Abstract
Background
The thick-skinned nose is still one of the most challenging aspects of rhinoplasty. The possible effects of oral isotretinoin on cosmetic results of rhinoplasty in patients with thick nasal skin have been considered during recent years.
Methods
In this double-blind placebo-controlled clinical trial, 48 cases were divided into two groups randomly. Oral isotretinoin (0.5-mg/kg) was started on the 31st day after surgery and given every other day for 1 month and after that daily for two additional months in the first group. The second group received a placebo in the same form, sequence and interval as the first group. The cosmetic results based on patient satisfaction and ranking by an expert surgeon were compared between the two groups at 3 months, 6 months and 1 year after surgery.
Results
Patient satisfaction and ranking by an expert surgeon in the isotretinoin group at 3 months and 6 months after surgery were significantly better than in the placebo group (p value < 0.05); however, at 12 months after surgery there was no statistically significant difference between the two groups (p value > 0.05).
Conclusion
Though postoperative use of oral isotretinoin in patients with thick nasal skin accelerates improvement in cosmetic results during the early months after surgery, it does not significantly affect the final cosmetic result 1 year after surgery.
Level of Evidence II
This journal requires that authors assign a level of evidence to each article. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266.
Similar content being viewed by others
Introduction
Rhinoplasty is one of the most popular facial plastic surgeries. In some races, such as African, African-American and middle-eastern people, a noticeable percent of cases that request rhinoplasty have thick nasal skin. On the other hand, thick skin rhinoplasty is still one of the most challenging topics with a paucity of relevant articles [1]. Patients with thick nasal skin usually also have oily skin with sebaceous hyperactivity that makes them acne prone. The possible effect of oral isotretinoin on the cosmetic results of rhinoplasty in patients with thick nasal skin has been considered by some authors during recent years, but as yet no placebo-controlled clinical trials have been published [1]. This study was designed to determine whether oral isotretinoin usage after rhinoplasty improves the cosmetic results in patients with thick skin nose.
Methods
The present study was a prospective double-blind placebo-controlled clinical trial. Patients from 17 to 45 years old who were candidates for septorhinoplasty in a tertiary referral university hospital from 2014 through 2016 were enrolled.
Inclusion criteria were acceptance of participators by signed written consent after complete description of the research and having a thick-skinned nose based on expert dermatologist examinations. Exclusion criteria of this study included absolute contraindication of isotretinoin (pregnancy, breast feeding and allergy to soya, peanut or paraben), inflammatory bowel disease, history of previous rhinoplasty and cases who decided to exit from the research.
The otorhinolaryngologist who evaluated outcomes, the surgeon, the dermatologist and the photographer were all blinded to the patient groups. The preoperative photographs were taken from all cases in standard views by one person. Complete blood count (CBC), blood urea nitrogen and creatinine (BUN/Cr), fasting blood sugar (FBS), triglyceride, cholesterol, liver function tests (LFTs) and beta HCG (for women) were checked in all patients after hospitalization. All of these tests were performed and checked again after 3 months.
All surgeries were done by the senior author (AAS) under general anesthesia using an open approach. Some common surgical steps used for all patients included autospreader flap placement, tongue in grove technique and suture technique tip plasty. Bony and cartilaginous hump removal, septoplasty, medial and lateral osteotomy, cephalic trim or creation of hinged flap [2] of cephalic part of lateral crus of lower lateral cartilage and alar base resection were done as needed. Different types of tip grafts were rarely placed. The nasal pack was not used for any patient. All patients got 8 mg dexamethasone intravenously 30 min before surgery and 1 g cefazolin intravenously 6 h after surgery. Cold compress and head elevation were advised for all patients for 48 h after surgery. All patients were discharged from the hospital between 8 and 18 h after surgery. Oral antibiotic (cephalexin 500 mg every 6 h for 5 days), oral analgesic (acetaminophen 325 mg every 4–6 h as needed) and nasal irrigation by normal saline (3–4 times per day for 4 weeks) were prescribed for all patients after their discharge from the hospital. Skin sutures, tapes and external splints were removed 6 days after surgery, and additional taping was not continued thereafter.
