Abstract
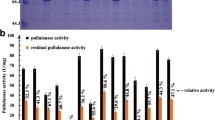
Pullulanases are widely used in food, medicine, and other industries because they specifically hydrolyze α-1,6-glycosidic linkages in starch and oligosaccharides. In addition, high-temperature thermostable pullulanase has multiple advantages, including decreasing saccharification solution viscosity accompanied with enhanced mass transfer and reducing microbial contamination in starch hydrolysis. However, thermophilic pullulanase availability remains limited. Additionally, most do not meet starch-manufacturing requirements due to weak thermostability. Here, we developed a computation-aided strategy to engineer the thermophilic pullulanase from Bacillus thermoleovorans. First, three computational design predictors (FoldX, I-Mutant 3.0, and dDFIRE) were combined to predict stability changes introduced by mutations. After excluding conserved and catalytic sites, 17 mutants were identified. After further experimental verification, we confirmed six positive mutants. Among them, the G692M mutant had the highest thermostability improvement, with 3.8 °C increased Tm and 2.1-fold longer half-life than the wild type at 70 °C. We then characterized the mechanism underlying increased thermostability, such as rigidity enhancement, closer conformation, and strengthened motion correlation using root mean square fluctuation (RMSF), principal component analysis (PCA), dynamic cross-correlation map (DCCM), and free energy landscape (FEL) analysis.
Key points
• A computation-aided strategy was developed to engineer pullulanase thermostability.
• Seventeen mutants were identified by combining three computational design predictors.
• The G692M mutant was obtained with increased Tmand half-life at 70 °C.







Similar content being viewed by others
References
Arnold FH (2019) Innovation by evolution: bringing new chemistry to life (Nobel Lecture). Angew Chem Int Ed Eng 58(41):14420–14426. https://doi.org/10.1002/anie.201907729
Bednar D, Beerens K, Sebestova E, Bendl J, Khare S, Chaloupkova R, Prokop Z, Brezovsky J, Baker D, Damborsky J (2015) FireProt: energy- and evolution-based computational design of thermostable multiple-point mutants. PLoS Comput Biol 11(11):e1004556–e1004556. https://doi.org/10.1371/journal.pcbi.1004556
Ben Messaoud E, Ben Ammar Y, Mellouli L, Bejar S (2002) Thermostable pullulanase type I from new isolated Bacillus thermoleovorans US105: cloning, sequencing and expression of the gene in E. coli. Enzym Microb Technol 31(6):827–832. https://doi.org/10.1016/S0141-0229(02)00185-0
Bommarius AS, Paye MF (2013) Stabilizing biocatalysts. Chem Soc Rev 42(15):6534–6565. https://doi.org/10.1039/c3cs60137d
Bornscheuer UT, Huisman GW, Kazlauskas RJ, Lutz S, Moore JC, Robins K (2012) Engineering the third wave of biocatalysis. Nature 485(7397):185–194. https://doi.org/10.1038/nature11117
Capriotti E, Fariselli P, Casadio R (2005) I-Mutant2.0: predicting stability changes upon mutation from the protein sequence or structure. Nucleic Acids Res 33(Web Server issue):W306–W310. https://doi.org/10.1093/nar/gki375
Carugo O, Pongor S (2001) A normalized root-mean-square distance for comparing protein three-dimensional structures. Protein Sci 10(7):1470–1473. https://doi.org/10.1110/ps.690101
Chakravarty S, Varadarajan R (2002) Elucidation of factors responsible for enhanced thermal stability of proteins: a structural genomics based study. Biochemistry 41(25):8152–8161. https://doi.org/10.1021/bi025523t
Chen VB, Arendall WB 3rd, Headd JJ, Keedy DA, Immormino RM, Kapral GJ, Murray LW, Richardson JS, Richardson DC (2010) MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr D Biol Crystallogr 66(Pt 1):12–21. https://doi.org/10.1107/s0907444909042073
Chen A, Li Y, Nie J, McNeil B, Jeffrey L, Yang Y, Bai Z (2015) Protein engineering of Bacillus acidopullulyticus pullulanase for enhanced thermostability using in silico data driven rational design methods. Enzym Microb Technol 78:74–83. https://doi.org/10.1016/j.enzmictec.2015.06.013
