Abstract
Purpose
To determine the differential features of inflammatory myofibroblastic tumor (IMT) and intrahepatic cholangiocarcinoma (ICC) manifesting as target appearance on gadoxetic acid-enhanced MRI.
Methods
Twenty-seven patients with 36 IMTs (1.2–6.0 cm) and 34 patients with 34 ICCs (1.5–6.0 cm) who underwent gadoxetic acid-enhanced MRI were enrolled in this study. Two reviewers evaluated morphology, signal intensity, and enhancement features of tumors on T1-weighted imaging (T1WI), T2-weighted imaging (T2WI), diffusion-weighted imaging (DWI), and gadoxetic acid-enhanced imaging.
Results
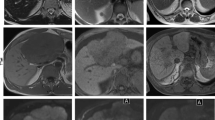
As for 32 IMTs with target appearance, IMTs most commonly demonstrated early target appearance characterized by a peripheral hypointense rim on unenhanced T1WI (n = 27, 84.4%), central enhanced area with a hypointense rim on arterial phase (AP) and portal venous phase (PVP) (n = 29, 90.6%), and transitional phase (TP) (n = 28, 87.5%). Meanwhile, most of the ICCs showed homogeneous hypointensity on T1WI (100%), a hyperenhancing rim on AP, late target appearance on TP (n = 32, 94.1%) and HBP (n = 32, 94.1%). Target appearance on DWI appearing as hyperintensity with central hypointense area was seen in 2 IMTs and 32 ICCs. On T2WI, 24 IMTs (n = 24, 75.0%) displayed central iso- and peripheral hyperintensity and 27 ICCs (84.4%) showed layered hyperintensity with either brighter or darker area in center. The remaining six IMTs with no target were observed as cystic appearing nodules (n = 3) or ill-defined hypovascular nodules (n = 2) and fibrotic mass (n = 1).
Conclusion
IMTs often show early target appearance on unenhanced T1WI, and early dynamic phases of gadoxetic acid-enhanced MRI. Target appearance on later phases, such as TP and HBP, and DWI target appearance were commonly in ICCs, but rare in IMTs.






Similar content being viewed by others
References
Yamamoto H, Yamaguchi H, Aishima S, Oda Y, Kohashi K, Oshiro Y, Tsuneyoshi M (2009) Inflammatory myofibroblastic tumor versus IgG4-related sclerosing disease and inflammatory pseudotumor: a comparative clinicopathologic study. Am J Surg Pathol 33:1330–1340. https://doi.org/10.1097/PAS.0b013e3181a5a207
Pannain VL, Passos JV, Rocha Filho A, Villela-Nogueira C, Caroli-Bottino A (2010) Agressive inflammatory myofibroblastic tumor of the liver with underlying schistosomiasis: a case report. World J Gastroenterol 16:4233–4236. https://doi.org/10.3748/wjg.v16.i33.4233
Al-Jabri T, Sanjay P, Shaikh I, Woodward A (2010) Inflammatory myofibroblastic pseudotumour of the liver in association with gall stones - a rare case report and brief review. Diagn Pathol 5:53. https://doi.org/10.1186/1746-1596-5-53
Papachristou GI, Wu T, Marsh W, Plevy SE (2004) Inflammatory pseudotumor of the liver associated with Crohn’s disease. J Clin Gastroenterol 38:818–822. https://doi.org/10.1016/j.crohns.2009.06.005
Iguchi H, Yamazaki H, Tsunoda H, Takahashi Y, Yokomori H (2013) A Case of Inflammatory Pseudotumor of the Liver Mimicking Hepatocellular Carcinoma on EOB-MRI and PET. Case Rep Med 2013:594254. https://doi.org/10.1155/2013/594254
Bae SK, Abiru S, Kamohara Y, Hashimoto S, Otani M, Saeki A, Nagaoka S, Yamasaki K, Komori A, Ito M, Fujioka H, Yatsuhashi H (2015) Hepatic inflammatory pseudotumor associated with xanthogranulomatous cholangitis mimicking cholangiocarcinoma. Inter Med 54:771–775. https://doi.org/10.2169/internalmedicine.54.2623
Deng FT, Li YX, Ye L, Tong L, Yang XP, Chai XQ (2010) Hilar inflammatory pseudotumor mimicking hilar cholangiocarcinoma. Hepatobiliary Pancreat Dis Int 9:219–221
Xiao Y, Zhou S, Ma C, Luo J, Zhu H, Tang F (2013) Radiological and histopathological features of hepatic inflammatory myofibroblastic tumour: analysis of 10 cases. Clin Radiol 68:1114–1120. https://doi.org/10.1016/j.crad.2013.05.097
Park HJ, Kim YK, Park MJ, Lee WJ (2013) Small intrahepatic mass-forming cholangiocarcinoma: target sign on diffusion-weighted imaging for differentiation from hepatocellular carcinoma. Abdom Imaging 38:793–801. https://doi.org/10.1007/s00261-012-9943-x
