Abstract
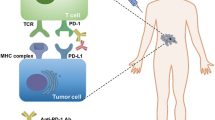
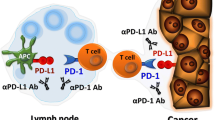
Tumor cells can evade immune surveillance through overexpressing the ligands of checkpoint receptors on tumor cells or adjacent cells, leading T cells to anergy or exhaustion. Growing evidence of the interaction between tumor cells and microenvironment promoted the emergence of immune-checkpoint blockade. By targeting programmed cell death-1 (PD-1) pathway, cytotoxic activity of T cell is enhanced significantly and tumor cell lysis is induced subsequently. Currently, various antibodies against PD-1 and programmed death-ligand 1 (PD-L1) are under clinical studies in lymphomas. In this review, we outline the rationale for investigation of PD-1-PD-L1 immune-checkpoint blockade in lymphomas and discuss their prospect of applications in clinical treatment.

Similar content being viewed by others
References
Hude I, Sasse S, Engert A, Brockelmann PJ (2017) The emerging role of immune checkpoint inhibition in malignant lymphoma. Haematologica 102(1):30–42. https://doi.org/10.3324/haematol.2016.150656
Ansell SM, Lesokhin AM, Borrello I, Halwani A, Scott EC, Gutierrez M, Schuster SJ, Millenson MM, Cattry D, Freeman GJ, Rodig SJ, Chapuy B, Ligon AH, Zhu L, Grosso JF, Kim SY, Timmerman JM, Shipp MA, Armand P (2015) PD-1 blockade with nivolumab in relapsed or refractory Hodgkin’s lymphoma. N Engl J Med 372(4):311–319. https://doi.org/10.1056/NEJMoa1411087
Armand P, Shipp MA, Ribrag V, Michot J, Zinzani PL, Kuruvilla J, Snyder E, Ricart AD, Balakumaran A, Rose S, Moskowitz CH (2016) Programmed death-1 blockade with pembrolizumab in patients with classical Hodgkin lymphoma after brentuximab vedotin failure. J Clin Oncol 34(31):3733–3739. https://doi.org/10.1200/JCO.2016.67.3467
Lesokhin AM, Ansell SM, Armand P, Scott EC, Halwani A, Gutierrez M, Millenson MM, Cohen AD, Schuster SJ, Lebovic D, Dhodapkar MV, Avigan D, Chapuy B, Ligon AH, Freeman GJ, Rodig SJ, Cattry D, Zhu L, Grosso JF, Garelik MB, Shipp MA, Borrello I, Timmerman JM (2016) Nivolumab in patients with relapsed or refractory hematologic malignancy: preliminary results of a phase Ib study. J Clin Oncol 34:2698–2704
Zinzani PL, Ribrag V, Moskowitz CH, Michot JM, Kuruvilla J, Balakumaran A, Zhang Y, Chlosta S, Shipp MA, Armand P (2017) Safety & tolerability of pembrolizumab in patients with relapsed/refractory primary mediastinal large B-cell lymphoma. Blood 130(3):267–270. https://doi.org/10.1182/blood-2016-12-758383
Ding W, Rademacher JL, Call TG, Parikh SA, Leis JF, Shanafelt TD, He R, Habermann TM, Lin Y, Laplant BR, Feldman AL, Witzig TE, Chanankhan AA, Asmus E, Dyke DLV, Conte MJ, Bowen DA, Sinha S, Zhang H, Liu X, Secreto CR, Tian S, Braggio E, Micallef IN, Yan H, Kay NE, Dong H, Ansell SM (2016) PD-1 blockade with pembrolizumab in relapsed CLL including Richter’s transformation: an update report from a phase 2 trial (MC1485). Blood 128:4392
Greaves P, Gribben JG (2013) The role of B7 family molecules in hematologic malignancy. Blood 121(5):734–744. https://doi.org/10.1182/blood-2012-10-385591
Schwartz RH (2003) T cell anergy. Annu Rev Immunol 21(1):305–334. https://doi.org/10.1146/annurev.immunol.21.120601.141110
Gardner D, Jeffery LE, Sansom DM (2014) Understanding the CD28/CTLA-4 (CD152) pathway and its implications for costimulatory blockade. Am J Transplant 14(9):1985–1991. https://doi.org/10.1111/ajt.12834
Chen L, Flies DB (2013) Molecular mechanisms of T cell co-stimulation and co-inhibition. Nat Rev Immunol 13(4):227–242. https://doi.org/10.1038/nri3405
