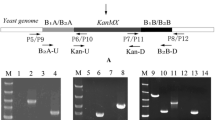
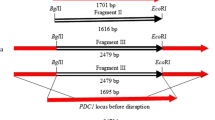
Abstract
Higher alcohols significantly influence the quality and flavor profiles of Chinese Baijiu. ILV1-encoded threonine deaminase, LEU1-encoded α-isopropylmalate dehydrogenase, and LEU2-encoded β-isopropylmalate dehydrogenase are involved in the production of higher alcohols. In this work, ILV1, LEU1, and LEU2 deletions in α-type haploid, a-type haploid, and diploid Saccharomyces cerevisiae strains and ILV1, LEU1, and LEU2 single-allele deletions in diploid strains were constructed to examine the effects of these alterations on the metabolism of higher alcohols. Results showed that different genetic engineering strategies influence carbon flux and higher alcohol metabolism in different manners. Compared with the parental diploid strain, the ILV1 double-allele-deletion diploid mutant produced lower concentrations of n-propanol, active amyl alcohol, and 2-phenylethanol by 30.33, 35.58, and 11.71%, respectively. Moreover, the production of isobutanol and isoamyl alcohol increased by 326.39 and 57.6%, respectively. The LEU1 double-allele-deletion diploid mutant exhibited 14.09% increased n-propanol, 33.74% decreased isoamyl alcohol, and 13.21% decreased 2-phenylethanol production, which were similar to those of the LEU2 mutant. Furthermore, the LEU1 and LEU2 double-allele-deletion diploid mutants exhibited 41.72 and 52.18% increased isobutanol production, respectively. The effects of ILV1, LEU1, and LEU2 deletions on the production of higher alcohols by α-type and a-type haploid strains were similar to those of double-allele deletion in diploid strains. Moreover, the isobutanol production of the ILV1 single-allele-deletion diploid strain increased by 27.76%. Variations in higher alcohol production by the mutants are due to the carbon flux changes in yeast metabolism. This study could provide a valuable reference for further research on higher alcohol metabolism and future optimization of yeast strains for alcoholic beverages.







Similar content being viewed by others
References
Baichwal VR, Cunningham TS, Gatzek PR, Kohlhaw GB (1983) Leucine biosynthesis in yeast: identification of two genes (LEU4, LEU5) that affect α-isopropylmalate synthase activity and evidence that LEU1 and LEU2 gene expression is controlled by α-isopropylmalate and the product of a regulatory gene. Curr Genet 7(5):369–377. https://doi.org/10.1007/BF00445877
Chen X, Nielsen KF, Borodina I, Kielland-Brandt MC, Karhumaa K (2011) Increased isobutanol production in Saccharomyces cerevisiae by overexpression of genes in valine metabolism. Biotechnol Biofuels 4(1):21. https://doi.org/10.1186/1754-6834-4-21
Derrick S, Large PJ (1993) Activities of the enzymes of the Ehrlich pathway and formation of branched-chain alcohols in Saccharomyces cerevisiae and Candida utilis grown in continuous culture on valine or ammonium as sole nitrogen source. J Gen Microbiol 139(11):2783–2792. https://doi.org/10.1099/00221287-139-11-2783
Dickinson JR, Norte V (1993) A study of branched-chain amino acid aminotransferase and isolation of mutations affecting the catabolism of branched-chain amino acids in Saccharomyces cerevisiae. FEBS Lett 326(1-3):29–32. https://doi.org/10.1016/0014-5793(93)81754-N
Eden A, Nedervelde LV, Drukker M, Benvenisty N, Debourg A (2001) Involvement of branched-chain amino acid aminotransferases in the production of fusel alcohols during fermentation in yeast. Appl Microbiol Biotechnol 55(3):296–300. https://doi.org/10.1007/s002530000506
Fan W, Qian MC (2005) Headspace solid phase microextraction and gas chromatography-olfactometry dilution analysis of young and aged Chinese “Yanghe Daqu” liquors. J Agric Food Chem 53(20):7931–7938. https://doi.org/10.1021/jf051011k
Fan W, Qian MC (2006) Characterization of aroma compounds of Chinese “Wuliangye” and “Jiannanchun” liquors by aroma extract dilution analysis. J Agric Food Chem 54(7):2695–2704. https://doi.org/10.1021/jf052635t
