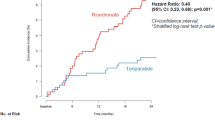
Abstract
The phase 3 teriparatide Fracture Prevention Trial showed significant reductions in vertebral (VF) and nonvertebral (NVF) fractures; however, patient exposure was insufficient for full analysis of low-incidence fractures, including hip. We assessed fracture results in pooled data from four prospective, observational teriparatide studies. Ambulatory women and men with osteoporosis received subcutaneous teriparatide 20 µg/day for up to 24 months per routine clinical practice. Fracture rates were compared between 6-month periods, using 0 to 6 months of treatment as the reference period. Analyses used a piecewise exponential model for first fracture. Hip, NVF, clinical VF (CVF), any clinical, and wrist fractures were assessed. For 8828 patients analyzed, mean age was 71 years; mean (SD) treatment duration was 17.4 (8.6) months. The rate of hip fracture decreased significantly for the > 12 to 18-month (− 47.7%) and > 18-month periods (-85.2%) versus the first 6 months of therapy, and for the > 18 versus the > 6 to 12-month period. NVF, CVF, and all clinical fractures were all significantly decreased in each post-reference period, with maximum decreases (> 18-month period) of 52.7%, 69.4%, and 61.2%, respectively, versus 0 to 6 months. No significant reduction was seen for rates of wrist fracture. Teriparatide treatment was associated with statistically significant decreases in hip fracture rate, particularly for > 18 months of treatment, and in NVF, CVF, and all clinical fracture rate in real-world patients. These results should be interpreted in the context of the non-controlled design of the source studies.

Similar content being viewed by others
References
Curtis EM, Moon RJ, Harvey NC, Cooper C (2017) The impact of fragility fracture and approaches to osteoporosis risk assessment worldwide. Bone 104:29–38
Boonen S, Autier P, Barette M, Vanderschueren D, Lips P, Haentjens P (2004) Functional outcome and quality of life following hip fracture in elderly women: a prospective controlled study. Osteoporos Int 15:87–94
Magaziner J, Simonsick EM, Kashner TM, Hebel JR, Kenzora JE (1990) Predictors of functional recovery one year following hospital discharge for hip fracture: a prospective study. J Gerontol 45:M101–M107
Magaziner J, Lydick E, Hawkes W, Fox KM, Zimmerman SI, Epstein RS, Hebel JR (1997) Excess mortality attributable to hip fracture in white women aged 70 years and older. Am J Public Health 87:1630–1636
van Staa TP, Leufkens HG, Cooper C (2002) Does a fracture at one site predict later fractures at other sites? A British cohort study. Osteoporos Int 13:624–629
Kanis JA, Oden A, McCloskey EV, Johansson H, Wahl DA, Cooper C (2012) A systematic review of hip fracture incidence and probability of fracture worldwide. Osteoporos Int 23:2239–2256
Neer RM, Arnaud CD, Zanchetta JR, Prince R, Gaich GA, Reginster JY, Hodsman AB, Eriksen EF, Ish-Shalom S, Genant HK, Wang O, Mitlak BH (2001) Effect of parathyroid hormone (1–34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N Engl J Med 344:1434–1441
Eriksen EF, Keaveny TM, Gallagher ER, Krege JH (2014) Literature review: The effects of teriparatide therapy at the hip in patients with osteoporosis. Bone 67:246–256
Burge RT, Disch DP, Gelwicks S, Zhang X, Krege JH (2017) Hip and other fragility fracture incidence in real-world teriparatide-treated patients in the United States. Osteoporos Int 28:799–809
Silverman S, Miller P, Sebba A, Weitz M, Wan X, Alam J, Masica D, Taylor KA, Ruff VA, Krohn K (2013) The direct assessment of nonvertebral fractures in community experience (DANCE) study: 2-year nonvertebral fragility fracture results. Osteoporos Int 24:2309–2317
Langdahl BL, Rajzbaum G, Jakob F, Karras D, Ljunggren O, Lems WF, Fahrleitner-Pammer A, Walsh JB, Barker C, Kutahov A, Marin F (2009) Reduction in fracture rate and back pain and increased quality of life in postmenopausal women treated with teriparatide: 18-month data from the European Forsteo Observational Study (EFOS). Calcif Tissue Int 85:484–493
Langdahl BL, Ljunggren O, Benhamou CL, Marin F, Kapetanos G, Kocjan T, Lespessailles E, Napoli N, Nikolic T, Petto H, Moll T, Lindh E (2016) Fracture rate, quality of life and back pain in patients with osteoporosis treated with teriparatide: 24-Month Results from the Extended Forsteo Observational Study (ExFOS). Calcif Tissue Int 99:259–271
Soen S, Fujiwara S, Takayanagi R, Kajimoto K, Tsujimoto M, Kimura S, Sato M, Krege JH, Enomoto H (2017) Real-world effectiveness of daily teriparatide in Japanese patients with osteoporosis at high risk for fracture: final results from the 24-month Japan Fracture Observational Study (JFOS). Curr Med Res Opin 33:2049–2056
