Abstract
Purpose
The purpose of this study was to determine the effects of naringin on osteoclastogenesis and osteolysis both in vitro and in vivo.
Methods
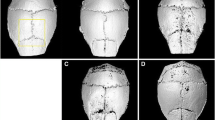
In this research osteoclasts were generated from mouse bone marrow monocytes with the receptor activator of NF-КB ligand and the macrophage colony stimulating factor. Naringin, at a concentration of 1, 10, 50, and 100 μg/mL, was respectively added to the medium. Seven days later, the osteoclasts were determined through tartrate-resistant acid phosphatase (TRAP) staining. Mature osteoclasts were isolated from newborn rabbits and cultured for three days on bone slices. Naringin at a concentration of 1, 10, 50, and 100 μg/mL was respectively added to the medium. The resorption bone slices were quantified, and the area was calculated after toluidine blue and Mayer-hematoxylin staining. Polymethyl methacrylate (PMMA) particles were implanted on the calvariae of C57BL/J6 mice. Naringin, at a dose of 50 μg/kg and 100 μg/kg, was respectively given intraperitoneally for seven. Seven days later, the calvariae were removed and processed for pathological analysis.
Results
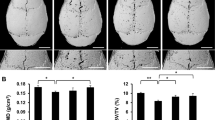
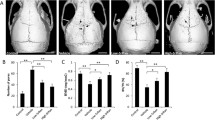
The result indicated that naringin treatment effectively inhibited in vitro osteoclastogenesis and inhibited mature osteoclasts. In vivo data indicated that naringin strongly inhibited PMMA-induced osteolysis.
Conclusion
Naringin can effectively inhibit osteoclastogenesis and suppress wear particles-induced osteolysis and might be useful in the treatment or prevention of wear particles-induced osteolysis and aseptic loosening for its effect on osteoclast generation and function.



Similar content being viewed by others
References
Huo MH, Dumont GD, Knight JR, Mont MA (2011) What’s new in total hip arthroplasty. J Bone Joint Surg Am 93:1944–1950
Billi F, Campbell P (2010) Nanotoxicology of metal wear particles in total joint arthroplasty: a review of current concepts. J Appl Biomater Biomech 8:1–6
Kaddick C, Catelas I, Pennekamp PH, Wimmer MA (2009) Implant wear and aseptic loosening. An overview. Orthopade 38:690–697
Zhang C, Tang T, Ren W, Zhang X, Dai K (2008) Influence of mouse genetic background on wear particle-induced in vivo inflammatory osteolysis. Inflamm Res 57:211–215
Baumann B, Rader CP, Seufert J, Noth U, Rolf O, Eulert J et al (2004) Effects of polyethylene and TiAlV wear particles on expression of RANK, RANKL and OPG mRNA. Acta Orthop Scand 75:295–302
Koreny T, Tunyogi-Csapo M, Gal I, Vermes C, Jacobs JJ, Glant TT (2006) The role of fibroblasts and fibroblast-derived factors in periprosthetic osteolysis. Arthritis Rheum 54:3221–3232
Wooley PH, Morren R, Andary J, Sud S, Yang SY, Mayton L et al (2002) Inflammatory responses to orthopaedic biomaterials in the murine air pouch. Biomaterials 23:517–526
Epstein NJ, Warme BA, Spanogle J, Ma T, Bragg B, Smith RL et al (2005) Interleukin-1 modulates periprosthetic tissue formation in an intramedullary model of particle-induced inflammation. J Orthop Res 23:501–510
Zhou X, Zhang C, Wang X, An B, Zhang P, Zhu Z (2012) Berberine inhibits lipopolysaccharide- and polyethylene particle-induced mouse calvarial osteolysis in vivo. J Surg Res 173:e47–e52
Zhang P, Qin L (2010) Potential use of erythromycin to prevent and treat prosthetic failure. J Clin Pharmacol 50:858–860
Catelas I, Jacobs JJ (2010) Biologic activity of wear particles. Instr Course Lect 59:3–16