Keir ME, Butte MJ, Freeman GJ, Sharpe AH (2008) PD-1 and its ligands in tolerance and immunity. Annu Rev Immunol 26(1):677–704. https://doi.org/10.1146/annurev.immunol.26.021607.090331
Pauken KE, Wherry EJ (2015) Overcoming T cell exhaustion in infection and cancer. Trends Immunol 36(4):265–276. https://doi.org/10.1016/j.it.2015.02.008
Wherry EJ, Kurachi M (2015) Molecular and cellular insights into T cell exhaustion. Nat Rev Immunol 15(8):486–499. https://doi.org/10.1038/nri3862
Yamamoto R, Nishikori M, Kitawaki T, Sakai T, Hishizawa M, Tashima M, Kondo T, Ohmori K, Kurata M, Hayashi T, Uchiyama T (2008) PD-1-PD-1 ligand interaction contributes to immunosuppressive microenvironment of Hodgkin lymphoma. Blood 111(6):3220–3224. https://doi.org/10.1182/blood-2007-05-085159
Greaves P, Clear A, Owen A, Iqbal S, Lee A, Matthews J, Wilson A, Calaminici M, Gribben JG (2013) Defining characteristics of classical Hodgkin lymphoma microenvironment T-helper cells. Blood 122(16):2856–2863. https://doi.org/10.1182/blood-2013-06-508044
Shi M, Roemer MG, Chapuy B, Liao X, Sun H, Pinkus GS, Shipp MA, Freeman GJ, Rodig SJ (2014) Expression of programmed cell death 1 ligand 2 (PD-L2) is a distinguishing feature of primary mediastinal (thymic) large B-cell lymphoma and associated with PDCD1LG2 copy gain. Am J Surg Pathol 38(12):1715–1723. https://doi.org/10.1097/PAS.0000000000000297
Chapuy B, Roemer MG, Stewart C, Tan Y, Abo RP, Zhang L, Dunford AJ, Meredith DM, Thorner AR, Jordanova ES, Liu G, Feuerhake F, Ducar MD, Illerhaus G, Gusenleitner D, Linden EA, Sun HH, Homer H, Aono M, Pinkus GS, Ligon AH, Ligon KL, Ferry JA, Freeman GJ, Hummelen PV, Golub TR, Getz G, Roding SJ, Jong DD, Monti S, Shipp MA (2016) Targetable genetic features of primary testicular and primary central nervous system lymphomas. Blood 127(7):869–881. https://doi.org/10.1182/blood-2015-10-673236
Kiyasu J, Miyoshi H, Hirata A, Arakawa F, Ichikawa A, Niino D, Sugita Y, Yufu Y, Choi I, Abe Y, Uike N, Nagafuji K, Okamura T, Akashi K, Takayanagi R, Shiratsuchi M, Ohshima K (2015) Expression of programmed cell death ligand 1 is associated with poor overall survival in patients with diffuse large B-cell lymphoma. Blood 126(19):2193–2201. https://doi.org/10.1182/blood-2015-02-629600
Twa DD, Chan FC, Benneriah S, Woolcock B, Mottok A, Tan KL, Slack GW, Gunawardana J, Lim RS, Mcpherson A, Kridel R, Telenius A, Scott DW, Savage KJ, Shah SP, Gascoyne RD, Steidl C (2014) Genomic rearrangements involving programmed death ligands are recurrent in primary mediastinal large B-cell lymphoma. Blood 123(13):2062–2065. https://doi.org/10.1182/blood-2013-10-535443
Twa DD, Mottok A, Chan FC, Benneriah S, Woolcock B, Tan KL, Mungall AJ, Mcdonald H, Zhao Y, Lim RS, Nelson BH, Milne K, Shah SP, Morin RD, Marra MA, Scott DW, Gascoyne RD, Steidl C (2015) Recurrent genomic rearrangements in primary testicular lymphoma. J Pathol 236(2):136–141. https://doi.org/10.1002/path.4522
Yang Z, Novak AJ, Stenson MJ, Witzig TE, Ansell SM (2006) Intratumoral CD4+CD25+ regulatory T-cell-mediated suppression of infiltrating CD4+ T cells in B-cell non-Hodgkin lymphoma. Blood 107(9):3639–3646. https://doi.org/10.1182/blood-2005-08-3376
Tian C, Yang H, Zhu L, Zhang Q, Cao Z, Zhang Y (2017) Anti-CD138 chimeric antigen receptor-modified T-cell therapy for multiple myeloma with extensive extramedullary involvement. Ann Hematol 96(8):1407–1410. https://doi.org/10.1007/s00277-017-3029-3
