Abstract
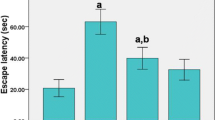
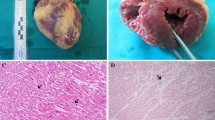
3,4-Methylenedioxymethamphetamine (MDMA or “ecstasy”) is a widespread drug of abuse with known neurotoxic properties. The present study aimed to evaluate the differential toxic effects of MDMA in adolescent and aged Wistar rats, using doses pharmacologically comparable to humans. Adolescent (post-natal day 40) (3 × 5 mg/kg, 2 h apart) and aged (mean 20 months old) (2 × 5 mg/kg, 2 h apart) rats received MDMA intraperitoneally. Animals were killed 7 days later, and the frontal cortex, hippocampus, striatum and cerebellum brain areas were dissected, and heart, liver and kidneys were collected. MDMA caused hyperthermia in both treated groups, but aged rats had a more dramatic temperature elevation. MDMA promoted serotonergic neurotoxicity only in the hippocampus of aged, but not in the adolescents’ brain, and did not change the levels of dopamine or serotonin metabolite in the striatum of both groups. Differential responses according to age were also seen regarding brain p-Tau levels, a hallmark of a degenerative brain, since only aged animals had significant increases. MDMA evoked brain oxidative stress in the hippocampus and striatum of aged, and in the hippocampus, frontal cortex, and striatum brain areas of adolescents according to protein carbonylation, but only decreased GSH levels in the hippocampus of aged animals. The brain maturational stage seems crucial for MDMA-evoked serotonergic neurotoxicity. Aged animals were more susceptible to MDMA-induced tissue damage in the heart and kidneys, and both ages had an increase in liver fibrotic tissue content. In conclusion, age is a determinant factor for the toxic events promoted by “ecstasy”. This work demonstrated special susceptibility of aged hippocampus to MDMA neurotoxicity, as well as impressive damage to the heart and kidney tissue following “ecstasy”.







Similar content being viewed by others
Abbreviations
- 5-HIAA:
-
5-Hydroxyindoleacetic acid
- 5-HT:
-
Serotonin
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- ATP:
-
Adenosine triphosphate
- BSA:
-
Bovine serum albumin
- CHAPS:
-
3-[(3-Cholamidopropyl)dimethylammonio]-1-propanesulfonate hydrate
- CKMB:
-
Creatine kinase MB isoenzyme
- DA:
-
Dopamine
- DTT:
-
Dithiothreitol
- EDTA:
-
Ethylenediamine tetraacetic acid
- GFAP:
-
Glial fibrillary acidic protein
- GSH:
-
Reduced glutathione
- GSHt:
-
Total glutathione
- GSSG:
-
Oxidized glutathione
- HEPES:
-
4-(2-Hydroxyethyl)piperazine-1-ethanesulfonic acid
- 5-HIAA:
-
5-Hydroxyindoleacetic acid
- HNE:
-
Hydroxynonenal
- HPLC:
-
High-performance liquid chromatography
- i.p.:
-
Intraperitoneal
- MDMA:
-
3,4-Methylenedioxymethamphetamine (MDMA; “ecstasy”)
- NA:
-
Noradrenaline
- PBS:
-
Phosphate-buffered saline
- OD:
-
Optic density
- PMSF:
-
Phenylmethanesulfonyl fluoride
- PND:
-
Post-natal day
- p-Tau:
-
Phosphorylated Tau
- ROS:
-
Reactive oxygen species
- SERT:
-
Serotonin transporter
- SDS:
-
Sodium dodecyl sulphate
- TCA:
-
Trichloroacetic acid
- TPH:
-
Tryptophan hydroxylase
References
Ádori C, Andó RD, Szekeres M, Gutknecht L, Kovács GG, Hunyady L, Lesch K-P, Bagdy G (2011) Recovery and aging of serotonergic fibers after single and intermittent MDMA treatment in dark agouti rat. J Comp Neurol 519(12):2353–2378. https://doi.org/10.1002/cne.22631
