Abstract
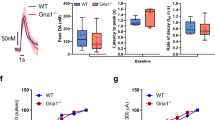
Schizophrenia is a disabling psychiatric disease characterized by symptoms including hallucinations, delusions, social withdrawal, loss of pleasure, and inappropriate affect. Although schizophrenia is marked by dysfunction in dopaminergic and glutamatergic signaling, it is not presently clear how these dysfunctions give rise to symptoms. The aberrant salience hypothesis of schizophrenia argues that abnormal attribution of motivational salience to stimuli is one of the main contributors to both positive and negative symptoms of schizophrenia. The proposed mechanisms for this hypothesis are overactive striatal dopaminergic and hypoactive glutamatergic signaling. The current study assessed salience attribution in mice (n = 72) using an oddball paradigm in which an infrequent stimulus either co-occurred with shock (conditioned group) or was presented alone (non-conditioned group). Behavioral response (freezing) and electroencephalogram (whole brain and amygdala) were used to assess salience attribution. Mice with pyramidal cell-selective knockout of ionotropic glutamate receptors (GluN1) were used to reproduce a prominent physiological change involved in schizophrenia. Non-conditioned knockout mice froze significantly more in response to the unpaired stimulus than non-conditioned wild-type mice, suggesting that this irrelevant cue acquired motivational salience for the knockouts. In accordance with this finding, low-frequency event-related spectral perturbation was significantly increased in non-conditioned knockout mice relative to both conditioned knockout and non-conditioned wild-type mice. These results suggest that pyramidal cell-selective GluN1 knockout leads to inappropriate attribution of salience for irrelevant stimuli as characterized by abnormalities in both behavior and brain circuitry functions.




Similar content being viewed by others
References
Barkus C, Sanderson DJ, Rawlins JN, Walton ME, Harrison PJ, Bannerman DM (2014) What causes aberrant salience in schizophrenia? A role for impaired short-term habituation and the GRIA1 (GluA1) AMPA receptor subunit. Mol Psychiatry 19:1060–1070. https://doi.org/10.1038/mp.2014.91
Billingslea EN, Tatard-Leitman VM, Anguiano J et al (2014) Parvalbumin cell ablation of NMDA-R1 causes increased resting network excitability with associated social and self-care deficits. Neuropsychopharmacology 39:1603–1613. https://doi.org/10.1038/npp.2014.7
Carlen M, Meletis K, Siegle JH et al (2012) A critical role for NMDA receptors in parvalbumin interneurons for gamma rhythm induction and behavior. Mol Psychiatry 17:537–548. https://doi.org/10.1038/mp.2011.31
Delorme A, Makeig S (2004) EEGLAB: an open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J Neurosci Methods 134:9–21. https://doi.org/10.1016/j.jneumeth.2003.10.009
Dowd EC, Barch DM (2010) Anhedonia and emotional experience in schizophrenia: neural and behavioral indicators. Biol Psychiatry 67:902–911. https://doi.org/10.1016/j.biopsych.2009.10.020
Goldman-Rakic PS, Selemon LD (1997) Functional and anatomical aspects of prefrontal pathology in schizophrenia. Schizophr Bull 23:437–458
Gur RE, Loughead J, Kohler CG et al (2007) Limbic activation associated with misidentification of fearful faces and flat affect in schizophrenia. Arch Gen Psychiatry 64:1356–1366. https://doi.org/10.1001/archpsyc.64.12.1356
Hall J, Whalley HC, McKirdy JW et al (2008) Overactivation of fear systems to neutral faces in schizophrenia. Biol Psychiatry 64:70–73. https://doi.org/10.1016/j.biopsych.2007.12.014
Hasegawa S, Furuichi T, Yoshida T et al (2009) Transgenic up-regulation of alpha-CaMKII in forebrain leads to increased anxiety-like behaviors and aggression. Mol Brain 2:6. https://doi.org/10.1186/1756-6606-2-6
Heinz A, Schlagenhauf F (2010) Dopaminergic dysfunction in schizophrenia: salience attribution revisited. Schizophr Bull 36:472–485. https://doi.org/10.1093/schbul/sbq031
Howes OD, Kapur S (2009) The dopamine hypothesis of schizophrenia: version III—the final common pathway. Schizophr Bull 35:549–562. https://doi.org/10.1093/schbul/sbp006
Howes O, McCutcheon R, Stone J (2015) Glutamate and dopamine in schizophrenia: an update for the 21st century. J Psychopharmacol 29:97–115. https://doi.org/10.1177/0269881114563634
Kapur S (2003) Psychosis as a state of aberrant salience: a framework linking biology, phenomenology, and pharmacology in schizophrenia. Am J Psychiatry 160:13–23
Kilts CD (2001) The changing roles and targets for animal models of schizophrenia. Biol Psychiatry 50:845–855
Korotkova T, Fuchs EC, Ponomarenko A, von Engelhardt J, Monyer H (2010) NMDA receptor ablation on parvalbumin-positive interneurons impairs hippocampal synchrony, spatial representations, and working memory. Neuron 68:557–569. https://doi.org/10.1016/j.neuron.2010.09.017