Carreras J, Lopez-Guillermo A, Roncador G, Villamor N, Colomo L, Martinez A, Hamoudi R, Howat WJ, Montserrat E, Campo E (2009) High numbers of tumor-infiltrating programmed cell death 1-positive regulatory lymphocytes are associated with improved overall survival in follicular lymphoma. J Clin Oncol 27(9):1470–1476. https://doi.org/10.1200/JCO.2008.18.0513
Xerri L, Chetaille B, Serriari N, Attias C, Guillaume Y, Arnoulet C, Olive D (2008) Programmed death 1 is a marker of angioimmunoblastic T-cell lymphoma and B-cell small lymphocytic lymphoma/chronic lymphocytic leukemia. Hum Pathol 39(7):1050–1058. https://doi.org/10.1016/j.humpath.2007.11.012
Brusa D, Serra S, Coscia M, Rossi D, Darena G, Laurenti L, Jaksic O, Fedele G, Inghirami G, Gaidano G, Malavasi F, Deaglio S (2013) The PD-1/PD-L1 axis contributes to T-cell dysfunction in chronic lymphocytic leukemia. Haematologica 98(6):953–963. https://doi.org/10.3324/haematol.2012.077537
Grzywnowicz M, Karczmarczyk A, Skorka K, Zajac M, Zaleska J, Chocholska S, Tomczak W, Giannopoulos K (2015) Expression of programmed death 1 ligand in different compartments of chronic lymphocytic leukemia. Acta Haematol 134(4):255–262. https://doi.org/10.1159/000430980
Shimauchi T, Kabashima K, Nakashima D, Sugita K, Yamada Y, Hino R, Tokura Y (2007) Augmented expression of programmed death-1 in both neoplastic and nonneoplastic CD4+ T-cells in adult T-cell leukemia/lymphoma. Int J Cancer 121:2585–2590
Wang L, Qian J, Lu Y, Li H, Bao H, He D, Liu Z, Zheng Y, He J, Li Y, Neelapu S, Yang J, Kwak LW, Yi Q, Cai Z (2013) Immune evasion of mantle cell lymphoma: expression of B7-H1 leads to inhibited T-cell response to and killing of tumor cells. Haematologica 98(9):1458–1466. https://doi.org/10.3324/haematol.2012.071340
Dorfman DM, Brown JA, Shahsafaei A, Freeman GJ (2006) Programmed death-1 (PD-1) is a marker of germinal center-associated T cells and angioimmunoblastic T-cell lymphoma. Am J Surg Pathol 30(7):802–810. https://doi.org/10.1097/01.pas.0000209855.28282.ce
Wilcox RA, Feldman AL, Wada DA, Yang Z, Comfere NI, Dong H, Kwon ED, Novak AJ, Markovic SN, Pittelkow MR, Witzig TE, Ansell SM (2009) B7-H1 (PD-L1, CD274) suppresses host immunity in T-cell lymphoproliferative disorders. Blood 114(10):2149–2158. https://doi.org/10.1182/blood-2009-04-216671
Han L, Liu F, Li R, Li Z, Chen X, Zhou Z, Zhang X, Hu T, Zhang Y, Young K, Sun S, Wen J, Zhang M (2014) Role of programmed death ligands in effective T-cell interactions in extranodal natural killer/T-cell lymphoma. Oncol Lett 8(4):1461–1469. https://doi.org/10.3892/ol.2014.2356
Kantekure K, Yang Y, Raghunath P, Schaffer A, Woetmann A, Zhang Q, Odum N, Wasik M (2012) Expression patterns of the immunosuppressive proteins PD-1/CD279 and PD-L1/CD274 at different stages of cutaneous T-cell lymphoma/mycosis fungoides. Am J Dermatopathol 34(1):126–128. https://doi.org/10.1097/DAD.0b013e31821c35cb
Munir S, Andersen GH, Woetmann A, Odum N, Becker JC, Andersen MH (2013) Cutaneous T cell lymphoma cells are targets for immune checkpoint ligand PD-L1-specific, cytotoxic T cells. Leukemia 27(11):2251–2253. https://doi.org/10.1038/leu.2013.118
Miyoshi H, Kiyasu J, Kato T, Yoshida N, Shimono J, Yokoyama S, Taniguchi H, Sasaki Y, Kurita D, Kawamoto K, Kato K, Imaizumi Y, Seto M, Ohshima K (2016) PD-L1 expression on neoplastic or stromal cell is respectively poor or good prognostic factor for adult T-cell leukemia/lymphoma. Blood 128(10):1374–1381. https://doi.org/10.1182/blood-2016-02-698936
