 1H spectra from the MTL of an identical healthy subject were measured. The same subject was also examined at one of the sites five times to assess intracenter stability. The protocol included water-suppressed spectra with TE 272 ms and TE 30 ms and unsuppressed spectra for absolute quantification of metabolite concentrations. The intracenter reproducibility of absolute NAA concentration, expressed as coefficient of variation (CV), was 1.8%. CV for the concentrations of creatine (Cr), choline (Cho), and myoinositol (MI) and for the ratios NAA/Cr, NAA/Cho, and MI/NAA varied by 11–16%. Intercenter CV was 3.9% for NAA and were below 10% for all other metabolites and metabolic ratios. Our study demonstrates that quantitative assessment of NAA with single-voxel MRS can be performed with high intercenter reproducibility. This is the basis for applying 1H-MRS in large-scale early recognition and treatment studies in MTL affecting disorders.


Similar content being viewed by others
References
Bonavita S, Di Salle F, Tedeschi G (1999) Proton MRS in neurological disorders. Eur J Radiol 30:125–131
Sijens P, Irwan R, Potze JH, Mostert JP, de Keyser J, Oudkerk M (2005) Analysis of the human brain in primary progressive multiple sclerosis with mapping of the spatial distributions using 1H MR spectroscopy and diffusion tensor imaging. Eur Radiol 15:1686–1693
Block W, Jessen F, Träber F et al (2002) Regional N-acetylaspartate reduction in the hippocampus detected with fast proton magnetic resonance spectroscopic imaging in patients with Alzheimer disease. Arch Neurol 59:828–834
Schuff N, Amend D, Ezekiel F et al (1997) Changes of hippocampal N-acetyl aspartate and volume in Alzheimer’s disease. A proton MR spectroscopic imaging and MRI study. Neurology 49:1513–1521
Jessen F, Block W, Träber F et al (2000) Proton MR spectroscopy detects a relative decrease of N-acetylaspartate in the medial temporal lobe of patients with AD. Neurology 55:684–688
Jessen F, Block W, Träber F et al (2001) Decrease of N-acetylaspartate in the MTL correlates with cognitive decline of AD patients. Neurology 57:930–932
Engelhardt E, Moreira DM, Laks J, Marinho VM, Rozenthal M, Oliveira AC (2001) [Alzheimer’s disease and magnetic resonance spectroscopy of the hippocampus]. Arq Neuropsiquiatr 59:865–870
Block W, Träber F, Kuhl CK et al (1995) [1H-MR spectroscopic imaging in patients with clinically diagnosed Alzheimer’s disease]. Röfo Fortschr Geb Röntgenstr Neuen Bildgeb Verfahr 163:230–237
Schuff N, Capizzano AA, Du AT et al (2002) Selective reduction of N-acetylaspartate in medial temporal and parietal lobes in AD. Neurology 58:928–935
Dixon RM, Bradley KM, Budge MM, Styles P, Smith AD (2002) Longitudinal quantitative proton magnetic resonance spectroscopy of the hippocampus in Alzheimer’s disease. Brain 125:2332–2341
Chantal S, Braun CM, Bouchard RW, Labelle M, Boulanger Y (2004) Similar 1H magnetic resonance spectroscopic metabolic pattern in the medial temporal lobes of patients with mild cognitive impairment and Alzheimer disease. Brain Res 1003:26–35
Krishnan KR, Charles HC, Doraiswamy PM et al (2003) Randomized, placebo-controlled trial of the effects of donepezil on neuronal markers and hippocampal volumes in Alzheimer’s disease. Am J Psychiatry 160:2003–2011
Sijens PE, Knopp MV, Brunetti A et al (1995) 1H MR spectroscopy in patients with metastatic brain tumors: a multicenter study. Magn Reson Med 33:818–826
Negendank WG, Sauter R, Brown TR et al (1996) Proton magnetic resonance spectroscopy in patients with glial tumors: a multicenter study. J Neurosurg 84:449–458
Paley M, Cozzonem PJ, Alonso J et al (1996) Multicenter proton magnetic resonance spectroscopy study of neurological complications of AIDS. AIDS Res Hum Retroviruses 12:213–222
Lee PL, Yiannoutsos CT, Ernst T et al (2003) A multicenter 1H MRS study of the AIDS dementia complex: validation and preliminary analysis. J Magn Reson Imaging 17:625–633