Cases were divided into two equal groups based on block randomization. Oral isotretinoin (0.5-mg/kg) was started on the 31st day after surgery and given every other day for 1 month and after that daily for two additional months in the first group. The second group received a placebo in the same form, sequence and interval as the first group. Postoperative photographs were taken 3 months, 6 months and 12 months after surgery. All preoperative and postoperative photographs were taken by one person with equal features and settings.
Finally, preoperative photography was compared with postoperative photography at 3 months, 6 months and 12 months, respectively, by an otorhinolaryngologist who was an expert in facial plastic and cosmetic surgery, but who had not participated in any surgical intervention procedure for this study and was blinded to the patient groups. The cosmetic surgical outcomes were ranked into five grades (excellent, good, fair, no change and poor) by the otorhinolaryngologist.
The patients were also requested to report their cosmetic satisfaction at 3 months, 6 months and 12 months after surgery, respectively. This patient satisfaction was evaluated by questionnaire and ranked into five grades (excellent, good, fair, no change and poor).
The surgeon, photographer, dermatologist and otorhinolaryngologist who evaluated outcomes were blinded to the patient groups.
The sample size was calculated based on a pilot study with 20 cases. According to the pilot study, the excellent satisfaction in the isotretinoin group was 80% and in the placebo group 40%. Therefore, 24 cases were needed in each group to get 80% power at a = 0.05.
Qualitative data were described as frequency and percent, and quantitative data were described as mean ± standard deviation. Data were analyzed using SPSS 18 software. Qualitative variables and quantitative variables were compared and analyzed by the Chi square test and the t test, respectively. Results were statistically considered meaningful at p < 0.05. The proposal of this study was approved by the Tehran University of Medical Sciences Ethics Committee.
Results
Possible confounding variables, such as age, sex, body mass index and previous use of isotretinoin, presented no statistically significant differences between the two groups. The results are summarized in Table 1.
Patient satisfaction in the isotretinoin group at 3 months and 6 months after surgery was significantly better than in the placebo group (p value < 0.05); however, at 12 months after surgery, there was no statistically significant difference between the two groups (p value > 0.05). These results are summarized in Table 2.
As summarized in Table 3, the cosmetic surgical outcome in the isotretinoin group at 3 months and 6 months after surgery based on the ranking by the expert surgeon was significantly better than in the placebo group (p value < 0.05); however, at 12 months after surgery, there was no statistically significant difference between the isotretinoin group and the placebo group (p value > 0.05).
Two patients in the isotretinoin group complained of nasal dryness, and one patient had bloody discharge. These complaints were managed by the use of a topical lubricant, and none of the patients had to stop medications. Additionally, patients’ laboratory findings at 3 months after surgery did not show any significant changes in comparison with preoperative tests requiring cessation of intervention in any participants.
Discussion
The quality and thickness of the nasal skin are some of the most important variables affecting the cosmetic result of rhinoplasty. Patients with thick nasal skin usually have weak lower lateral cartilages. This weakness of the cartilaginous framework and thickness of nasal skin are the main causes that make achievement of desirable tip definition difficult and sometimes impossible. In addition, open septorhinoplasty can exacerbate the frequency and severity of acne after surgery [3, 4].
Isotretinoin is one of the most effective drugs for the treatment of acne especially in its severe forms [5]. Isotretinoin decreases sebum production and colonization of Propionibacterium acnes [6]. Dispenza et al. [7] represented a potential anti-inflammatory mechanism that decreases the inflammatory reaction to the bacteria for up to 6 months after treatment. One of the main concerns about the perioperative use of isotretinoin is its possible interference in skin healing [8]. Plastic surgeons believe oral isotretinoin should be stopped at least 6–12 months before elective surgery to avoid possible wound healing impairment by delayed healing, development of excess granulation tissue or hypertrophic scarring. On the other hand, some studies have demonstrated that isotretinoin does not affect wound healing [9]. Ungarelli et al. [8] reported a review article about the safety of operating on patients taking oral isotretinoin. They concluded: “Effects of isotretinoin in surgical patients require more clinical data, but recent studies are challenging the belief that surgery in patients who have taken or take isotretinoin has unacceptable risks of poor healing. Also, the currently recommended time between discontinuation of isotretinoin and surgery should be reviewed.”