Alves E, Summavielle T, Alves CJ, Gomes-da-Silva J, Barata JC, Fernandes E, Bastos ML, Tavares MA, Carvalho F (2007) Monoamine oxidase-B mediates ecstasy-induced neurotoxic effects to adolescent rat brain mitochondria. J Neurosci 27(38):10203–10210. https://doi.org/10.1523/jneurosci.2645-07.2007
Alves E, Binienda Z, Carvalho F, Alves CJ, Fernandes E, Bastos ML, Tavares MA, Summavielle T (2009) Acetyl-l-carnitine provides effective in vivo neuroprotection over 3,4-methylenedioximethamphetamine-induced mitochondrial neurotoxicity in the adolescent rat brain. Neuroscience 158(2):514–523. https://doi.org/10.1016/j.neuroscience.2008.10.041
Andreu V, Mas A, Bruguera M, Salmeron JM, Moreno V, Nogue S, Rodes J (1998) Ecstasy: a common cause of severe acute hepatotoxicity. J Hepatol 29(3):394–397. https://doi.org/10.1016/S0168-8278(98)80056-1
Armenian P, Mamantov TM, Tsutaoka BT, Gerona RR, Silman EF, Wu AH, Olson KR (2013) Multiple MDMA (Ecstasy) overdoses at a rave event: a case series. J Intensive Care Med 28(4):252–258. https://doi.org/10.1177/0885066612445982
Badon LA, Hicks A, Lord K, Ogden BA, Meleg-Smith S, Varner KJ (2002) Changes in cardiovascular responsiveness and cardiotoxicity elicited during binge administration of Ecstasy. J Pharmacol Exp Ther 302(3):898–907. https://doi.org/10.1124/jpet.302.3.898
Bantel H, Ruck P, Gregor M, Schulze-Osthoff K (2001) Detection of elevated caspase activation and early apoptosis in liver diseases. Eur J Cell Biol 80(3):230–239. https://doi.org/10.1078/0171-9335-00154
Barbosa DJ, Capela JP, Oliveira JM, Silva R, Ferreira LM, Siopa F, Branco PS, Fernandes E, Duarte JA, Bastos ML, Carvalho F (2012) Pro-oxidant effects of Ecstasy and its metabolites in mouse brain synaptosomes. Br J Pharmacol 165(4b):1017–1033. https://doi.org/10.1111/j.1476-5381.2011.01453.x
Barbosa DJ, Capela JP, Silva R, Vilas-Boas V, Ferreira LM, Branco PS, Fernandes E, Bastos ML, Carvalho F (2014a) The mixture of “ecstasy” and its metabolites is toxic to human SH-SY5Y differentiated cells at in vivo relevant concentrations. Arch Toxicol 88(2):455–473. https://doi.org/10.1007/s00204-013-1120-7
Barbosa DJ, Serrat R, Mirra S, Quevedo M, de Barreda EG, Avila J, Ferreira LM, Branco PS, Fernandes E, Bastos ML, Capela JP, Soriano E, Carvalho F (2014b) The mixture of “ecstasy” and its metabolites impairs mitochondrial fusion/fission equilibrium and trafficking in hippocampal neurons, at in vivo relevant concentrations. Toxicol Sci 139(2):407–420. https://doi.org/10.1093/toxsci/kfu042
Barbosa DJ, Serrat R, Mirra S, Quevedo M, Gómez de Barreda E, Ávila J, Fernandes E, Bastos ML, Capela JP, Carvalho F, Soriano E (2014c) MDMA impairs mitochondrial neuronal trafficking in a Tau- and Mitofusin2/Drp1-dependent manner. Arch Toxicol 88(8):1561–1572. https://doi.org/10.1007/s00204-014-1209-7
Barbosa DJ, Capela JP, Feio-Azevedo R, Teixeira-Gomes A, Bastos ML, Carvalho F (2015) Mitochondria: key players in the neurotoxic effects of amphetamines. Arch Toxicol 89(10):1695–1725. https://doi.org/10.1007/s00204-015-1478-9
Beck BD, Seeley M, Calabrese EJ (2014) The use of toxicology in the regulatory process. In: Wallace Hayes A, Kruger CL (eds) Haye’s principles and methods of toxicology, 6th edn. CRC Press, Boca Raton, pp 35–87
