Abstract
Key message
Global transcriptome analysis revealed common regulons for biotic/abiotic stresses, and some of these regulons encoding signaling components in both stresses were newly identified in this study.
Abstract
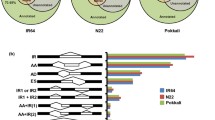
In this study, we aimed to identify plant responses to multiple stress conditions and discover the common regulons activated under a variety of stress conditions. Global transcriptome analysis revealed that salicylic acid (SA) may affect the activation of abiotic stress-responsive genes in pepper. Our data indicate that methyl jasmonate (MeJA) and ethylene (ET)-responsive genes were primarily activated by biotic stress, while abscisic acid (ABA)-responsive genes were activated under both types of stresses. We also identified differentially expressed gene (DEG) responses to specific stress conditions. Biotic stress induces more DEGs than those induced by abiotic and hormone applications. The clustering analysis using DEGs indicates that there are common regulons for biotic or abiotic stress conditions. Although SA and MeJA have an antagonistic effect on gene expression levels, SA and MeJA show a largely common regulation as compared to the regulation at the DEG expression level induced by other hormones. We also monitored the expression profiles of DEG encoding signaling components. Twenty-two percent of these were commonly expressed in both stress conditions. The importance of this study is that several genes commonly regulated by both stress conditions may have future applications for creating broadly stress-tolerant pepper plants. This study revealed that there are complex regulons in pepper plant to both biotic and abiotic stress conditions.


Similar content being viewed by others
References
Abuqamar S, Luo H, Laluk K, Mickelbart MV, Mengiste T (2009) Crosstalk between biotic and abiotic stress responses in tomato is mediated by the AIM1 transcription factor. Plant J 58:347–360
Anderson JP, Badruzsaufari E, Schenk PM, Manners JM, Desmond OJ, Ehlert C, Maclean DJ, Ebert PR, Kazan K (2004) Antagonistic interaction between abscisic acid and jasmonate–ethylene signaling pathways modulates defense gene expression and disease resistance in Arabidopsis. Plant Cell 16:3460–3479
Asselbergh B, De Vleesschauwer D, Höfte M (1992) Global switches and fine-tuning-ABA modulates plant pathogen defense. Mol Plant Microbe Interact 21:709–719
Atkinson NJ, Urwin PE (2012) The interaction of plant biotic and abiotic stresses: from genes to the field. J Exp Bot 63:3523–3543
Beaudoin N, Serizet C, Gosti F, Giraudat J (2000) Interactions between abscisic acid and ethylene signaling cascades. Plant Cell 12:1103–1115
Boudsocq M, Sheen J (2012) CDPKs in immune and stress signaling. Trends Plant Sci. doi:10.1016/j.tplants.2012.08.008
Cheong YH, Chang HS, Gupta R, Wang X, Zhu T, Luan S (2002) Transcriptional profiling reveals novel interactions between wounding, pathogen, abiotic stress, and hormonal responses in Arabidopsis. Plant Physiol 129:661–677
Chinnusamy V, Gong Z, Zhu JK (2008) Abscisic acid-mediated epigenetic processes in plant development and stress responses. J Integr Plant Biol 50:1187–1195
Dartan B (2004) A novel AP2 domain transcription factor from Lycopersicon esculentum, functions in tobacco mosaic virus pathogenesis. Dissertation, Sabanchi University
Davies PJ (2010) Their nature, occurrence, and functions. In: Davies PJ (ed) The plant hormones. Springer, Netherlands, pp 1–15
Eisen MB, Spellman PT, Brown PO, Botstein D (1998) Cluster analysis and display of genome-wide expression patterns. PNAS 95:14863–14868
Fraire-Velázquez S, Rodríguez-Guerra R, Sánchez-Calderón L (2011) Abiotic and biotic stress response crosstalk in plants. In: Shanker A, Venkateswarlu B (eds) Abiotic stress response in plants—physiological, biochemical and genetic perspectives. Intech, New York, pp 1–24
Fujita M, Fujita Y, Noutoshi Y, Takahashi F, Narusaka Y, Yamaguchi-Shinozaki K, Shinozaki K (2006) Crosstalk between abiotic and biotic stress responses: a current view from the points of convergence in the stress signaling networks. Curr Opin Plant Biol 9:436–442
Gago GM, Almoguera C, Jordano J, Gonzalez DH, Chan RL (2002) Hahb-4, a homeobox-leucine zipper gene potentially involved in abscisic acid-dependent responses to water stress in sunflower. Plant, Cell Environ 25:633–640
Glazebrook J (2005) Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu Rev Phytopathol 43:205–227
Hegedus D, Yu M, Baldwin D, Gruber M, Sharpe A, Parkin I, Whitwill S, Lydiate D (2003) Molecular characterization of Brassica napus NAC domain transcriptional activators induced in response to biotic and abiotic stress. Plant Mol Biol 53:383–397
Heidrich K, Blanvillain-Baufum S, Parker JE (2012) Molecular and spatial constraints on NB-LRR receptor signaling. Curr Opin Plant Biol 15:385–391
Herms DA, Mattson WJ (1992) The dilemma of plants: to grow or defend. Q Rev Biol 67:83–335