In this study, oral isotretinoin was started 1 month after surgery to eliminate any possible risk of skin healing impairment. Although the standard dosage of oral isotretinoin is 0.5–1 mg/kg per day to reach a cumulative dosage of 120–180 mg/kg (the treatment course lasting for 3–6 months), there are some classical low-dose regimens which include: daily regimen (0.25–0.5 mg/kg per day), alternate day regimen (0.15–0.5 mg/kg every other day) and intermittent dose regimen (0.5–0.75 mg/kg per day, application 1 week or 10 days a month) [10]. We started with 0.5 mg/kg every other day for 1 month to reduce the risk of possible complications and after that continued with 0.5 mg/kg per day for two additional months.
Guyuron and Lee [1] have attempted to present a systemic algorithm to manage rhinoplasty patients with thick nasal skin. Based on their article, one of the most important steps for the management of this group of patients is evaluation and treatment of the skin. They believe that diet alteration, topical Retin-A and ultimately oral isotretinoin should be considered in patients with sebaceous overactivity.
In a retrospective study, Cobo and Vitery [11] reported 17 patients who received oral isotretinoin (dose ranging from 0.25 to 0.5 mg/kg) which was started during the first month after rhinoplasty and was continued for 4–6 months. They reported a significant improvement in the appearance and texture of nasal and facial skin and a better looking tip definition in all patients based on the comparison of photographs before surgery and 2 years after surgery. Although patient follow-up was over 1 year, there was no control group in this study.
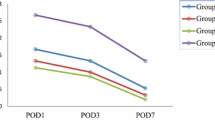
It seems that our study is the first placebo-controlled clinical trial with regard to the role of isotretinoin in thick-skinned rhinoplasty. Our findings show that cosmetic results based on expert surgeon ranking in patients who had received oral isotretinoin were significantly better than the placebo group at 3 months after surgery (Figs. 1, 2). Similarly, at 6 months after surgery, cosmetic results of the isotretinoin group were significantly better than the placebo group. On the other hand, at 12 months after surgery, although the cosmetic results in the isotretinoin group were relatively better than in the placebo group, this difference was not statistically significant. Interestingly, patient satisfaction results were quite similar to the cosmetic results that were ranked by an expert surgeon. Although patient satisfaction in patients who had received oral isotretinoin was significantly better than the placebo group at 3 months and 6 months after surgery, the difference between the two groups was not statistically meaningful at 12 months after surgery (Fig. 3).
Preoperative and postoperative photographs of a female patient who underwent cosmetic rhinoplasty. Preoperative frontal (a), lateral (d) and basal (g) views show droopy thick skin nose. The patient early results are shown in frontal (b), lateral (e) and basal (h) views after 1 month. Oral isotretinoin was started after 1 month. The frontal (c), lateral (f) and basal (i) views of the patient after 1 month of using isotretinoin were shown. Notice the early effect of the isotretinoin in postoperative skin edema
Preoperative and postoperative photographs of a female patient who underwent cosmetic rhinoplasty. Preoperative frontal (a), lateral (d) and basal (g) views show wide thick-skinned nose. The patient’s early results are shown in frontal (b), lateral (e) and basal (h) views after 1 month. Placebo was started after 1 month. The frontal (c), lateral (f) and basal (i) views of the patient after the use of the placebo for 1 month are also shown
Based on our findings, postoperative oral isotretinoin started 1 month after surgery and continued for 3 months at a dose of 0.5 mg/kg/day can accelerate the improvement in cosmetic results and patient satisfaction at least during the first 6 months after surgery (Fig. 1); however, it does not significantly affect the final result 12 months after surgery. It seems that long-term results are affected more by creation of a strong cartilaginous framework using the proper surgical technique. The acceleration in patient satisfaction and cosmetic outcome can be helpful to decrease patient anxiety and make better interaction between surgeon and patient. In addition, the patient might return more rapidly to usual social relations. On the other hand, these advantages should be compared with the disadvantages of possible side effects of oral isotretinoin. Isotretinoin has a variety of potential side effects that include fetal malformations; mucocutaneous effects on the lips, eyes, mouth and other epidermal surfaces; psychiatric disorders; pseudotumor cerebri; decreased night vision; corneal opacities; inflammatory bowel disease; hyperostosis; hepatotoxicity and hypertriglyceridemia [12]. Allen and Rhee [13] reported three patients with nasal tip deformity that presented within 6 months after starting isotretinoin. In the present study, there were no patients with nasal tip deformity at the 11-month follow-up. Therefore, it seems that the surgeon should make the decision to prescribe oral isotretinoin after thick skin rhinoplasty according to the specific features of each patient.