Muenst S, Hoeller S, Dirnhofer S, Tzankov A (2009) Increased programmed death-1+ tumor-infiltrating lymphocytes in classical Hodgkin lymphoma substantiate reduced overall survival. Hum Pathol 40(12):1715–1722. https://doi.org/10.1016/j.humpath.2009.03.025
Roemer MG, Advani RH, Ligon AH, Natkunam Y, Redd RA, Homer H, Connelly CF, Sun H, Daadi SE, Freeman GJ, Armand P, Chapuy B, Jong DD, Hoppe RT, Neuberg D, Rodig SJ, Shipp MA (2016) PD-L1 and PD-L2 genetic alterations define classical Hodgkin lymphoma and predict outcome. J Clin Oncol 34(23):2690–2697. https://doi.org/10.1200/JCO.2016.66.4482
Menter T, Bodmerhaecki A, Dirnhofer S, Tzankov A (2016) Evaluation of the diagnostic and prognostic value of PDL1 expression in Hodgkin and B-cell lymphomas. Hum Pathol 54:17–24. https://doi.org/10.1016/j.humpath.2016.03.005
Tian C, Zheng G, Zhuang H, Li X, Hu D, Zhu L, Wang T, You M, Zhang Y (2017) MicroRNA-494 activation suppresses bone marrow stromal cell-mediated drug resistance in acute myeloid leukemia cells. J Cell Physiol 232(6):1387–1395. https://doi.org/10.1002/jcp.25628
Richendollar BG, Pohlman B, Elson P, Hsi ED (2011) Follicular programmed death 1-positive lymphocytes in the tumor microenvironment are an independent prognostic factor in follicular lymphoma. Hum Pathol 42(4):552–557. https://doi.org/10.1016/j.humpath.2010.08.015
Kosmaczewska A, Ciszak L, Suwalska K, Wolowiec D, Frydecka I (2005) CTLA-4 overexpression in CD19+/CD5+ cells correlates with the level of cell cycle regulators and disease progression in B-CLL patients. Leukemia 19(2):301–304. https://doi.org/10.1038/sj.leu.2403588
Riches JC, Davies JK, Mcclanahan F, Fatah R, Iqbal S, Agrawal SG, Ramsay AG, Gribben JG (2013) T cells from CLL patients exhibit features of T-cell exhaustion but retain capacity for cytokine production. Blood 121(9):1612–1621. https://doi.org/10.1182/blood-2012-09-457531
Green MR, Rodig SJ, Juszczynski P, Ouyang J, Sinha P, Odonnell E, Neuberg D, Shipp MA (2012) Constitutive AP-1 activity and EBV infection induce PD-L1 in Hodgkin lymphomas and posttransplant lymphoproliferative disorders: implications for targeted therapy. Clin Cancer Res 18(6):1611–1618. https://doi.org/10.1158/1078-0432.CCR-11-1942
Sagivbarfi I, Kohrt HE, Czerwinski DK, Ng PP, Chang BY, Levy R (2015) Therapeutic anti-tumor immunity by checkpoint blockade is enhanced by ibrutinib, an inhibitor of both BTK and ITK. Proc Natl AcadSci U S A112:E966–E972
Tian C, Wang X, Zhang Y (2016) Glomerular infiltration by intravascular large B-cell lymphoma. Br J Haematol 174(6):833. https://doi.org/10.1111/bjh.14204
Nishijima TF, Shachar SS, Nyrop KA, Muss HB (2017) Safety and tolerability of PD-1/PD-L1 inhibitors compared with chemotherapy in patients with advanced cancer: a meta-analysis. Oncologist 22(4):470–479. https://doi.org/10.1634/theoncologist.2016-0419
Atkins MB, Clark JI, Quinn DI (2017) Immune checkpoint inhibitors in advanced renal cell carcinoma: experience to date and future directions. Ann Oncol 28(7):1484–1494. https://doi.org/10.1093/annonc/mdx151
Massard C, Gordon MS, Sharma S, Rafii S, Wainberg ZA, Luke J, Curiel TJ, Colon-Otero G, Hamid O, Sanborn RE, O'Donnell PH, Drakaki A, Tan W, Kurland JF, Rebelatto MC, Jin X, Blake-Haskins JA, Gupta A, Segal NH (2016) Safety and efficacy of durvalumab (MEDI4736), an anti-programmed cell death Ligand-1 immune checkpoint inhibitor, in patients with advanced urothelial bladder cancer. J Clin Oncol 34(26):3119–3125. https://doi.org/10.1200/JCO.2016.67.9761