Miller PD, Silverman SL, Gold DT, Taylor KA, Chen P, Wagman RB (2006) Rationale, objectives and design of the direct analysis of nonvertebral fracture in the community experience (DANCE) study. Osteoporos Int 17:85–90
Rajzbaum G, Jakob F, Karras D, Ljunggren O, Lems WF, Langdahl BL, Fahrleitner-Pammer A, Walsh JB, Gibson A, Tynan AJ, Marin F (2008) Characterization of patients in the European Forsteo Observational Study (EFOS): postmenopausal women entering teriparatide treatment in a community setting. Curr Med Res Opin 24:377–384
Ljunggren O, Benhamou CL, Dekker J, Kapetanos G, Kocjan T, Langdahl BL, Napoli N, Petto H, Nikolic T, Lindh E (2014) Study description and baseline characteristics of the population enrolled in a multinational observational study of extended teriparatide use (ExFOS). Curr Med Res Opin 30:1607–1616
Soen S, Fujiwara S, Takayanagi R, Sato M, Tsujimoto M, Yamamoto T, Enomoto H, Krege JH (2015) Japan Fracture Observational Study (JFOS): patient characteristics and interim data on the use of daily teriparatide in Japanese patients with osteoporosis. Curr Med Res Opin 31:1771–1779
Lindsay R, Miller P, Pohl G, Glass EV, Chen P, Krege JH (2009) Relationship between duration of teriparatide therapy and clinical outcomes in postmenopausal women with osteoporosis. Osteoporos Int 20:943–948
Schwartz AV, Pavo I, Alam J, Disch DP, Schuster D, Harris JM, Krege JH (2016) Teriparatide in patients with osteoporosis and type 2 diabetes. Bone 91:152–158
Prevrhal S, Krege JH, Chen P, Genant H, Black DM (2009) Teriparatide vertebral fracture risk reduction determined by quantitative and qualitative radiographic assessment. Curr Med Res Opin 25:921–928
Kendler DL, Marin F, Zerbini CA, Russo LA, Greenspan SL, Zikan V, Bagur A, Malouf-Sierra J, Lakatos P, Fahrleitner-Pammer A, Lespessailles E, Minisola S, Body JJ, Geusens P, Moricke R, Lopez-Romero P (2017) Effects of teriparatide and risedronate on new fractures in post-menopausal women with severe osteoporosis (VERO): a multicentre, double-blind, double-dummy, randomised controlled trial. Lancet 391:230–240
Haentjens P, Magaziner J, Colon-Emeric CS, Vanderschueren D, Milisen K, Velkeniers B, Boonen S (2010) Meta-analysis: excess mortality after hip fracture among older women and men. Ann Intern Med 152:380–390
Magaziner J, Hawkes W, Hebel JR, Zimmerman SI, Fox KM, Dolan M, Felsenthal G, Kenzora J (2000) Recovery from hip fracture in eight areas of function. J Gerontol A 55:M498–M507
Dyer SM, Crotty M, Fairhall N, Magaziner J, Beaupre LA, Cameron ID, Sherrington C (2016) A critical review of the long-term disability outcomes following hip fracture. BMC Geriatr 16:158
Johnell O, Kanis JA (2004) An estimate of the worldwide prevalence, mortality and disability associated with hip fracture. Osteoporos Int 15:897–902
Hopkins RB, Goeree R, Pullenayegum E, Adachi JD, Papaioannou A, Xie F, Thabane L (2011) The relative efficacy of nine osteoporosis medications for reducing the rate of fractures in post-menopausal women. BMC Musculoskelet Disord 12:209
Miller PD, Hattersley G, Riis BJ, Williams GC, Lau E, Russo LA, Alexandersen P, Zerbini CA, Hu MY, Harris AG, Fitzpatrick LA, Cosman F, Christiansen C (2016) Effect of Abaloparatide vs Placebo on new vertebral fractures in postmenopausal women with osteoporosis: a randomized clinical trial. JAMA 316:722–733
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
S.S. is on the Speaker’s Bureau and is a consultant for and has received research support from Eli Lilly and Co.; K.S. has received consultant fees and has research Grants from Eli Lilly and Co.; B.L.L. has received fees for serving on advisory boards, speaking at symposia, and has received research grants from Eli Lilly and Co. Amgen, UCB, Merck, and Novo Nordisk; NN has received consultant and speaker fees from Eli Lilly and Co.; S.F. and S.S. have nothing to declare; T.E.M. is an employee of Syneos Health under contract to Eli Lilly and Co.; D.P.D., F.M., H.E., and J.H.K. are employees of Eli Lilly and Co.
Ethical Approval
The studies analyzed herein were approved by local ethics committees or review boards, depending on local requirements, and were conducted in compliance with all applicable laws, regulations and guidelines.
Human and Animal Rights and Informed Consent
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Written informed consent was obtained from all individual patients included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Silverman, S., Langdahl, B.L., Fujiwara, S. et al. Reduction of Hip and Other Fractures in Patients Receiving Teriparatide in Real-World Clinical Practice: Integrated Analysis of Four Prospective Observational Studies. Calcif Tissue Int 104, 193–200 (2019). https://doi.org/10.1007/s00223-018-0485-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-018-0485-2