Gietz RD, Woods RA (2002) Transformation of yeast by lithium acetate/single-stranded carrier DNA/polyethylene glycol method. Methods Enzymol 350:87–96. https://doi.org/10.1016/S0076-6879(02)50957-5
Güldener U, Heck S, Fielder T, Beinhauer J, Hegemann JH (1996) A new efficient gene disruption cassette for repeated use in budding yeast. Nucleic Acids Res 24(13):2519–2524. https://doi.org/10.1093/nar/24.13.2519
Gueldener U, Heinisch J, Koehler GJ, Voss D, Hegemann JH (2002) A second set of loxP marker cassettes for Cre-mediated multiple gene deletions in budding yeast. Nucleic Acids Res 30(6):88–94. https://doi.org/10.1093/nar/30.6.e23
Holmberg S, Petersen JG (1988) Regulation of isoleucine-valine biosynthesis in Saccharomyces cerevisiae. Curr Genet 13(3):207–217. https://doi.org/10.1007/BF00387766
Hsu YP, Schimmel P (1984) Yeast LEU1. Repression of mRNA levels by leucine and relationship of 5′-noncoding region to that of LEU2. J Biol Chem 259(6):3714–3719
Ida K, Ishii J, Matsuda F, Kondo T, Kondo A (2015) Eliminating the isoleucine biosynthetic pathway to reduce competitive carbon outflow during isobutanol production by Saccharomyces cerevisiae. Microb Cell Factories 14(1):1–9. https://doi.org/10.1186/s12934-015-0240-6
Kobayashi M, Shimizu H, Shioya S (2008) Beer volatile compounds and their application to low-malt beer fermentation. J Biosci Bioeng 106(4):317–323. https://doi.org/10.1263/jbb.106.317
Kondo T, Tezuka H, Ishii J, Matsuda F, Ogino C, Kondo A (2012) Genetic engineering to enhance the Ehrlich pathway and alter carbon flux for increased isobutanol production from glucose by Saccharomyces cerevisiae. J Biotechnol 159(1-2):32–37. https://doi.org/10.1016/j.jbiotec.2012.01.022
Li W, Wang JH, Zhang CY, Ma HX, Xiao DG (2017) Regulation of Saccharomyces cerevisiae genetic engineering on the production of acetate esters and higher alcohols during Chinese baijiu fermentation. J Ind Microbiol Biotechnol 44(6):949–960. https://doi.org/10.1007/s10295-017-1907-2
Lu J, Dong J, Wu D, Chen Y, Guo X, Shi Y, Sun X, Xiao D (2012) Construction of recombinant industrial brewer’s yeast with lower diacetyl production and proteinase A activity. Eur Food Res Technol 235(5):951–961. https://doi.org/10.1007/s00217-012-1821-9
Nigavekar SS, Cannon JF (2002) Characterization of genes that are synthetically lethal with ade3 or leu2 in Saccharomyces cerevisiae. Yeast 19(2):115–122. https://doi.org/10.1002/yea.807
Park SH, Kim S, Hahn JS (2014) Metabolic engineering of Saccharomyces cerevisiae for the production of isobutanol and 3-methyl-1-butanol. Appl Microbiol Biotechnol 98(21):9139–9147. https://doi.org/10.1007/s00253-014-6081-0
Petersen JGL, Holmberg S, Nilsson-Tillgren T, Kielland-Brandt MC (1983) Molecular cloning and characterization of the threonine deaminase (ILV1) gene of Saccharomyces cerevisiae. Carlsb Res Commun 48(3):149–159. https://doi.org/10.1007/BF02907764
Pires EJ, Teixeira JA, Brányik T, Vicente AA (2014) Yeast: the soul of beer's aroma-a review of flavour-active esters and higher alcohols produced by the brewing yeast. Appl Microbiol Biotechnol 98(5):1937–1949. https://doi.org/10.1007/s00253-013-5470-0
Ryan ED, Kohlhaw GB (1974) Subcellular localization of isoleucine-valine biosynthetic enzymes in yeast. J Bacteriol 120(2):631–637
Sakai Y, Tani Y (1992) Directed mutagenesis in an asporogenous methylotrophic yeast: cloning, sequencing, and one-step gene disruption of the 3-isopropylmalate dehydrogenase gene (LEU2) of Candida boidinii to derive doubly auxotrophic marker strains. J Bacteriol 174(18):5988–5993. https://doi.org/10.1128/jb.174.18.5988-5993.1992
Shi S, Tong S, Liu Z, Zhang H, Ang EL, Zhao H (2016) Metabolic engineering of a synergistic pathway for n-butanol production in Saccharomyces cerevisiae. Sci Rep 6(1):25675. https://doi.org/10.1038/srep25675
