Abstract
Purpose
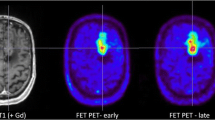
Current guidelines for glioma imaging by positron emission tomography (PET) using the amino acid analogue O-(2-[18F]fluoroethyl)-L-tyrosine (18F-FET) recommend image acquisition from 20–40 min post injection (p.i.). The maximal tumour-to-background evaluation (TBRmax) obtained in these summation images does not enable reliable differentiation between low and high grade glioma (LGG and HGG), which, however, can be achieved by dynamic 18F-FET-PET. We investigated the accuracy of tumour grading using TBRmax values at different earlier time points after tracer injection.
Methods
Three hundred and fourteen patients with histologically proven primary diagnosis of glioma (131 LGG, 183 HGG) who had undergone 40-min dynamic 18F-FET-PET scans were retrospectively evaluated. TBRmax was assessed in the standard 20–40 min summation images, as well as in summation images from 0–10 min, 5–15 min, 5–20 min, and 15–30 min p.i., and kinetic analysis was performed. TBRmax values and kinetic analysis were correlated with histological classification. ROC analyses were performed for each time frame and sensitivity, specificity, and accuracy were assessed.
Results
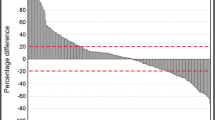
TBRmax values in the earlier summation images were significantly better for tumour grading (P < 0.001) when compared to standard 20–40 min scans, with best results for the early 5–15 min scan. This was due to higher TBRmax in the HGG (3.9 vs. 3.3; p < 0.001), while TBRmax remained nearly stable in the LGG (2.2 vs. 2.1). Overall, accuracy increased from 70 % in the 20–40 min analysis to 77 % in the 5–15 min images, but did not reach the accuracy of dynamic analysis (80 %).
Conclusions
Early TBRmax assessment (5–15 min p.i.) is more accurate for the differentiation between LGG and HGG than the standard static scan (20–40 min p.i.) mainly caused by the characteristic high 18F-FET uptake of HGG in the initial phase. Therefore, when dynamic 18F-FET-PET cannot be performed, early TBRmax assessment can be considered as an alternative for tumour grading.





Similar content being viewed by others
References
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, et al. The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol. 2007;114:97–109. doi:10.1007/s00401-007-0243-4.
Jansen NL, Graute V, Armbruster L, Suchorska B, Lutz J, Eigenbrod S, et al. MRI-suspected low-grade glioma: is there a need to perform dynamic FET PET? Eur J Nucl Med Mol Imaging. 2012;39:1021–9. doi:10.1007/s00259-012-2109-9.
Scott JN, Brasher PM, Sevick RJ, Rewcastle NB, Forsyth PA. How often are nonenhancing supratentorial gliomas malignant? A population study. Neurology. 2002;59:947–9.
Kondziolka D, Lunsford LD, Martinez AJ. Unreliability of contemporary neurodiagnostic imaging in evaluating suspected adult supratentorial (low-grade) astrocytoma. J Neurosurg. 1993;79:533–6. doi:10.3171/jns.1993.79.4.0533.
Bisdas S, Ritz R, Bender B, Braun C, Pfannenberg C, Reimold M, et al. Metabolic mapping of gliomas using hybrid MR-PET imaging: feasibility of the method and spatial distribution of metabolic changes. Investig Radiol. 2013;48:295–301. doi:10.1097/RLI.0b013e31827188d6.
Pauleit D, Floeth F, Hamacher K, Riemenschneider MJ, Reifenberger G, Muller HW, et al. O-(2-[18F]fluoroethyl)-L-tyrosine PET combined with MRI improves the diagnostic assessment of cerebral gliomas. Brain. 2005;128:678–87. doi:10.1093/brain/awh399.
la Fougere C, Suchorska B, Bartenstein P, Kreth FW, Tonn JC. Molecular imaging of gliomas with PET: opportunities and limitations. Neuro Oncol. 2011;13:806–19. doi:10.1093/neuonc/nor054.