Broening HW, Bowyer JF, Slikker W (1995) Age-dependent sensitivity of rats to the long-term effects of the serotonergic neurotoxicant (+/−)-3,4-methylenedioxymethamphetamine (MDMA) correlates with the magnitude of the MDMA-induced thermal response. J Pharmacol Exp Ther 275(1):325–333
Busceti CL, Biagioni F, Riozzi B, Battaglia G, Storto M, Cinque C, Molinaro G, Gradini R, Caricasole A, Canudas AM, Bruno V, Nicoletti F, Fornai F (2008) Enhanced tau phosphorylation in the Hippocampus of Mice Treated with 3,4-methylenedioxymethamphetamine (“Ecstasy”). J Neurosci 28(12):3234–3245. https://doi.org/10.1523/jneurosci.0159-08.2008
Capela JP, Meisel A, Abreu AR, Branco PS, Ferreira LM, Lobo AM, Remiao F, Bastos ML, Carvalho F (2006) Neurotoxicity of Ecstasy metabolites in rat cortical neurons, and influence of hyperthermia. J Pharmacol Exp Ther 316(1):53–61. https://doi.org/10.1124/jpet.105.092577
Capela JP, Macedo C, Branco PS, Ferreira LM, Lobo AM, Fernandes E, Remiao F, Bastos ML, Dirnagl U, Meisel A, Carvalho F (2007) Neurotoxicity mechanisms of thioether ecstasy metabolites. Neuroscience 146(4):1743–1757. https://doi.org/10.1016/j.neuroscience.2007.03.028
Capela JP, Carmo H, Remiao F, Bastos ML, Meisel A, Carvalho F (2009) Molecular and cellular mechanisms of ecstasy-induced neurotoxicity: an overview. Mol Neurobiol 39(3):210–271. https://doi.org/10.1007/s12035-009-8064-1
Carvalho M, Carmo H, Costa VM, Capela JP, Pontes H, Remião F, Carvalho F, Bastos ML (2012) Toxicity of amphetamines: an update. Arch Toxicol 86(8):1167–1231. https://doi.org/10.1007/s00204-012-0815-5
Che S, Johnson M, Hanson GR, Gibb JW (1995) Body temperature effect on methylenedioxymethamphetamine-induced acute decrease in tryptophan hydroxylase activity. Eur J Pharmacol 293(4):447–453
Colado MI, O’Shea E, Granados R, Misra A, Murray TK, Green AR (1997) A study of the neurotoxic effect of MDMA (‘ecstasy’) on 5-HT neurones in the brains of mothers and neonates following administration of the drug during pregnancy. Br J Pharmacol 121(4):827–833. https://doi.org/10.1038/sj.bjp.0701201
Costa VM, Silva R, Ferreira LM, Branco PS, Carvalho F, Bastos ML, Carvalho RA, Carvalho M, Remiao F (2007) Oxidation process of adrenaline in freshly isolated rat cardiomyocytes: formation of adrenochrome, quinoproteins, and GSH adduct. Chem Res Toxicol 20(8):1183–1191. https://doi.org/10.1021/tx7000916
Costa VM, Carvalho F, Bastos ML, Carvalho RA, Carvalho M, Remiao F (2011) Contribution of catecholamine reactive intermediates and oxidative stress to the pathologic features of heart diseases. Curr Med Chem 18(15):2272–2314. https://doi.org/10.2174/092986711795656081
Cowan RL, Lyoo IK, Sung SM, Ahn KH, Kim MJ, Hwang J, Haga E, Vimal RLP, Lukas SE, Renshaw PF (2003) Reduced cortical gray matter density in human MDMA (Ecstasy) users: a voxel-based morphometry study. Drug Alcohol Depend 72(3):225–235. https://doi.org/10.1016/j.drugalcdep.2003.07.001
Cuyas E, Robledo P, Pizarro N, Farre M, Puerta E, Aguirre N, de la Torre R (2014) 3,4-methylenedioxymethamphetamine induces gene expression changes in rats related to serotonergic and dopaminergic systems, but not to neurotoxicity. Neurotox Res 25(2):161–169. https://doi.org/10.1007/s12640-013-9416-1
de Champlain J, Krakoff LR, Axelrod J (1967) Catecholamine Metabolism in Experimental Hypertension in the Rat. Circ Res 20(1):136–145. https://doi.org/10.1161/01.res.20.1.136