Stribny J, Romagnoli G, Pérez-Torrado R, Daran JM, Querol A (2016) Characterisation of the broad substrate specificity 2-keto acid decarboxylase Aro10p of Saccharomyces kudriavzevii and its implication in aroma development. Microb Cell Factories 15(1):1–12. https://doi.org/10.1186/s12934-016-0449-z
Swiegers JH, Pretorius IS (2005) Yeast modulation of wine flavor. Adv Appl Microbiol 57:131–175. https://doi.org/10.1016/S0065-2164(05)57005-9
Valero E, Moyano L, Millan MC, Medinab M, Ortega JM (2002) Higher alcohols and esters production by Saccharomyces cerevisiae. Influence of the initial oxygenation of the grape must. Food Chem 78(1):57–61. https://doi.org/10.1016/S0308-8146(01)00361-2
Vollbrecht D (1974) Three pathways of isoleucine biosynthesis in mutant strains of Saccharomyces cerevisiae. Biochim Biophys Acta 362(2):382–389. https://doi.org/10.1016/0304-4165(74)90231-1
Wang CL, Shi DJ, Gong GL (2008) Microorganisms in Daqu: a starter culture of Chinese Maotai-flavor liquor. World J Microbiol Biotechnol 24(10):2183–2190. https://doi.org/10.1007/s11274-008-9728-0
Wu Q, Kong Y, Xu Y (2015) Flavor profile of Chinese liquor is altered by interactions of intrinsic and extrinsic microbes. Appl Environ Microbiol 82(2):422–430. https://doi.org/10.1128/AEM.02518-15
Xiao ZB, Yu D, Niu YW, Chen F, Song SQ, Zhu JC, Zhu GY (2014) Characterization of aroma compounds of Chinese famous liquors by gas chromatography-mass spectrometry and flash GC electronic-nose. J Chromatogr B 945-946:92–100. https://doi.org/10.1016/j.jchromb.2013.11.032
Yang D, Luo X, Wang X (2014) Characteristics of traditional Chinese shanlan wine fermentation. J Biosci Bioeng 117(2):203–207. https://doi.org/10.1016/j.jbiosc.2013.07.010
Yuan J, Ching CB (2015) Combinatorial assembly of large biochemical pathways into yeast chromosomes for improved production of value-added compounds. ACS Synth Biol 4(1):23–31. https://doi.org/10.1021/sb500079f
Yuan J, Mishra P, Ching CB (2016) Metabolically engineered Saccharomyces cerevisiae for branched-chain ester productions. J Biotechnol 239:90–97. https://doi.org/10.1016/j.jbiotec.2016.10.013
Yuan J, Chen X, Mishra P, Ching CB (2017a) Metabolically engineered Saccharomyces cerevisiae for enhanced isoamyl alcohol production. Appl Microbiol Biotechnol 101(1):465–474. https://doi.org/10.1007/s00253-016-7970-1
Yuan J, Mishra P, Ching CB (2017b) Engineering the leucine biosynthetic pathway for isoamyl alcohol overproduction in Saccharomyces cerevisiae. J Ind Microbiol Biotechnol 44(1):107–117. https://doi.org/10.1007/s10295-016-1855-2
Zheng XW, Tabrizi MR, Nout MJR, Han BZ (2011) Daqu—a traditional Chinese liquor fermentation starter. J I Brewing 117(1):82–90. https://doi.org/10.1002/j.2050-0416.2011.tb00447.x
Zhu S, Lu X, Ji K, Guo K, Li Y, Wu C, Xu G (2007) Characterization of flavor compounds in Chinese liquor Moutai by comprehensive two-dimensional gas chromatography/time-of-flight mass spectrometry. Anal Chim Acta 597(2):340–348. https://doi.org/10.1016/j.aca.2007.07.007
Acknowledgments
This work was supported by the National Key Research and Development Program of China (2016YFD0400505), the National Natural Science Foundation of China (31471724), the Major Project of Research Program on Applied Fundamentals and Advanced Technologies of Tianjin (14JCZDJC32900), and the National High Technology Research and Development Program of China (2013AA102108).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical statement
This manuscript is in compliance with ethical standards. This manuscript does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
ESM 1
(PDF 410 kb)
Rights and permissions
About this article
Cite this article
Li, W., Chen, SJ., Wang, JH. et al. Genetic engineering to alter carbon flux for various higher alcohol productions by Saccharomyces cerevisiae for Chinese Baijiu fermentation. Appl Microbiol Biotechnol 102, 1783–1795 (2018). https://doi.org/10.1007/s00253-017-8715-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-017-8715-5