Domań-Pytka M, Bardowski J (2004) Pullulan degrading enzymes of bacterial origin. Crit Rev Microbiol 30(2):107–121. https://doi.org/10.1080/10408410490435115
Ericsson UB, Hallberg BM, Detitta GT, Dekker N, Nordlund P (2006) Thermofluor-based high-throughput stability optimization of proteins for structural studies. Anal Biochem 357(2):289–298. https://doi.org/10.1016/j.ab.2006.07.027
Gao Y, Li JJ, Zheng L, Du Y (2017) Rational design of Pleurotus eryngii versatile ligninolytic peroxidase for enhanced pH and thermal stability through structure-based protein engineering. Protein Eng Des Sel 30(11):743–751. https://doi.org/10.1093/protein/gzx055
Goldenberg O, Erez E, Nimrod G, Ben-Tal N (2009) The ConSurf-DB: pre-calculated evolutionary conservation profiles of protein structures. Nucleic Acids Res 37(Database issue):D323–D327. https://doi.org/10.1093/nar/gkn822
Grant BJ, Rodrigues AP, ElSawy KM, McCammon JA, Caves LS (2006) Bio3d: an R package for the comparative analysis of protein structures. Bioinformatics 22(21):2695–2696. https://doi.org/10.1093/bioinformatics/btl461
Gribenko AV, Patel MM, Liu J, McCallum SA, Wang C, Makhatadze GI (2009) Rational stabilization of enzymes by computational redesign of surface charge-charge interactions. Proc Natl Acad Sci U S A 106(8):2601–2606. https://doi.org/10.1073/pnas.0808220106
Gromiha MM, Pathak MC, Saraboji K, Ortlund EA, Gaucher EA (2013) Hydrophobic environment is a key factor for the stability of thermophilic proteins. Proteins 81(4):715–721. https://doi.org/10.1002/prot.24232
Guerois R, Nielsen JE, Serrano L (2002) Predicting changes in the stability of proteins and protein complexes: a study of more than 1000 mutations. J Mol Biol 320(2):369–387. https://doi.org/10.1016/s0022-2836(02)00442-4
Hua H, Luo H, Bai Y, Wang K, Niu C, Huang H, Shi P, Wang C, Yang P, Yao B (2014) A thermostable glucoamylase from Bispora sp. MEY-1 with stability over a broad pH range and significant starch hydrolysis capacity. PLoS One 9(11):e113581. https://doi.org/10.1371/journal.pone.0113581
Johnson RJ, Savas CJ, Kartje Z, Hoops GC (2014) Rapid and adaptable measurement of protein thermal stability by differential scanning fluorimetry: updating a common biochemical laboratory experiment. J Chem Educ 91(7):1077–1080. https://doi.org/10.1021/ed400783e
Khan S, Vihinen M (2010) Performance of protein stability predictors. Hum Mutat 31(6):675–684. https://doi.org/10.1002/humu.21242
Korkegian A, Black ME, Baker D, Stoddard BL (2005) Computational thermostabilization of an enzyme. Science 308(5723):857–860. https://doi.org/10.1126/science.1107387
Kumar V (2010) Analysis of the key active subsites of glycoside hydrolase 13 family members. Carbohydr Res 345(7):893–898. https://doi.org/10.1016/j.carres.2010.02.007
Laskowski R, Macarthur MW, Moss DS, Thornton J (1993) PROCHECK: a program to check the stereochemical quality of protein structures. J Appl Crystallogr 26:283–291. https://doi.org/10.1107/S0021889892009944
Li WF, Zhou XX, Lu P (2005) Structural features of thermozymes. Biotechnol Adv 23(4):271–281. https://doi.org/10.1016/j.biotechadv.2005.01.002
Li Y, Zhang L, Ding Z, Gu Z, Shi G (2016) Engineering of isoamylase: improvement of protein stability and catalytic efficiency through semi-rational design. J Ind Microbiol Biotechnol 43(1):3–12. https://doi.org/10.1007/s10295-015-1708-4
Li G, Chen Y, Fang X, Su F, Yunjun Y (2018a) Identification of a hot-spot to enhance Candida rugosa lipase thermostability by rational design methods. RSC Adv 8(4):1948–1957. https://doi.org/10.1039/C7RA11679A
Li L, Dong F, Lin L, He D, Wei W, Wei D (2018b) N-terminal domain truncation and domain insertion-based engineering of a novel thermostable type I pullulanase from Geobacillus thermocatenulatus. J Agric Food Chem 66(41):10788–10798. https://doi.org/10.1021/acs.jafc.8b03331