Dores-Sousa JL, Duarte JA, Seabra V, Bastos ML, Carvalho F, Costa VM (2015) The age factor for mitoxantrone’s cardiotoxicity: Multiple doses render the adult mouse heart more susceptible to injury. Toxicology 329:106–119. https://doi.org/10.1016/j.tox.2015.01.006
EMCDDA (2017) European Drug Report 2017: Trends and developments. European Monitoring Centre for Drugs and Drug Addiction, Luxembourg
Garcia-Cabrerizo R, Garcia-Fuster MJ (2015) Chronic MDMA induces neurochemical changes in the hippocampus of adolescent and young adult rats: down-regulation of apoptotic markers. Neurotoxicology 49:104–113. https://doi.org/10.1016/j.neuro.2015.06.001
Goni-Allo B, Mathuna BO, Segura M, Puerta E, Lasheras B, de la Torre R, Aguirre N (2008a) The relationship between core body temperature and 3,4-methylenedioxymethamphetamine metabolism in rats: implications for neurotoxicity. Psychopharmacology 197(2):263–278. https://doi.org/10.1007/s00213-007-1027-1
Goni-Allo B, Puerta E, Mathuna BO, Hervias I, Lasheras B, de la Torre R, Aguirre N (2008b) On the role of tyrosine and peripheral metabolism in 3,4-methylenedioxymethamphetamine-induced serotonin neurotoxicity in rats. Neuropharmacology 54(5):885–900. https://doi.org/10.1016/j.neuropharm.2008.01.007
Greene SL, Dargan PI, O’Connor N, Jones AL, Kerins M (2003) Multiple toxicity from 3,4-methylenedioxymethamphetamine (“ecstasy”). Am J Emerg Med 21(2):121–124. https://doi.org/10.1053/ajem.2003.50028
Hall AP, Henry JA (2006) Acute toxic effects of ‘Ecstasy’ (MDMA) and related compounds: overview of pathophysiology and clinical management. Br J Anaesth 96(6):678–685. https://doi.org/10.1093/bja/ael078
Henry JA, Jeffreys KJ, Dawling S (1992) Toxicity and deaths from 3,4-methylenedioxymethamphetamine (“ecstasy”). Lancet 340(8816):384–387. https://doi.org/10.1016/0140-6736(92)91469-O
Iqbal K, Liu F, Gong C-X, Alonso AdC, Grundke-Iqbal I (2009) Mechanisms of tau-induced neurodegeneration. Acta Neuropathol 118(1):53–69. https://doi.org/10.1007/s00401-009-0486-3
Karami M, Karimian Nokabadi F, Ebrahimzadeh MA, Naghshvar F (2014) Nephroprotective effects of Feijoa sellowiana leaves extract on renal injury induced by acute dose of ecstasy (MDMA) in mice. Iran J Basic Med Sci 17(1):69–72
Kelly PA, Ritchie IM, Quate L, McBean DE, Olverman HJ (2002) Functional consequences of perinatal exposure to 3,4-methylenedioxymethamphetamine in rat brain. Br J Pharmacol 137(7):963–970. https://doi.org/10.1038/sj.bjp.0704961
Kindlundh-Hogberg AM, Schioth HB, Svenningsson P (2007) Repeated intermittent MDMA binges reduce DAT density in mice and SERT density in rats in reward regions of the adolescent brain. Neurotoxicology 28(6):1158–1169. https://doi.org/10.1016/j.neuro.2007.07.002
Klomp A, den Hollander B, de Bruin K, Booij J, Reneman L (2012) The effects of ecstasy (MDMA) on brain serotonin transporters are dependent on age-of-first exposure in recreational users and animals. PLoS One 7(10):e47524. https://doi.org/10.1371/journal.pone.0047524
Kolyaduke OV, Hughes RN (2013) Increased anxiety-related behavior in male and female adult rats following early and late adolescent exposure to 3,4-methylenedioxymethamphetamine (MDMA). Pharmacol Biochem Behav 103(4):742–749. https://doi.org/10.1016/j.pbb.2012.12.004