Lau CI, Wang HC, Hsu JL, Liu ME (2013) Does the dopamine hypothesis explain schizophrenia? Rev Neurosci 24:389–400. https://doi.org/10.1515/revneuro-2013-0011
Lawrie SM, Abukmeil SS (1998) Brain abnormality in schizophrenia. A systematic and quantitative review of volumetric magnetic resonance imaging studies. Br J Psychiatry J Ment Sci 172:110–120
Leitman DI, Loughead J, Wolf DH et al (2008) Abnormal superior temporal connectivity during fear perception in schizophrenia. Schizophr Bull 34:673–678. https://doi.org/10.1093/schbul/sbn052
Lewis DA, Glantz LA, Pierri JN, Sweet RA (2003) Altered cortical glutamate neurotransmission in schizophrenia: evidence from morphological studies of pyramidal neurons. Ann N Y Acad Sci 1003:102–112
Mier D, Lis S, Zygrodnik K, Sauer C, Ulferts J, Gallhofer B, Kirsch P (2014) Evidence for altered amygdala activation in schizophrenia in an adaptive emotion recognition task. Psychiatry Res 221:195–203. https://doi.org/10.1016/j.pscychresns.2013.12.001
Moessnang C, Habel U, Schneider F, Siegel SJ (2012) The electrophysiological signature of motivational salience in mice and implications for schizophrenia. Neuropsychopharmacology 37:2846–2854. https://doi.org/10.1038/npp.2012.156
Moriya T, Kouzu Y, Shibata S, Kadotani H, Fukunaga K, Miyamoto E, Yoshioka T (2000) Close linkage between calcium/calmodulin kinase II alpha/beta and NMDA-2A receptors in the lateral amygdala and significance for retrieval of auditory fear conditioning. Eur J Neurosci 12:3307–3314
Palaniyappan L, White TP, Liddle PF (2012) The concept of salience network dysfunction in schizophrenia: from neuroimaging observations to therapeutic opportunities. Curr Top Med Chem 12:2324–2338
Phillips ML, Williams L, Senior C et al (1999) A differential neural response to threatening and non-threatening negative facial expressions in paranoid and non-paranoid schizophrenics. Psychiatry Res 92:11–31
Roiser JP, Stephan KE, den Ouden HE, Barnes TR, Friston KJ, Joyce EM (2009) Do patients with schizophrenia exhibit aberrant salience? Psychol Med 39:199–209. https://doi.org/10.1017/S0033291708003863
Satterthwaite TD, Wolf DH, Loughead J et al (2010) Association of enhanced limbic response to threat with decreased cortical facial recognition memory response in schizophrenia. Am J Psychiatry 167:418–426. https://doi.org/10.1176/appi.ajp.2009.09060808
Sobota R, Mihara T, Forrest A, Featherstone RE, Siegel SJ (2015) Oxytocin reduces amygdala activity, increases social interactions, and reduces anxiety-like behavior irrespective of NMDAR antagonism. Behav Neurosci 129:389–398. https://doi.org/10.1037/bne0000074
Tatard-Leitman VM, Jutzeler CR, Suh J et al (2015) Pyramidal cell selective ablation of N-methyl-d-aspartate receptor 1 causes increase in cellular and network excitability. Biol Psychiatry 77:556–568. https://doi.org/10.1016/j.biopsych.2014.06.026
Taylor SF, Phan KL, Britton JC, Liberzon I (2005) Neural response to emotional salience in schizophrenia. Neuropsychopharmacology 30:984–995. https://doi.org/10.1038/sj.npp.1300679
Tsien JZ, Chen DF, Gerber D et al (1996) Subregion- and cell type-restricted gene knockout in mouse brain. Cell 87:1317–1326. https://doi.org/10.1016/s0092-8674(00)81826-7
Volk DW, Lewis DA (2010) Prefrontal cortical circuits in schizophrenia. Curr Top Behav Neurosci 4:485–508
Wang X, Zhang C, Szabo G, Sun QQ (2013) Distribution of CaMKIIalpha expression in the brain in vivo, studied by CaMKIIalpha-GFP mice. Brain Res 1518:9–25. https://doi.org/10.1016/j.brainres.2013.04.042
Wehner JM, Radcliffe RA (2004) Cued and contextual fear conditioning in mice. Curr Protoc Neurosci Chap 8 Unit 8 5C. https://doi.org/10.1002/0471142301.ns0805cs27
Wild B, Erb M, Bartels M (2001) Are emotions contagious? Evoked emotions while viewing emotionally expressive faces: quality, quantity, time course and gender differences. Psychiatry Res 102:109–124
Acknowledgements
Funding for the current study was provided by 1-P50-MH-096891-01.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Steven Siegel reports grant support from Astellas and Merck that is unrelated to the content of this paper. Alexandra Forrest, Jakyung Bang, Jonathan Bloom, Rebecca Zhang, Christopher Gajewski, and Matthew Luminais were undergraduate students supported on the Education Core of this P50. Mary McMullen, Yuling Liang, and Robert Featherstone have no disclosures.
Rights and permissions
About this article
Cite this article
Forrest, A.D., Bang, J., Featherstone, R.E. et al. Pyramidal cell-selective GluN1 knockout causes impairments in salience attribution and related EEG activity. Exp Brain Res 236, 837–846 (2018). https://doi.org/10.1007/s00221-017-5152-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-017-5152-8