Mahadevi AS, Sastry GN (2013) Cation-π interaction: its role and relevance in chemistry, biology, and material science. Chem Rev 113(3):2100–2138. https://doi.org/10.1021/cr300222d
Morley KL, Kazlauskas RJ (2005) Improving enzyme properties: when are closer mutations better? Trends Biotechnol 23(5):231–237. https://doi.org/10.1016/j.tibtech.2005.03.005
Moroder L, Budisa N (2010) Synthetic biology of protein folding. Chemphyschem 11(6):1181–1187. https://doi.org/10.1002/cphc.201000035
Pace CN, Fu H, Fryar KL, Landua J, Trevino SR, Shirley BA, Hendricks MM, Iimura S, Gajiwala K, Scholtz JM, Grimsley GR (2011) Contribution of hydrophobic interactions to protein stability. J Mol Biol 408(3):514–528. https://doi.org/10.1016/j.jmb.2011.02.053
Piovesan D, Minervini G, Tosatto SC (2016) The RING 2.0 web server for high quality residue interaction networks. Nucleic Acids Res 44(W1):W367–W374. https://doi.org/10.1093/nar/gkw315
Reetz MT, Carballeira JD, Vogel A (2006) Iterative saturation mutagenesis on the basis of B factors as a strategy for increasing protein thermostability. Angew Chem Int Ed Eng 45(46):7745–7751. https://doi.org/10.1002/anie.200602795
Renugopalakrishnan V, Garduño-Juárez R, Narasimhan G, Verma CS, Wei X, Li P (2005) Rational design of thermally stable proteins: relevance to bionanotechnology. J Nanosci Nanotechnol 5(11):1759–1767. https://doi.org/10.1166/jnn.2005.441
Ruggiero A, Smaldone G, Esposito L, Balasco N, Vitagliano L (2019) Loop size optimization induces a strong thermal stabilization of the thioredoxin fold. FEBS J 286(9):1752–1764. https://doi.org/10.1111/febs.14767
Shafee T (2014) Evolvability of a viral protease: experimental evolution of catalysis, robustness and specificity. Dissertation, University of Cambridge
Shoulders MD, Satyshur KA, Forest KT, Raines RT (2010) Stereoelectronic and steric effects in side chains preorganize a protein main chain. Proc Natl Acad Sci U S A 107(2):559–564. https://doi.org/10.1073/pnas.0909592107
Skjærven L, Yao XQ, Scarabelli G, Grant BJ (2014) Integrating protein structural dynamics and evolutionary analysis with Bio3D. BMC Bioinf 15(1):399. https://doi.org/10.1186/s12859-014-0399-6
Stepankova V, Bidmanova S, Koudelakova T, Prokop Z, Chaloupkova R, Damborsky J (2013) Strategies for stabilization of enzymes in organic solvents. ACS Catal 3(12):2823–2836. https://doi.org/10.1021/cs400684x
Sun YZ, Chen XB, Wang RR, Li WY, Ma Y (2019a) Exploring the effect of N308D mutation on protein tyrosine phosphatase-2 cause gain-of-function activity by a molecular dynamics study. J Cell Biochem 120(4):5949–5961. https://doi.org/10.1002/jcb.27883
Sun Z, Liu Q, Qu G, Feng Y, Reetz MT (2019b) Utility of B-factors in protein science: interpreting rigidity, flexibility, and internal motion and engineering thermostability. Chem Rev 119(3):1626–1665. https://doi.org/10.1021/acs.chemrev.8b00290
Swift ML (1997) GraphPad prism, data analysis, and scientific graphing. J Chem Inf Comput Sci 37(2):411–412. https://doi.org/10.1021/ci960402j
Wang X, Nie Y, Xu Y (2018) Improvement of the activity and stability of starch-debranching pullulanase from Bacillus naganoensis via tailoring of the active sites lining the catalytic pocket. J Agric Food Chem 66(50):13236–13242. https://doi.org/10.1021/acs.jafc.8b06002
Wang X, Nie Y, Xu Y (2019) Industrially produced pullulanases with thermostability: discovery, engineering, and heterologous expression. Bioresour Technol 278:360–371. https://doi.org/10.1016/j.biortech.2019.01.098
Webb B, Sali A (2016) Comparative protein structure modeling using MODELLER. Curr Protoc Bioinformatics 54:5.6.1–5.6.37. https://doi.org/10.1002/cpbi.3
Whitley MJ, Lee AL (2009) Frameworks for understanding long-range intra-protein communication. Curr Protein Pept Sci 10(2):116–127. https://doi.org/10.2174/138920309787847563
