Abstract
Purpose
Nodal involvement is an independent risk factor of recurrence in papillary thyroid cancer (PTC). Neither the international guidelines nor the recently introduced ongoing risk adaptation concept consider the extent of initial surgical clearance of radioiodine sensitive lymph node metastases in their stratification systems. We investigated the prognostic relevance of incomplete initial surgical clearance in patients with purely lymphogeneous metastatic PTC (pN1 M0) despite successful radioiodine therapy. Accurate assessment of pre-ablative nodal status was attempted using PET/CT studies with both 124I-NaI and 18F-FDG along with high-resolution cervical ultrasound.
Methods
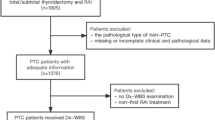
Sixty-five patients with histologically diagnosed lymph node metastases (pN1 M0) were retrospectively analyzed. Patients with iodine-negative lymph node metastases diagnosed by 18F-FDG PET/CT or distant metastases were excluded from the analysis. The association of disease recurrence with the pre-ablative nodal status, as well as other baseline characteristics, were examined applying nonparametric tests for independent samples and multiple regression analysis. Patients with persistent lymph node metastases in 124I-NaI PET/CT were further divided according to the additional presence or absence of FDG-uptake in 18F-FDG PET/CT. Survival analyses were performed using Kaplan–Meier curves and the Cox proportional hazards model for uni- and multivariate analyses to assess the influence of prognostic factors on progression free survival (PFS).
Results
Incomplete metastatic lymph node resection captured by 124I-NaI PET/CT (n = 33) was an independent risk factor for recurrence (61 % vs 25 %, p = 0.006) and shorter PFS (46 months vs not reached, HR 4.0 [95 %-CI, 1.7–9.2], p = 0.001). Ultrasound could detect lymph node metastases only in 19/33 patients (58 %). Among patients with positive nodal status, FDG-avidity of metastatic iodine positive lymph nodes worsened the outcome (16 vs 69 months, p = 0.047). From all other investigated factors including age, N-stage (N1a vs N1b), and T-Stage (T4 vs T1-3), only large tumor size (pT4) had a significant impact on PFS (HR 2.9 [95 %-CI, 1.3–6.4], p = 0.007).
Conclusions
Incomplete initial surgical clearance of lymph node metastases even after successful radioiodine therapy may increase the chances of recurrence and is an independent risk factor for impaired survival of patients with PTC. Pre-ablative (dual tracer PET/CT) imaging with 124I-Na and 18F provides a prognostic tool for these patients and may considerably complement the current risk stratification systems.




Similar content being viewed by others
References
Schlumberger MJ. Papillary and follicular thyroid carcinoma. N Engl J Med. 1998;338:297–306. doi:10.1056/NEJM199801293380506.
Sherman SI. Thyroid carcinoma. Lancet. 2003;361:501–11.
Dean DS, Hay ID. Prognostic indicators in differentiated thyroid carcinoma. Cancer Control. 2000;7:229–39.
Yang L, Shen W, Sakamoto N. Population-based study evaluating and predicting the probability of death resulting from thyroid cancer and other causes among patients with thyroid cancer. J Clin Oncol. 2013;31:468–74. doi:10.1200/JCO.2012.42.4457.
Coburn MC, Wanebo HJ. Prognostic factors and management considerations in patients with cervical metastases of thyroid cancer. Am J Surg. 1992;164:671–6.
Lundgren CI, Hall P, Dickman PW, Zedenius J. Clinically significant prognostic factors for differentiated thyroid carcinoma: a population-based, nested case–control study. Cancer. 2006;106:524–31. doi:10.1002/cncr.21653.
Guo K, Wang Z. Risk factors influencing the recurrence of papillary thyroid carcinoma: a systematic review and meta-analysis. Int J Clin Exp Pathol. 2014;7:5393–403.
Dralle H, Musholt TJ, Schabram J, Steinmuller T, Frilling A, Simon D, et al. German association of endocrine surgeons practice guideline for the surgical management of malignant thyroid tumors. Langenbecks Arch Surg. 2013;398:347–75. doi:10.1007/s00423-013-1057-6.
Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, et al. 2015 american thyroid association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the american thyroid association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid. 2016;26:1–133. doi:10.1089/thy.2015.0020.
Cooper DS, Doherty GM, Haugen BR, Kloos RT, Lee SL, Mandel SJ, et al. Revised American thyroid association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid. 2009;19:1167–214. doi:10.1089/thy.2009.0110.
Pacini F, Castagna MG, Brilli L, Pentheroudakis G, Group EGW. Differentiated thyroid cancer: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol. 2009;20 Suppl 4:143–6. doi:10.1093/annonc/mdp156.
Bockisch A, Rosenbaum-Krumme S. Cancer: the effect of radioiodine therapy after total thyroidectomy. Nat Rev Endocrinol. 2013;9:511–2. doi:10.1038/nrendo.2013.153.