Kosins and Obagi [14] concluded that the Obagi “skin pinch test” is a reliable test for evaluating nasal skin thickness. They classified patients based on pinch thickness of the nasolabial fold. In our study, thick-skinned nose patients were selected based on the review of ancestral and clinical history, visual examination and physical examination of the patient’s skin by an expert dermatologist [15, 16]. In future studies regarding this topic, it could be helpful to determine skin thickness before intervention by means of ultrasonography or computed tomography scan [14, 17] and also evaluate patient satisfaction using other validated questionnaires such as FACE-Q.
Conclusion
Based on our findings, oral isotretinoin usage after rhinoplasty in patients with thick nasal skin significantly accelerates improvement in cosmetic results during the first months after surgery though it does not significantly affect the final cosmetic result 1 year after surgery.
References
Guyuron B, Lee M (2017) An effective algorithm for management of noses with thick skin. Aesth Plast Surg 41:381–387
Sazgar AA, Most SP (2011) Stabilization of nasal tip support in nasal tip reduction surgery. Otolaryngol Head Neck Surg 145:932–934
Nemati S, Golchay J, Iranfar K, Alizadeh A (2013) Frequency of acne vulgaris and its exacerbation in facial and periorbital area after septorhinoplasty. Am J Otolaryngol 34:378–381
Sadeghi M, Saedi B, Safavi A, Iri MR (2012) Postrhinoplasty acne formation: a case–control study. B-ENT 9:95–100
Beckenbach L, Baron JM, Merk HF, Löffler H, Amann PM (2015) Retinoid treatment of skin diseases. Eur J Dermatol 25:384–391
Fox LP, Merk HF, Bickers DR (2006) Dermatopharmakologie. In: Brunton LL, Lazo JS, Parker KL (eds) Goodman & Gilman’s—Pharmakologische Grundlagen der Arzneimitteltherapie. ABW Wissenschaftsverlag GmbH, Berlin, pp 1–73
Dispenza MC, Wolpert EB, Gilliland KL, Dai JP, Cong Z, Nelson AM, Thiboutot DM (2012) Systemic isotretinoin therapy normalizes exaggerated TLR-2-mediated innate immune responses in acne patients. J Invest Dermatol 132:2198–2205
Ungarelli LF, Hetem CM, Farina Junior JA (2016) Is it safe to operate on patients taking isotretinoin? Aesthet Plast Surg 40:139–148
Bagatin E, Guadanhim LR, Yarak S, Kamamoto CS, Almeida FA (2010) Dermabrasion for acne scars during treatment with oral isotretinoin. Dermatol Surg 36:483–489
Sardana K, Garg VK (2011) Low-dose isotretinoin in acne vulgaris: a critical review. Br J Dermatol 165:698–700
Cobo R, Vitery L (2016) Isotretinoin use in thick-skinned rhinoplasty patients. Facial Plast Surg 32:656–661
McLane J (2001) Analysis of common side effects of isotretinoin. J Am Acad Dermatol 45:S188–S194
Allen BC, Rhee JS (2005) Complications associated with isotretinoin use after rhinoplasty. Aesthet Plast Surg 29:102–106
Kosins AM, Obagi ZE (2017) Managing the difficult soft tissue envelope in facial and rhinoplasty surgery. Aesthet Surg J 37(2):143–157
Roberts WE (2008) The Roberts skin type classification system. J Drugs Dermatol 7:452–456
Roberts WE (2009) Skin type classification systems old and new. Dermatol Clin 27:529–533
Cho GS, Kim JH, Yeo NK, Kim SH, Jang YJ (2011) Nasal skin thickness measured using computed tomography and its effect on tip surgery outcomes. Otolaryngol Head Neck Surg 144(4):522–527
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest to disclose.
Rights and permissions
About this article
Cite this article
Sazgar, A.A., Majlesi, A., Shooshtari, S. et al. Oral Isotretinoin in the Treatment of Postoperative Edema in Thick-Skinned Rhinoplasty: A Randomized Placebo-Controlled Clinical Trial. Aesth Plast Surg 43, 189–195 (2019). https://doi.org/10.1007/s00266-018-1252-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-018-1252-5