Kregel KC (1994) Influence of aging on tissue-specific noradrenergic activity at rest and during nonexertional heating in rats. J Appl Physiol 76(3):1226–1231. https://doi.org/10.1152/jappl.1994.76.3.1226
Loureiro-Vieira S, Costa VM, Duarte JA, Duarte-Araújo M, Gonçalves-Monteiro S, Bastos ML, Carvalho F, Capela JP (2018) Methylphenidate clinically oral doses improved brain and heart glutathione redox status and evoked renal and cardiac tissue injury in rats. Biomed Pharmacother 100:551–563. https://doi.org/10.1016/j.biopha.2018.02.017
Malberg JE, Seiden LS (1998) Small changes in ambient temperature cause large changes in 3,4-methylenedioxymethamphetamine (MDMA)-induced serotonin neurotoxicity and core body temperature in the rat. J Neurosci 18(13):5086–5094
Malberg JE, Sabol KE, Seiden LS (1996) Co-administration of MDMA with drugs that protect against MDMA neurotoxicity produces different effects on body temperature in the rat. J Pharmacol Exp Ther 278(1):258–267
Mills EM, Rusyniak DE, Sprague JE (2004) The role of the sympathetic nervous system and uncoupling proteins in the thermogenesis induced by 3,4-methylenedioxymethamphetamine. J Mol Med (Berl) 82(12):787–799. https://doi.org/10.1007/s00109-004-0591-7
Navarro-Zaragoza J, Ros-Simo C, Milanes MV, Valverde O, Laorden ML (2015) Binge ethanol and MDMA combination exacerbates toxic cardiac effects by inducing cellular stress. PLoS One 10(10):e0141502. https://doi.org/10.1371/journal.pone.0141502
Nishimura A, Ueda S, Takeuchi Y, Sawada T, Kawata M (1995) Age-related decrease of serotonergic fibres and S-100[beta] immunoreactivity in the rat dentate gyrus. Neuro Rep 6(10):1445–1448
Pereira FC, Gough B, Macedo TR, Ribeiro CF, Ali SF, Binienda ZK (2011) Buprenorphine modulates methamphetamine-induced dopamine dynamics in the rat caudate nucleus. Neurotox Res 19(1):94–101. https://doi.org/10.1007/s12640-009-9143-9
Reveron ME, Monks TJ, Duvauchelle CL (2005) Age-dependent (+)MDMA-mediated neurotoxicity in mice. Neurotoxicology 26(6):1031–1040. https://doi.org/10.1016/j.neuro.2005.05.006
Shenouda SK, Lord KC, McIlwain E, Lucchesi PA, Varner KJ (2008) Ecstasy produces left ventricular dysfunction and oxidative stress in rats. Cardiovasc Res 79(4):662–670. https://doi.org/10.1093/cvr/cvn129
Shintani-ishida K, Saka K, Yamaguchi K, Hayashida M, Nagai H, Takemura G, Yoshida K (2014) MDMA induces cardiac contractile dysfunction through autophagy upregulation and lysosome destabilization in rats. Biochim Biophys Acta 1842(5):691–700. https://doi.org/10.1016/j.bbadis.2014.01.013
Slotkin TA, Seidler FJ, Ali SF (2000) Cellular determinants of reduced adaptability of the aging brain: neurotransmitter utilization and cell signaling responses after MDMA lesions. Brain Res 879(1):163–173. https://doi.org/10.1016/S0006-8993(00)02767-0
Szabo G, Csak T (2012) Inflammasomes in liver diseases. J Hepatol 57(3):642–654. https://doi.org/10.1016/j.jhep.2012.03.035
Teixeira-Gomes A, Costa VM, Feio-Azevedo R, Bastos ML, Carvalho F, Capela JP (2015) The neurotoxicity of amphetamines during the adolescent period. Int J Dev Neurosci 41:44–62. https://doi.org/10.1016/j.ijdevneu.2014.12.001
Teixeira-Gomes A, Costa VM, Feio-Azevedo R, Duarte JA, Duarte-Araújo M, Fernandes E, Bastos ML, Carvalho F, Capela JP (2016) “Ecstasy” toxicity to adolescent rats following an acute low binge dose. BMC Pharmacol Toxicol 17(1):1–14. https://doi.org/10.1186/s40360-016-0070-0
