Abstract
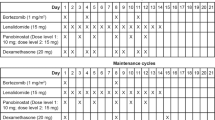
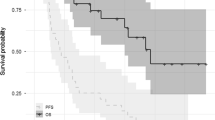
The clinical efficacy and safety of a four-drug combination of bortezomib, cyclophosphamide, thalidomide, and dexamethasone was assessed for patients with relapsed or refractory multiple myeloma. Seventy patients received at least two cycles of treatment with bortezomib 1.3 mg/m2 intravenously on days 1, 4, 8, and 11; cyclophosphamide 150 mg/m2 orally on days 1–4; thalidomide 50 mg/day orally every day; and dexamethasone 20 mg/m2 intravenously on days 1, 4, 8, and 11. The overall best response rate was 88%, with 46% complete response, 9% very good partial response, and 33% partial response. After a median follow-up of 12.6 months, the median progression-free survival (PFS) was 14.6 months with a 3-year PFS of 14% and the median overall survival (OS) was 31.6 months with a 3-year OS of 47%. Grade 3 or 4 adverse events included thrombocytopenia (12%), neutropenia (4%), peripheral sensory neuropathy (3%), with thrombosis being very rare (<1%). Bortezomib combined with cyclophosphamide, thalidomide, and dexamethasone is a highly effective salvage therapy with manageable toxicity for patients with relapsed or refractory multiple myeloma.




Similar content being viewed by others
References
Kyle RA, Rajkumar SV (2004) Multiple myeloma. N Engl J Med 351:1860–1873
Galton DA, Peto R (1968) A progress report on the Medical Research Council's therapeutic trial in myelomatosis. Br J Haematol 15:319–320
Sirohi B, Powles R (2004) Multiple myeloma. Lancet 363:875–887
Alexanian R, Barlogie B, Tucker S (1990) VAD-based regimens as primary treatment for multiple myeloma. Am J Hematol 33:86–89
Barlogie B, Jagannath S, Desikan KR et al (1999) Total therapy with tandem transplants for newly diagnosed multiple myeloma. Blood 93:55–65
Samson D, Gaminara E, Newland A et al (1989) Infusion of vincristine and doxorubicin with oral dexamethasone as first-line therapy for multiple myeloma. Lancet 2:882–885
Sirohi B, Powles R, Mehta J et al (1999) Complete remission rate and outcome after intensive treatment of 177 patients under 75 years of age with IgG myeloma defining a circumscribed disease entity with a new staging system. Br J Haematol 107:656–666
Goldschmidt H, Hegenbart U, Wallmeier M et al (1997) Factors influencing collection of peripheral blood progenitor cells following high-dose cyclophosphamide and granulocyte colony stimulating factor in patients with multiple myeloma. Br J Haematol 98:736–744
Richardson PG, Hideshima T, Mitsiades C et al (2007) The emerging role of novel therapies for the treatment of relapsed myeloma. J Natl Compr Canc Netw 5:149–162
San Miguel JF, Schlag R, Khuageva NK et al (2008) Bortezomib plus melphalan and prednisone for initial treatment of multiple myeloma. N Engl J Med 359:906–917
Dimopoulos M, Spencer A, Attal M et al (2007) Lenalidomide plus dexamethasone for relapsed or refractory multiple myeloma. N Engl J Med 357:2123–2132
Weber DM, Chen C, Niesvizky R et al (2007) Lenalidomide plus dexamethasone for relapsed multiple myeloma in North America. N Engl J Med 357:2133–2142
Richardson PG, Barlogie B, Berenson J et al (2003) A phase 2 study of bortezomib in relapsed, refractory myeloma. N Engl J Med 348:2609–2617
Jagannath S, Barlogie B, Berenson J (2004) A phase 2 study of two doses of bortezomib in relapsed or refractory myeloma. Br J Haematol 127:165–172
Richardson PG, Sonneveld P, Schuster M et al (2007) Extended follow-up of a phase 3 trial in relapsed multiple myeloma: final time-to-event results of the APEX trial. Blood 110:3557–3560
Wang LM, Weber DM, Delasalle KB et al (2004) VTD (Velcade, thalidomide, dexamethasone) as primary therapy for newly diagnosed multiple myeloma. Blood 104:210a
Oakervee HE, Popat R, Curry N et al (2005) PAD combination therapy (PS-341/bortezomib, doxorubicin and dexamethasone) for previously untreated patients with multiple myeloma. Br J Haematol 129:755–762
Dimopoulos MA, Zervas K, Kouvatseas G et al (2001) Thalidomide and dexamethasone combination for refractory multiple myeloma. Ann Oncol 12:991–995
Weber DM, Rankin K, Gavino M et al (2000) Thalidomide with dexamethasone for resistant multiple myeloma. Blood 96:167a
Palumbo A, Giaccone L, Bertola A et al (2001) Low-dose thalidomide plus dexamethasone is an effective salvage therapy for advanced myeloma. Haematologica 86:399–403
Garcia-Sanz R, Gonzalez-Fraile MI, Sierra M, Lopez C et al (2003) The combination of thalidomide, cyclophosphamide and dexamethasone (ThaCyDex) is feasible and can be an option for relapsed/refractory multiple myeloma. Hematol J 3:43–48
Srkalovic G, Elson P, Trebisky B et al (2002) Use of melphalan, thalidomide, and dexamethasone in treatment of refractory and relapsed multiple myeloma. Med Oncol 19:219–226
Richardson PG, Briemberg H, Jagannath S et al (2006) Frequency, characteristics, and reversibility of peripheral neuropathy during treatment of advanced multiple myeloma with bortezomib. J Clin Oncol 24:3113–3120
Durie BG, Harousseau JL, Miguel JS et al (2006) International uniform response criteria for multiple myeloma. Leukemia 20:1467–1473
Chanan-Khan A, Sonneveld P, Schuster MW et al (2008) Analysis of herpes zoster events among bortezomib-treated patients in the phase III APEX study. J Clin Oncol 26:4784–4790
Terpos E, Kastritis E, Roussou M et al (2008) The combination of bortezomib, melphalan, dexamethasone and intermittent thalidomide is an effective regimen for relapsed/refractory myeloma and is associated with improvement of abnormal bone metabolism and angiogenesis. Leukemia 22:2247–2256
Palumbo A, Ambrosini MT, Benevolo G et al (2007) Bortezomib, melphalan, prednisone, and thalidomide for relapsed multiple myeloma. Blood 109:2767–2772
Musto P, Falcone A, Sanpaolo G et al (2006) Bortezomib (Velcade) for progressive myeloma after autologous stem cell transplantation and thalidomide. Leuk Res 30:283–285
Popat R, Oakervee H, Williams C et al (2009) Bortezomib, low-dose intravenous melphalan, and dexamethasone for patients with relapsed multiple myeloma. Br J Haematol 144:887–894
Reece DE, Rodriguez GP, Chen C et al (2008) Phase I-II trial of bortezomib plus oral cyclophosphamide and prednisone in relapsed and refractory multiple myeloma. J Clin Oncol 26:4777–4783
Acknowledgments
The authors thank Dr. Paul Richardson, Dana-Faber Cancer Institute, for critically review the manuscript.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, YK., Sohn, SK., Lee, JH. et al. Clinical efficacy of a bortezomib, cyclophosphamide, thalidomide, and dexamethasone (Vel-CTD) regimen in patients with relapsed or refractory multiple myeloma: a phase II study. Ann Hematol 89, 475–482 (2010). https://doi.org/10.1007/s00277-009-0856-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-009-0856-x