Pafundi DH, Laack NN, Youland RS, Parney IF, Lowe VJ, Giannini C, et al. Biopsy validation of 18F-DOPA PET and biodistribution in gliomas for neurosurgical planning and radiotherapy target delineation: results of a prospective pilot study. Neuro Oncol. 2013;15:1058–67. doi:10.1093/neuonc/not002.
Vees H, Senthamizhchelvan S, Miralbell R, Weber DC, Ratib O, Zaidi H. Assessment of various strategies for 18F-FET PET-guided delineation of target volumes in high-grade glioma patients. Eur J Nucl Med Mol Imaging. 2009;36:182–93. doi:10.1007/s00259-008-0943-6.
Kracht LW, Miletic H, Busch S, Jacobs AH, Voges J, Hoevels M, et al. Delineation of brain tumor extent with [11C]L-methionine positron emission tomography: local comparison with stereotactic histopathology. Clin Cancer Res. 2004;10:7163–70. doi:10.1158/1078-0432.CCR-04-0262.
Jansen NL, Suchorska B, Schwarz SB, Eigenbrod S, Lutz J, Graute V, et al. [18F]fluoroethyltyrosine-positron emission tomography-based therapy monitoring after stereotactic iodine-125 brachytherapy in patients with recurrent high-grade glioma. Mol Imaging. 2013;12:137–47.
Galldiks N, Stoffels G, Filss C, Rapp M, Blau T, Tscherpel C, et al. The use of dynamic O-(2-18F-fluoroethyl)-l-tyrosine PET in the diagnosis of patients with progressive and recurrent glioma. Neuro Oncol. 2015. doi:10.1093/neuonc/nov088.
Galldiks N, Dunkl V, Stoffels G, Hutterer M, Rapp M, Sabel M, et al. Diagnosis of pseudoprogression in patients with glioblastoma using O-(2-[18F]fluoroethyl)-L-tyrosine PET. Eur J Nucl Med Mol Imaging. 2015;42:685–95. doi:10.1007/s00259-014-2959-4.
Herrmann K, Czernin J, Cloughesy T, Lai A, Pomykala KL, Benz MR, et al. Comparison of visual and semiquantitative analysis of 18F-FDOPA-PET/CT for recurrence detection in glioblastoma patients. Neuro Oncol. 2014;16:603–9. doi:10.1093/neuonc/not166.
Glaudemans AW, Enting RH, Heesters MA, Dierckx RA, van Rheenen RW, Walenkamp AM, et al. Value of 11C-methionine PET in imaging brain tumours and metastases. Eur J Nucl Med Mol Imaging. 2013;40:615–35. doi:10.1007/s00259-012-2295-5.
Popperl G, Kreth FW, Mehrkens JH, Herms J, Seelos K, Koch W, et al. FET PET for the evaluation of untreated gliomas: correlation of FET uptake and uptake kinetics with tumour grading. Eur J Nucl Med Mol Imaging. 2007;34:1933–42. doi:10.1007/s00259-007-0534-y.
Langen KJ, Bartenstein P, Boecker H, Brust P, Coenen HH, Drzezga A, et al. German guidelines for brain tumour imaging by PET and SPECT using labelled amino acids. Nuklearmedizin. 2011;50:167–73.
Vander Borght T, Asenbaum S, Bartenstein P, Halldin C, Kapucu Ö, Van Laere K, et al. EANM Procedure Guidelines for Brain Tumour Imaging using Labelled Amino Acid Analogues. Eur J Nucl Med Mol Imaging. 2006;33:1374–80.
Popperl G, Kreth FW, Herms J, Koch W, Mehrkens JH, Gildehaus FJ, et al. Analysis of 18F-FET PET for grading of recurrent gliomas: is evaluation of uptake kinetics superior to standard methods? J Nucl Med. 2006;47:393–403.
Jansen NL, Suchorska B, Wenter V, Eigenbrod S, Schmid-Tannwald C, Zwergal A, et al. Dynamic 18F-FET PET in newly diagnosed astrocytic low-grade glioma identifies high-risk patients. J Nucl Med. 2014;55:198–203. doi:10.2967/jnumed.113.122333.