Moskowitz CH, Ribrag V, Michot JM, Martinelli G, Zinzani PL, Gutierrez M, Maeyer GD, Jacob AG, GiallellaK AJW, Derosier M, Wang J, Yang Z, Rubin EH, Rose S, Shipp MA, Armand P (2014) PD-1 blockade with the monoclonal anti-body pembrolizumab (MK-3475) in patients with classical Hodgkin lymphoma after brentuximab vedotin failure: preliminary results from a phase 1b study (keynote-013). Blood 124:290
Lesokhin AM, Ansell SM, Armand P, Scott EC, Halwani A, Gutierrez M, Millenson MM, Cohen AD, Schuster SJ, Lebovic D, Dhodapkar MV, Avigan D, Chapuy B, Ligon AH, Rodig SJ, Cattry D, Zhu L, Grosso JF, Kim SY, Shipp MA, Borrello I, Timmerman JM (2014) Preliminary results of a phase I study of nivolumab (BMS-936558) in patients with relapsed or refractory lymphoid malignancies. Blood 124:291–291
Ansell S, Armand P, Timmerman JM, Shipp MA, Garelik MB, Zhu L, Lesokhin AM (2015) Nivolumab in patients (pts) with relapsed or refractory classical Hodgkin lymphoma (R/R cHL): clinical outcomes from extended follow-up of a phase 1 study (CA209-039). Blood 126:583
Hawkes EA, Grigg A, Chong G (2015) Programmed cell death-1 inhibition in lymphoma. Lancet Oncol 16:234–245
Johnson DB, Balko JM, Compton ML, Chalkias S, Gorham J, Xu Y, Hicks M, Puzanov I, Alexander MR, Bloomer TL, Becker JR, Slosky DA, Phillips EJ, Pilkinton MA, Craig-Owens L, Kola N, Plautz G, Reshef DS, Deutsch JS, Deering RP, Olenchock BA, Lichtman AH, Roden DM, Seidman CE, Koralnik IJ, Seidman JG, Hoffman RD, Taube JM, Diaz LA Jr, Anders RA, Sosman JA, Moslehi JJ (2016) Fulminant myocarditis with combination immune checkpoint blockade. N Engl J Med 375(18):1749–1755. https://doi.org/10.1056/NEJMoa1609214
Jelinek T, Mihalyova J, Kascak M, Duras J, Hajek R (2017) PD-1/PD-L1 inhibitors in haematological malignancies: update 2017. Immunology 152(3):357–371. https://doi.org/10.1111/imm.12788
Osta BE, Hu F, Sadek RF (2016) Immune checkpoint inhibitors (ICI): a meta-analysis of immune-related adverse events (irAE) from cancer clinical trials. ASCO
Larkin J, Chiarionsileni V, Gonzalez R, Grob JJ, Cowey CL, Lao CD, Schadendorf D, Dummer R, Smylie M, Rutkowski P, Ferrucci PF, Hill A, Wagstaff J, Carlino MS, Haanen JB, Maio M, Marquezrodas I, Mcarthur GA, Ascierto PA, LongGV CMK, Postow MA, Grossmann KF, Sznol M, Dreno B, Bastholt L, Yang A, Rollin L, Horak CE, Hodi FS, Wolchok JD (2015) Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N Engl J Med 373(1):23–34. https://doi.org/10.1056/NEJMoa1504030
Herbaux C, Gauthier J, Brice P, Fornecker L, Bouabdallah K, Manson GF, Ghesquieres H, Thiebautbertrand A, Demarquette H, Boyle EM, Ysebaert L, Houot R, Yakoubagha I, Morschhauser F (2015) Nivolumab is effective and reasonably safe in relapsed or refractory Hodgkin’s lymphoma after allogeneic hematopoietic cell transplantation: a study from the Lysa and SFGM-TC. Blood 126:3979–3979
Funding
This work was supported by grants 81670104 and 81570201 from the National Natural Science Foundation of China (NSFC).
Author information
Authors and Affiliations
Contributions
Y. W. and L. W. drafted the paper. C. T. and Y. Z. critically revised the paper. All authors approved all versions including the final version and are responsible for the accuracy and integrity of all aspects of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Wang, Y., Wu, L., Tian, C. et al. PD-1-PD-L1 immune-checkpoint blockade in malignant lymphomas. Ann Hematol 97, 229–237 (2018). https://doi.org/10.1007/s00277-017-3176-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-017-3176-6