Sabet A, Kim M. Postoperative management of differentiated thyroid cancer. Otolaryngol Clin North Am. 2010;43:329–51. doi:10.1016/j.otc.2010.02.003. viii-ix.
Tuttle RM, Tala H, Shah J, Leboeuf R, Ghossein R, Gonen M, et al. Estimating risk of recurrence in differentiated thyroid cancer after total thyroidectomy and radioactive iodine remnant ablation: using response to therapy variables to modify the initial risk estimates predicted by the new American thyroid association staging system. Thyroid: Off J Am Thyroid Assoc. 2010;20:1341–9. doi:10.1089/thy.2010.0178.
Castagna MG, Maino F, Cipri C, Belardini V, Theodoropoulou A, Cevenini G, et al. Delayed risk stratification, to include the response to initial treatment (surgery and radioiodine ablation), has better outcome predictivity in differentiated thyroid cancer patients. Eur J Endocrinol. 2011;165:441–6. doi:10.1530/EJE-11-0466.
Cano-Palomares A, Castells I, Capel I, Bella MR, Barcons S, Serrano A, et al. Response to initial therapy of differentiated thyroid cancer predicts the long-term outcome better than classical risk stratification systems. Int J Endocrinol. 2014;2014:591285. doi:10.1155/2014/591285.
de Pont C, Halders S, Bucerius J, Mottaghy F, Brans B. 124I PET/CT in the pretherapeutic staging of differentiated thyroid carcinoma: comparison with posttherapy 131I SPECT/CT. Eur J Nucl Med Mol Imaging. 2013;40:693–700. doi:10.1007/s00259-012-2331-5.
Jentzen W, Moldovan AS, Ruhlmann M, Gorges R, Bockisch A, Rosenbaum-Krumme S. Lowest effective 131I activity for thyroid remnant ablation of differentiated thyroid cancer patients. Dosimetry-based model for estimation. Nuklearmedizin. 2015;54:137–43. doi:10.3413/Nukmed-0711-14-12.
Jentzen W, Freudenberg L, Eising EG, Sonnenschein W, Knust J, Bockisch A. Optimized 124I PET dosimetry protocol for radioiodine therapy of differentiated thyroid cancer. J Nucl Med. 2008;49:1017–23. doi:10.2967/jnumed.107.047159.
Jentzen W, Hoppenbrouwers J, van Leeuwen P, van der Velden D, van de Kolk R, Poeppel TD, et al. Assessment of lesion response in the initial radioiodine treatment of differentiated thyroid cancer using 124I PET imaging. J Nucl Med. 2014;55:1759–65. doi:10.2967/jnumed.114.144089.
Leenhardt L, Erdogan MF, Hegedus L, Mandel SJ, Paschke R, Rago T, et al. 2013 European thyroid association guidelines for cervical ultrasound scan and ultrasound-guided techniques in the postoperative management of patients with thyroid cancer. Eur Thyroid J. 2013;2:147–59. doi:10.1159/000354537.
Adam MA, Pura J, Goffredo P, Dinan MA, Reed SD, Scheri RP, et al. Presence and number of lymph node metastases Are associated with compromised survival for patients younger than Age 45 years with papillary thyroid cancer. J Clin Oncol. 2015;33:2370–5. doi:10.1200/JCO.2014.59.8391.
Ryu IS, Song CI, Choi SH, Roh JL, Nam SY, Kim SY. Lymph node ratio of the central compartment is a significant predictor for locoregional recurrence after prophylactic central neck dissection in patients with thyroid papillary carcinoma. Ann Surg Oncol. 2014;21:277–83. doi:10.1245/s10434-013-3258-1.
Jeon MJ, Yoon JH, Han JM, Yim JH, Hong SJ, Song DE, et al. The prognostic value of the metastatic lymph node ratio and maximal metastatic tumor size in pathological N1a papillary thyroid carcinoma. Eur J Endocrinol. 2013;168:219–25. doi:10.1530/EJE-12-0744.
Young S, Harari A, Smooke-Praw S, Ituarte PH, Yeh MW. Effect of reoperation on outcomes in papillary thyroid cancer. Surgery. 2013;154:1354–61. doi:10.1016/j.surg.2013.06.043. discussion 61–2.
Tufano RP, Bishop J, Wu G. Reoperative central compartment dissection for patients with recurrent/persistent papillary thyroid cancer: efficacy, safety, and the association of the BRAF mutation. Laryngoscope. 2012;122:1634–40. doi:10.1002/lary.23371.
Al-Saif O, Farrar WB, Bloomston M, Porter K, Ringel MD, Kloos RT. Long-term efficacy of lymph node reoperation for persistent papillary thyroid cancer. J Clin Endocrinol Metab. 2010;95:2187–94. doi:10.1210/jc.2010-0063.
Jonklaas J, Sarlis NJ, Litofsky D, Ain KB, Bigos ST, Brierley JD, et al. Outcomes of patients with differentiated thyroid carcinoma following initial therapy. Thyroid: Off J Am Thyroid Assoc. 2006;16:1229–42. doi:10.1089/thy.2006.16.1229.