Geng DC, Zhu XS, Mao HQ, Meng B, Chen L, Yang HL et al (2011) Protection against titanium particle-induced osteoclastogenesis by cyclooxygenase-2 selective inhibitor. J Biomed Mater Res A 99:516–522
Markel DC, Zhang R, Shi T, Hawkins M, Ren W (2009) Inhibitory effects of erythromycin on wear debris-induced VEGF/Flt-1 gene production and osteolysis. Inflamm Res 58:413–421
Wong RW, Rabie B, Bendeus M, Hagg U (2007) The effects of Rhizoma Curculiginis and Rhizoma Drynariae extracts on bones. Chin Med 2:13
Jeong JC, Lee JW, Yoon CH, Lee YC, Chung KH, Kim MG et al (2005) Stimulative effects of Drynariae Rhizoma extracts on the proliferation and differentiation of osteoblastic MC3T3-E1 cells. J Ethnopharmacol 96:489–495
Jeong JC, Kang SK, Youn CH, Jeong CW, Kim HM, Lee YC et al (2003) Inhibition of Drynariae Rhizoma extracts on bone resorption mediated by processing of cathepsin K in cultured mouse osteoclasts. Int Immunopharmacol 3:1685–1697
Yasuda H, Shima N, Nakagawa N, Yamaguchi K, Kinosaki M, Mochizuki S et al (1998) Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc Natl Acad Sci USA 95:3597–3602
Sobue T, Hakeda Y, Kobayashi Y, Hayakawa H, Yamashita K, Aoki T et al (2001) Tissue inhibitor of metalloproteinases 1 and 2 directly stimulate the bone-resorbing activity of isolated mature osteoclasts. J Bone Miner Res 16:2205–2214
Schwarz EM, Benz EB, Lu AP, Goater JJ, Mollano AV, Rosier RN et al (2000) Quantitative small-animal surrogate to evaluate drug efficacy in preventing wear debris-induced osteolysis. J Orthop Res 18:849–855
Goodman SB, Gomez Barrena E, Takagi M, Konttinen YT (2009) Biocompatibility of total joint replacements: a review. J Biomed Mater Res A 90:603–618
Ren W, Zhang R, Wu B, Wooley PH, Hawkins M, Markel DC (2011) Effects of SU5416 and a vascular endothelial growth factor neutralizing antibody on wear debris-induced inflammatory osteolysis in a mouse model. J Inflamm Res 4:29–38
Zhang W, Zhao H, Peng X, Cheng T, Zhang X (2011) Low-dose captopril inhibits wear debris-induced inflammatory osteolysis. J Int Med Res 39:798–804
Childs LM, Goater JJ, O’Keefe RJ, Schwarz EM (2001) Efficacy of etanercept for wear debris-induced osteolysis. J Bone Miner Res 16:338–347
Schwarz EM, Looney RJ, O’Keefe RJ (2000) Anti-TNF-alpha therapy as a clinical intervention for periprosthetic osteolysis. Arthritis Res 2:165–168
Tsutsumi R, Hock C, Bechtold CD, Proulx ST, Bukata SV, Ito H et al (2008) Differential effects of biologic versus bisphosphonate inhibition of wear debris-induced osteolysis assessed by longitudinal micro-CT. J Orthop Res 26:1340–1346
Zhang P, Dai KR, Yan SG, Yan WQ, Zhang C, Chen DQ et al (2009) Effects of naringin on the proliferation and osteogenic differentiation of human bone mesenchymal stem cell. Eur J Pharmacol 607:1–5
Acknowledgments
This research was supported by the Shanghai Postdoctoral Sustentation Fund, China (Grant No. 11R21414400), China Postdoctoral Science Foundation (Grant No. 20100480598) and National Natural Science Foundation of China (Grant No. 81071487).
Conflicts of interest
The authors declare that they have no conflict of interest with this paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Xiaowei Yu and Xinwei Zhao contributed equally as the first author.
Rights and permissions
About this article
Cite this article
Yu, X., Zhao, X., Wu, T. et al. Inhibiting wear particles-induced osteolysis with naringin. International Orthopaedics (SICOT) 37, 137–143 (2013). https://doi.org/10.1007/s00264-012-1668-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-012-1668-5