UNODC (2017) World Drug Report 2017. United Nations, Vienna
van Luijtelaar MGPA, Steinbusch HWM, Tonnaer JADM (1988) Aberrant morphology of serotonergic fibers in the forebrain of the aged rat. Neurosci Lett 95(1–3):93–96. https://doi.org/10.1016/0304-3940(88)90638-6
Vlad M, Ionescu N, Ispas AT, Giuvarasteanu I, Ungureanu E, Stoica C (2010) Morphological changes during acute experimental short-term hyperthermia. Rom J Morphol Embryol 51(4):739–744
Yin C, Pettigrew A, Loftus JP, Black SJ, Belknap JK (2009) Tissue concentrations of 4-HNE in the black walnut extract model of laminitis: indication of oxidant stress in affected laminae. Vet Immunol Immunopathol 129(3–4):211–215. https://doi.org/10.1016/j.vetimm.2008.11.016
Acknowledgements
We greatly acknowledge Dr. ª Bárbara Oliveira of ICBAS-UP animal house facility, and Dr.a Laura Pereira of FFUP biochemistry for their technical assistance. This work was supported by the Fundação para a Ciência e Tecnologia (FCT)—Project PTDC/SAU-FCF/102958/2008—QREN initiative with EU/FEDER financing through under the frame of ‘‘Programa Operacional Temático Factores de Competitividade (COMPETE) do Quadro Comunitário de Apoio III’’ and ‘‘Fundo Comunitário Europeu (FEDER)’’ (FCOMP-01-0124-FEDER-011079). It was also supported by FEDER funds through the COMPETE and by national funds by the Fundação para a Ciência e Tecnologia (FCT) within the Project “PTDC/DTP-FTO/1489/2014—POCI-01-0145-FEDER-016537”. This work also received financial support from the European Union (FEDER funds POCI/01/0145/FEDER/007728) and National Funds (FCT/MEC, Fundação para a Ciência e a Tecnologia and Ministério da Educação e Ciência) under the Partnership Agreement PT2020 UID/MULTI/04378/2013. Additionally, supported by project NORTE-01-0145-FEDER-000024, supported by Norte Portugal Regional Operational Programme (NORTE 2020), under the PORTUGAL 2020 Partnership Agreement (DESignBIOtecHealth—New Technologies for three Health Challenges of Modern Societies: Diabetes, Drug Abuse and Kidney Diseases), through the European Regional Development Fund (ERDF). VM Costa and DJ Barbosa acknowledge FCT for their post-doc (SFRH/BPD/110001/2015) and PhD grants (SFRH/BD/64939/2009), respectively.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Disclosure
Parts of this work have been presented orally at the Portuguese Pharmacology Society annual meeting, and in the form of an abstract and poster at the Congress of the European Societies of Toxicology (Eurotox). Some results included in this article were incorporated in Armanda Gomes Master thesis entitled “Brain and Peripheral Organ Toxicity of “Ecstasy” in Adolescent Rats in Human Relevant Doses” printed last September 2014. The thesis document is available online at the university of Porto repository: https://repositorio-aberto.up.pt/bitstream/10216/76843/2/32960.pdf.
Rights and permissions
About this article
Cite this article
Feio-Azevedo, R., Costa, V.M., Barbosa, D.J. et al. Aged rats are more vulnerable than adolescents to “ecstasy”-induced toxicity. Arch Toxicol 92, 2275–2295 (2018). https://doi.org/10.1007/s00204-018-2226-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-018-2226-8