Pacini F, Schlumberger M, Harmer C, Berg GG, Cohen O, Duntas L, et al. Post-surgical use of radioiodine (131I) in patients with papillary and follicular thyroid cancer and the issue of remnant ablation: a consensus report. Eur J Endocrinol. 2005;153:651–9. doi:10.1530/eje.1.02014.
Pelizzo MR, Boschin IM, Toniato A, Piotto A, Pagetta C, Gross MD, et al. Papillary thyroid carcinoma: 35-year outcome and prognostic factors in 1858 patients. Clin Nucl Med. 2007;32:440–4. doi:10.1097/RLU.0b013e31805375ca.
Pacini F, Schlumberger M, Dralle H, Elisei R, Smit JW, Wiersinga W. European consensus for the management of patients with differentiated thyroid carcinoma of the follicular epithelium. Eur J Endocrinol. 2006;154:787–803. doi:10.1530/eje.1.02158.
Jeon MJ, Kim WG, Park WR, Han JM, Kim TY, Song DE, et al. Modified dynamic risk stratification for predicting recurrence using the response to initial therapy in patients with differentiated thyroid carcinoma. Eur J Endocrinol. 2014;170:23–30. doi:10.1530/EJE-13-0524.
Lepoutre-Lussey C, Maddah D, Golmard JL, Russ G, Tissier F, Tresallet C, et al. Post-operative neck ultrasound and risk stratification in differentiated thyroid cancer patients with initial lymph node involvement. Eur J Endocrinol. 2014;170:837–46. doi:10.1530/EJE-13-0888.
Freudenberg LS, Jentzen W, Stahl A, Bockisch A, Rosenbaum-Krumme SJ. Clinical applications of 124I-PET/CT in patients with differentiated thyroid cancer. Eur J Nucl Med Mol Imaging. 2011;38 Suppl 1:S48–56. doi:10.1007/s00259-011-1773-5.
Van Nostrand D, Moreau S, Bandaru VV, Atkins F, Chennupati S, Mete M, et al. (124)I positron emission tomography versus (131)I planar imaging in the identification of residual thyroid tissue and/or metastasis in patients who have well-differentiated thyroid cancer. Thyroid. 2010;20:879–83. doi:10.1089/thy.2009.0430.
Phan HT, Jager PL, Paans AM, Plukker JT, Sturkenboom MG, Sluiter WJ, et al. The diagnostic value of 124I-PET in patients with differentiated thyroid cancer. Eur J Nucl Med Mol Imaging. 2008;35:958–65. doi:10.1007/s00259-007-0660-6.
Haslerud T, Brauckhoff K, Reisaeter L, Kufner-Lein R, Heinecke A, Varhaug JE, et al. F18-FDG-PET for recurrent differentiated thyroid cancer: a systematic meta-analysis. Acta Radiol. 2015. doi:10.1177/0284185115594645.
Wang W, Larson SM, Fazzari M, Tickoo SK, Kolbert K, Sgouros G, et al. Prognostic value of [18F]fluorodeoxyglucose positron emission tomographic scanning in patients with thyroid cancer. J Clin Endocrinol Metab. 2000;85:1107–13. doi:10.1210/jcem.85.3.6458.
Grunwald F, Kalicke T, Feine U, Lietzenmayer R, Scheidhauer K, Dietlein M, et al. Fluorine-18 fluorodeoxyglucose positron emission tomography in thyroid cancer: results of a multicentre study. Eur J Nucl Med. 1999;26:1547–52.
Rosenbaum-Krumme SJ, Gorges R, Bockisch A, Binse I. (1)(8)F-FDG PET/CT changes therapy management in high-risk DTC after first radioiodine therapy. Eur J Nucl Med Mol Imaging. 2012;39:1373–80. doi:10.1007/s00259-012-2065-4.
Iagaru A, Kalinyak JE, McDougall IR. F-18 FDG PET/CT in the management of thyroid cancer. Clin Nucl Med. 2007;32:690–5. doi:10.1097/RLU.0b013e318125037a.
Caetano R, Bastos CR, de Oliveira IA, da Silva RM, Fortes CP, Pepe VL, et al. Accuracy of positron emission tomography and positron emission tomography-CT in the detection of differentiated thyroid cancer recurrence with negative I whole-body scan results: a meta-analysis. Head Neck. 2014. doi:10.1002/hed.23881.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Research involving Human Participants and/or Animals
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the principles of the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
This article does not contain any studies with animals performed by any of the authors.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Amir Sabet and Ina Binse contributed equally to this work.
Rights and permissions
About this article
Cite this article
Sabet, A., Binse, I., Grafe, H. et al. Prognostic impact of incomplete surgical clearance of radioiodine sensitive local lymph node metastases diagnosed by post-operative 124I-NaI-PET/CT in patients with papillary thyroid cancer. Eur J Nucl Med Mol Imaging 43, 1988–1994 (2016). https://doi.org/10.1007/s00259-016-3400-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-016-3400-y