Wijma HJ, Floor RJ, Jekel PA, Baker D, Marrink SJ, Janssen DB (2014) Computationally designed libraries for rapid enzyme stabilization. Protein Eng Des Sel 27(2):49–58. https://doi.org/10.1093/protein/gzt061
Wu H, Yu X, Chen L, Wu G (2014) Cloning, overexpression and characterization of a thermostable pullulanase from Thermus thermophilus HB27. Protein Expr Purif 95:22–27. https://doi.org/10.1016/j.pep.2013.11.010
Wu Z, Kan SBJ, Lewis RD, Wittmann BJ, Arnold FH (2019) Machine learning-assisted directed protein evolution with combinatorial libraries. Proc Natl Acad Sci U S A 116(18):8852–8858. https://doi.org/10.1073/pnas.1901979116
Xu Z, Cen YK, Zou SP, Xue YP, Zheng YG (2020) Recent advances in the improvement of enzyme thermostability by structure modification. Crit Rev Biotechnol 40(1):83–98. https://doi.org/10.1080/07388551.2019.1682963
Yang Y, Zhou Y (2008a) Ab initio folding of terminal segments with secondary structures reveals the fine difference between two closely related all-atom statistical energy functions. Protein Sci 17(7):1212–1219. https://doi.org/10.1110/ps.033480.107
Yang Y, Zhou Y (2008b) Specific interactions for ab initio folding of protein terminal regions with secondary structures. Proteins 72(2):793–803. https://doi.org/10.1002/prot.21968
You C, Shi T, Li Y, Han P, Zhou X, Zhang YP (2017) An in vitro synthetic biology platform for the industrial biomanufacturing of myo-inositol from starch. Biotechnol Bioeng 114(8):1855–1864. https://doi.org/10.1002/bit.26314
Yu H, Dalby PA (2018a) Coupled molecular dynamics mediate long- and short-range epistasis between mutations that affect stability and aggregation kinetics. Proc Natl Acad Sci U S A 115(47):E11043–E11052. https://doi.org/10.1073/pnas.1810324115
Yu H, Dalby PA (2018b) Exploiting correlated molecular-dynamics networks to counteract enzyme activity-stability trade-off. Proc Natl Acad Sci U S A 115(52):E12192–E12200. https://doi.org/10.1073/pnas.1812204115
Yu H, Huang H (2014) Engineering proteins for thermostability through rigidifying flexible sites. Biotechnol Adv 32(2):308–315. https://doi.org/10.1016/j.biotechadv.2013.10.012
Zouari Ayadi D, Ben Ali M, Jemli S, Ben Mabrouk S, Mezghani M, Ben Messaoud E, Bejar S (2008) Heterologous expression, secretion and characterization of the Geobacillus thermoleovorans US105 type I pullulanase. Appl Microbiol Biotechnol 78(3):473–481. https://doi.org/10.1007/s00253-007-1318-9
Acknowledgments
We thank Editage (www.editage.cn) for the English language editing.
Funding
This work was supported by the National Natural Science Foundation of China (31872891, 21676120), the Program of Introducing Talents of Discipline to Universities (111-2-06), the High-End Foreign Experts Recruitment Program (G20190010083), the Program for Advanced Talents within Six Industries of Jiangsu Province (2015-NY-007), the National Program for Support of Top-notch Young Professionals, the Fundamental Research Funds for the Central Universities (JUSRP51504), the Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions, Top-notch Academic Programs Project of Jiangsu Higher Education Institutions, the Program for the Key Laboratory of Enzymes of Suqian (M201803), and the National First-Class Discipline Program of Light Industry Technology and Engineering (LITE2018-09).
Author information
Authors and Affiliations
Contributions
J.B., Y.N., and Y.X. conceived and designed the experiments. J.B., S.C., and X.Z. performed the experiments. J.B. and Y.N. analyzed experimental data. J.B. and Y.N. wrote the main manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 1894 kb)
Rights and permissions
About this article
Cite this article
Bi, J., Chen, S., Zhao, X. et al. Computation-aided engineering of starch-debranching pullulanase from Bacillus thermoleovorans for enhanced thermostability. Appl Microbiol Biotechnol 104, 7551–7562 (2020). https://doi.org/10.1007/s00253-020-10764-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10764-z