Kilian J, Whitehead D, Horak J, Wanke D, Weinl S, Batistic O, D’Angelo C, Bornberg-Bauer E, Kudla J, Harter K (2007) The AtGenExpress global stress expression data set: protocols, evaluation and model data analysis of UV-B light, drought and cold stress responses. Plant J 50:347–363
Knight H, Knight MR (2001) Abiotic stress signalling pathways: specificity and cross-talk. Trends Plant Sci 6:262–267
Koornneef A, Pieterse CM (2008) Cross talk in defense signaling. Plant Physiol 146:839–844
Lee S, Chung EJ, Joung YH, Choi D (2010) Non-climacteric fruit ripening in pepper: increased transcription of EIL-like genes normally regulated by ethylene. Funct Integr Genomics 10:135–146
Lee S, Hwang S, Seo Y, Jeon W, Oh B-J (2012) Molecular characterization of the AtCXE8 gene, which promotes resistance to Botrytis cinerea infection. Plant Biotechnol Rep. doi:10.1007/s11816-012-0253-0
Leek JT, Monsen EC, Dabney AR, Storey JD (2006) EDGE: extraction and analysis of differential gene expression. Bioinformatics 22:507–508
Liu H, Zhang H, Yang Y, Li G, Wang X, Basnayake BM, Li D, Song F (2008) Functional analysis reveals pleiotropic effects of rice RING-H2 finger protein gene OsBIRF1 on regulation of growth and defense responses against abiotic and biotic stresses. Plant Mol Biol 68:17–30
Mittler R (2006) Abiotic stress, the field environment and stress combination. Trends Plant Sci 11:15–19
Nakaminami K, Matsui A, Shinozaki K, Seki M (2012) RNA regulation in plant abiotic stress responses. Biochim Biophys Acta 1819:149–153
Park C-J, Shin R, Park J, Lee G-J, You J-S, Paek K-H (2002) Induction of pepper cDNA encoding a lipid transfer protein during the resistance response to tobacco mosaic virus. Plant Mol Biol 48:243–254
Pieterse CM, van Loon LC (1999) Salicylic acid-independent plant defence pathways. Trends Plant Sci 4:52–58
Pieterse CM, Leon-Reyes A, Van der Ent S, Van Wees SC (2009) Networking by small-molecule hormones in plant immunity. Nat Chem Biol 5:308–316
Rizhsky L, Liang H, Shuman J, Shulaev V, Davletova S, Mittler R (2004) When defense pathways collide. The response of Arabidopsis to a combination of drought and heat stress. Plant Physiol 134:1683–1696
Saeed AI, Sharov V, White J, Li J, Liang W, Bhagabati N, Braisted J, Klapa M, Currier T, Thiagarajan M, Sturn A, Snuffin M, Rezantsev A, Popov D, Ryltsov A, Kostukovich E, Borisovsky I, Liu Z, Vinsavich A, Trush V, Quackenbush J (2003) TM4: a free, open-source system for microarray data management and analysis. Biotechniques 34:2003
Seki M, Narusaka M, Ishida J, Nanjo T, Fujita M, Oono Y, Kamiya A, Nakajima M, Enju A, Sakurai T, Satou M, Akiyama K, Taji T, Yamaguchi-Shinozaki K, Carninci P, Kawai J, Hayashizaki Y, Shinozaki K (2002) Monitoring the expression profiles of 7000 Arabidopsis genes under drought, cold and high-salinity stresses using a full-length cDNA microarray. Plant J 31:279–292
Shah J (2003) The salicylic acid loop in plant defense. Curr Opin Plant Biol 6:365–371
Singh K, Foley RC, Oñate-Sánchez L (2002) Transcription factors in plant defense and stress responses. Curr Opin Plant Biol 5:430–436
Spoel SH, Dong X (2008) Making sense of hormone crosstalk during plant immune responses. Cell Host Microbe 3:348–351
Sunkar R, Li YF, Jagadeeswaran G (2012) Functions of microRNAs in plant stress responses. Trends Plant Sci 17:196–203
Swindell WR (2006) The association among gene expression responses to nine abiotic stress treatments in Arabidopsis thaliana. Genetics 174:1811–1824
Thurow C, Schiermeyer A, Krawczyk S, Butterbrodt T, Nickolov K, Gatz C (2005) Tobacco bZIP transcription factor TGA2.2 and related factor TGA2.1 have distinct roles in plant defense responses and plant development. Plant J 44:100–113
Tusher VG, Tibshirani R, Chu G (2001) Significance analysis of microarrays applied to the ionizing radiation response. PNAS 98:5116–5121
Wang W, Vinocur B, Altman A (2003) Plant responses to drought, salinity and extreme temperatures: towards genetic engineering for stress tolerance. Planta 218:1–14
Yoon J, Chung WI, Choi D (2009) NbHB1, Nicotiana benthamiana homeobox 1, is a jasmonic acid-dependent positive regulator of pathogen-induced plant cell death. New Phytol 184:71–84
Yuan S, Lin HH (2008) Role of salicylic acid in plant abiotic stress. Z Naturforsch C 63:313–320
Zhang H, Huang Z, Xie B, Chen Q, Tian X, Zhang X, Lu X, Huang D, Huang R (2004) The ethylene-, jasmonate-, abscisic acid- and NaCl-responsive tomato transcription factor JERF1 modulates expression of GCC box-containing genes and salt tolerance in tobacco. Planta 220:262–270
Acknowledgments
We thank Professor CJ. Park for his useful discussions and critical reading of this manuscript. This work was supported by a faculty research fund for S. Lee from Sejong University in 2011.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by E. Guiderdoni.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lee, S., Choi, D. Comparative transcriptome analysis of pepper (Capsicum annuum) revealed common regulons in multiple stress conditions and hormone treatments. Plant Cell Rep 32, 1351–1359 (2013). https://doi.org/10.1007/s00299-013-1447-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-013-1447-9