Holshouser BA, Komu M, Moller HE et al (1995) Localized proton NMR spectroscopy in the striatum of patients with idiopathic Parkinson’s disease: a multicenter pilot study. Magn Reson Med 33:589–594
Chard DT, McLean MA, Parker GJM, MacManus DG, Miller DH (2002) Reproducibility of in vivo metabolite quantification with proton magnetic resonance spectroscopic imaging. J Magn Reson Imaging 15:219–225
Komoroski RA, Kotrla KJ, Lemen L, Lindquist D, Diaz P, Foundas A (2004) Brain metabolite concentration ratios in vivo: multisite reproducibility by single-voxel 1H MR spectroscopy. Magn Reson Imaging 22:721–725
Podo F, Henriksen O, Bovee WM, Leach MO, Leibfritz D, de Certaines JD (1998) Absolute metabolite quantification by in vivo NMR spectroscopy: I. Introduction, objectives and activities of a concerted action in biomedical research. Magn Reson Imaging 16:1085–1092
Keevil SF, Barbiroli B, Brooks JC et al (1998) Absolute metabolite quantification by in vivo NMR spectroscopy: II. A multicentre trial of protocols for in vivo localised proton studies of human brain. Magn Reson Imaging 16:1093–1106
de Beer R, Barbiroli B, Gobbi G et al (1998) Absolute metabolite quantification by in vivo NMR spectroscopy: III. Multicentre 1H MRS of the human brain addressed by one and the same data-analysis protocol. Magn Reson Imaging 16:1107–1111
Bottomley PA (1987) Spatial localization in NMR spectroscopy. Ann NY Acad Sci 508:333–348
Vanhamme L, van den Boogaart A, van Huffel S (1997) Improved method for accurate and efficient quantification of MRS data with use of prior knowledge. J Magn Reson 129:35–43
van den Boogaart A, van Hecke P, van Huffel S, Graveron-Demilly D, van Ormondt D, de Beer R (1996) MRUI: a graphical user interface for accurate routine MRS data analysis. Proc 13th Ann Meeting ESMRMB Prague, Czech Republic, p.318 (abstract)
Bartha R, Drost DJ, Williamson PC (1999) Factors affecting the quantification of short echo in-vivo 1H MR spectra: prior knowledge, peak elimination, and filtering. NMR Biomed 12:205–216
Naressi A, Couturier C, Devos JM et al (2001) Java-based graphical user interface for the MRUI quantitation package. MAGMA 12:141–152
Gredal O, Rosenbaum S, Topp S, Karlsborg M, Strange P, Werdelin L (1997) Quantification of brain metabolites in amyotrophic lateral sclerosis by localized proton magnetic resonance spectroscopy. Neurology 48:878–881
Schirmer T, Auer DP (2000) On the reliability of quantitative clinical magnetic resonance spectroscopy of the human brain. NMR Biomed 13:28–36
Brooks WM, Friedman SD, Stidley CA (1999) Reproducibility of 1H-MRS in vivo. Magn Reson Med 41:193–197
Li BSY, Wang H, Gonen O (2003) Metabolite ratios to assumed stable creatine level may confound the quantification of proton brain MR spectroscopy. Magn Reson Imaging 21:923–928
Wellard RM, Briellmann RS, Jennings C, Jackson GD (2005) Physiologic variability of single-voxel proton MR spectroscopic measurements at 3T. Am J Neuroradiol 26:585–590
Kantarci K, Jack CR, Xu YC et al (2000) Regional metabolic patterns in mild cognitive impairment and Alzheimer’s disease: a 1H MRS study. Neurology 55:210–217
Shonk TK, Moats RA, Gifford P et al (1995) Probable Alzheimer disease: diagnosis with proton MR spectroscopy. Radiology 195:65–72
Moats RA, Ernst T, Shonk TK, Ross BD (1994) Abnormal cerebral metabolite concentrations in patients with probable Alzheimer disease. Magn Reson Med 32:110–115
Author information
Authors and Affiliations
Corresponding author
Additional information
The study is part of the German Research Network on Dementia and was funded by the German Federal Ministry for Education and Research (grant O1GI 0102)
Rights and permissions
About this article
Cite this article
Träber, F., Block, W., Freymann, N. et al. A multicenter reproducibility study of single-voxel 1H-MRS of the medial temporal lobe. Eur Radiol 16, 1096–1103 (2006). https://doi.org/10.1007/s00330-005-0108-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-005-0108-y