Elsayes KM, Hooker JC, Agrons MM, Kielar AZ, Tang A, Fowler KJ, Chernyak V, Bashir MR, Kono Y, Do RK, Mitchell DG, Kamaya A, Hecht EM, Sirlin CB (2017) 2017 Version of LI-RADS for CT and MR Imaging: An Update. Radiographics 37:1994–2017. https://doi.org/10.1148/rg.2017170098
Karahan OI, Isin S, Baykara M, Coskun A (2003) Case report: Inflammatory pseudotumor of the liver with target-like appearance. Tani Girisim Radyol 9:75–77
Min JH, Kim YK, Choi SY, Jeong WK, Lee WJ, Ha SY, Ahn S, Ahn HS (2017) Differentiation between cholangiocarcinoma and hepatocellular carcinoma with target sign on diffusion-weighted imaging and hepatobiliary phase gadoxetic acid-enhanced MR imaging: Classification tree analysis applying capsule and septum. Eur J Radiol 92:1–10. https://doi.org/10.1016/j.ejrad.2017.04.008
Yu JS, Park C, Kim JH, Chung JJ, Kim KW (2007) Inflammatory myofibrobalstic tumors in the liver: MRI of two immunohistochemically-verified cases. J Mag Reson Imaging 26:418–421
Zhao JJ, Ling JQ, Fang Y, Gao XD, Shu P, Shen KT, Qin J, Sun YH, Qin XY (2014) Intra-abdominal inflammatory myofibroblastic tumor: spontaneous regression. World J Gastroenterol 20:13625–13631. https://doi.org/10.3748/wjg.v20.i37.13625
Eun HW, Kim JH, Hong SS, Kim YJ (2012) Malignant versus benign hepatic masses in patients with recurrent pyogenic cholangitis: MR differential diagnosis. Abdom Imaging 37:767–774. https://doi.org/10.1007/s00261-011-9833-7
Matsubara O, Tan-Liu NS, Kenney RM, Mark EJ (1988) Inflammatory pseudotumors of the lung: progression from organizing pneumonia to fibrous histiocytoma or to plasma cell granuloma in 32 cases. Hum Pathol 19:807–814. https://doi.org/10.1016/S0046-8177(88)80264-8
Kim YK, Kim CS, Lee JM, Ko SW, Moon WS, Yu HC (2006) Solid organizing hepatic abscesses mimic hepatic tumor: Multiphasic computed tomography and magnetic resonance imaging findings with histopathologic correlation. J Comput Assist Tomogr 30:189–196
Kang TW, Kim SH, Jang KM, Choi D, Choi JY, Park CK (2014) Inflammatory myofibroblastic tumours of the liver: gadoxetic acid-enhanced and diffusion-weighted MRI findings with 18F-FDG PET/CT and clinical significance of regression on follow-up. Clin Radiol 69:509–518. https://doi.org/10.1016/j.crad.2013.12.018
Kim YK, Lee YH, Kim CS, Lee MW (2011) Differentiating focal eosinophilic liver disease from hepatic metastases using unenhanced and gadoxetic acid-enhanced MRI. Abdom Imaging 36:425–432. https://doi.org/10.1007/s00261-011-9752-7
Hiwatashi A, Kinoshita T, Moritani T, Wang HZ, Shrier DA, Numaguchi Y, Ekholm SE, Westesson PL (2003) Hypointensity on diffusion-weighted MRI of the brain related to T2 shortening and susceptibility effects. AJR Am J Roentgenol 181:1705–1709. https://doi.org/10.2214/ajr.181.6.1811705
Maetani Y, Itoh K, Watanabe C, Shibata T, Ametani F, Yamabe H, Konishi J (2001) MR imaging of intrahepatic cholangiocarcinoma with pathologic correlation. AJR Am J Roentgenol 176:1499–1507. https://doi.org/10.2214/ajr.176.6.1761499
Chong YS, Kim YK, Lee MW, Kim SH, Lee WJ, Rhim HC, Lee SJ (2012) Differentiating mass-forming intrahepatic cholangiocarcinoma from atypical hepatocellular carcinoma using gadoxetic acid-enhanced MRI. Clin Radiol 67:766–773. https://doi.org/10.1016/j.crad.2012.01.004
Kim SH, Lee CH, Kim BH, Kim WB, Yeom SK, Kim KA, Park CM (2012) Typical and atypical imaging findings of intrahepatic cholangiocarcinoma using gadolinium ethoxybenzyl diethylenetriamine pentaacetic acid-enhanced magnetic resonance imaging. J Comput Assist Tomogr 36:704–709. https://doi.org/10.1097/RCT.0b013e3182706562
Gabata T, Matsui O, Kadoya M, Yoshikawa J, Ueda K, Kawamori Y, Takashima T, Nonomura A (1998) Delayed MR imaging of the liver: correlation of delayed enhancement of hepatic tumors and pathologic appearance. Abdom Imaging 23:309–313. https://doi.org/10.1007/s002619900347
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was waived by IRB.
Rights and permissions
About this article
Cite this article
Chang, A.I., Kim, Y.K., Min, J.H. et al. Differentiation between inflammatory myofibroblastic tumor and cholangiocarcinoma manifesting as target appearance on gadoxetic acid-enhanced MRI. Abdom Radiol 44, 1395–1406 (2019). https://doi.org/10.1007/s00261-018-1847-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-018-1847-y