Kunz M, Thon N, Eigenbrod S, Hartmann C, Egensperger R, Herms J, et al. Hot spots in dynamic (18)FET-PET delineate malignant tumor parts within suspected WHO grade II gliomas. Neuro Oncol. 2011;13:307–16. doi:10.1093/neuonc/noq196.
Wen PY, Norden AD, Drappatz J, Quant E. Response assessment challenges in clinical trials of gliomas. Current oncology reports. 2010;12:68–75. doi:10.1007/s11912-009-0078-3.
Galldiks N, Stoffels G, Ruge MI, Rapp M, Sabel M, Reifenberger G, et al. Role of O-(2-18F-fluoroethyl)-L-tyrosine PET as a diagnostic tool for detection of malignant progression in patients with low-grade glioma. J Nucl Med. 2013;54:2046–54. doi:10.2967/jnumed.113.123836.
Dunkl V, Cleff C, Stoffels G, Judov N, Sarikaya-Seiwert S, Law I, et al. The usefulness of dynamic O-(2-18F-fluoroethyl)-L-tyrosine PET in the clinical evaluation of brain tumors in children and adolescents. J Nucl Med. 2015;56:88–92. doi:10.2967/jnumed.114.148734.
Calcagni ML, Galli G, Giordano A, Taralli S, Anile C, Niesen A, et al. Dynamic O-(2-[18F]fluoroethyl)-L-tyrosine (F-18 FET) PET for glioma grading: assessment of individual probability of malignancy. Clin Nucl Med. 2011;36:841–7. doi:10.1097/RLU.0b013e3182291b40.
Jansen NL, Suchorska B, Wenter V, Schmid-Tannwald C, Todica A, Eigenbrod S, et al. Prognostic significance of dynamic 18F-FET PET in newly diagnosed astrocytic high-grade glioma. J Nucl Med. 2015;56:9–15. doi:10.2967/jnumed.114.144675.
Derlon JM, Chapon F, Noel MH, Khouri S, Benali K, Petit-Taboue MC, et al. Non-invasive grading of oligodendrogliomas: correlation between in vivo metabolic pattern and histopathology. Eur J Nucl Med. 2000;27:778–87.
Kracht LW, Friese M, Herholz K, Schroeder R, Bauer B, Jacobs A, et al. Methyl-[11C]- l-methionine uptake as measured by positron emission tomography correlates to microvessel density in patients with glioma. Eur J Nucl Med Mol Imaging. 2003;30:868–73. doi:10.1007/s00259-003-1148-7.
Bisdas S, Kirkpatrick M, Giglio P, Welsh C, Spampinato MV, Rumboldt Z. Cerebral blood volume measurements by perfusion-weighted MR imaging in gliomas: ready for prime time in predicting short-term outcome and recurrent disease? AJNR Am J Neurorad. 2009;30:681–8. doi:10.3174/ajnr.A1465.
Jansen NL, Schwartz C, Graute V, Eigenbrod S, Lutz J, Egensperger R, et al. Prediction of oligodendroglial histology and LOH 1p/19q using dynamic [(18)F]FET-PET imaging in intracranial WHO grade II and III gliomas. Neuro Oncol. 2012;14:1473–80. doi:10.1093/neuonc/nos259.
Lohmann P, Herzog H, Rota Kops E, Stoffels G, Judov N, Filss C, et al. Dual-time-point O-(2-[F]fluoroethyl)-L-tyrosine PET for grading of cerebral gliomas. Eur Radiol. 2015. doi:10.1007/s00330-015-3691-6.
Acknowledgment
Parts of this paper originate from the doctoral thesis of Isabel Winkelmann. We thank Dr. Markus Diemling for the technical support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Albert, N.L., Winkelmann, I., Suchorska, B. et al. Early static 18F-FET-PET scans have a higher accuracy for glioma grading than the standard 20–40 min scans. Eur J Nucl Med Mol Imaging 43, 1105–1114 (2016). https://doi.org/10.1007/s00259-015-3276-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-015-3276-2