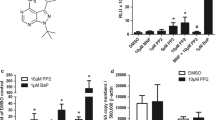
Abstract
The maintenance of cellular homeostasis is a complex process that is governed by the receipt of prototypical growth and death signals. The endogenous functions of aryl hydrocarbon receptor (AHR) in cellular homeostasis are not well understood. We aimed to establish whether the disturbance of endogenously activated AHR can influence cell growth, and if so, what mechanism(s) are involved. Cell growth was measured in mouse hepatoma Hepa-1 wild-type and cytochrome P4501A1 (CYP1A1)-deficient c37 cells. In other sets of experiments, HepG2 cells were exposed to different doses of FICZ (0.01nM–1 µM) alone or in combination with 50 nM of the CYP1A1 inhibitor 3′methoxy-4′nitro-flavone (MNF). CYP1A1 enzyme activity, cell viability, oxidative stress, and several endpoints of apoptosis were measured. FICZ treatment at a high concentration or in combination with MNF induced sustained CYP1A1 activity and led to oxidative stress and activation of apoptosis via a mitochondrial-dependent pathway. In comparison with the wild-type Hepa-1 cells, c37 cells lacking CYP1A1 activity proliferated faster in normal medium which contains trace levels of FICZ. Besides, in HepG2 cells, FICZ stimulated cell growth at low concentrations but inhibited cell growth at high concentrations. Based on these findings, we propose that CYP1A1 inhibitors, by increasing the levels of the endogenous ligand FICZ, change the cell growth kinetics and trigger cell death and apoptosis through a mitochondrial-dependent pathway. Since AHR controls multiple cellular functions, a wide range of toxicity can be expected by disturbing its endogenous functions.





Similar content being viewed by others
References
Abbott BD (1995) Review of the interaction between TCDD and glucocorticoids in embryonic palate. Toxicology 105(2–3):365–373
Abdelrahim M, Smith R 3rd, Safe S (2003) Aryl hydrocarbon receptor gene silencing with small inhibitory RNA differentially modulates Ah-responsiveness in MCF-7 and HepG2 cancer cells. Mol Pharmacol 63(6):1373–1381. doi:10.1124/mol.63.6
Anderson ME (1985) Determination of glutathione and glutathione disulfide in biological samples. Methods Enzymol 113:548–555
Bergander L, Wincent E, Rannug A, Foroozesh M, Alworth W, Rannug U (2004) Metabolic fate of the Ah receptor ligand 6-formylindolo[3,2-b]carbazole. Chem Biol Interact 149(2–3):151–164. doi:10.1016/j.cbi.2004.08.005
Calabrese EJ, Baldwin LA (2003) Inorganics and hormesis. Crit Rev Toxicol 33(3–4):215–304. doi:10.1080/713611040
Camacho IA, Singh N, Hegde VL, Nagarkatti M, Nagarkatti PS (2005) Treatment of mice with 2,3,7,8-tetrachlorodibenzo-p-dioxin leads to aryl hydrocarbon receptor-dependent nuclear translocation of NF-kappaB and expression of Fas ligand in thymic stromal cells and consequent apoptosis in T cells. J Immunol 175(1):90–103
Caruso JA, Mathieu PA, Joiakim A, Zhang H, Reiners JJ Jr (2006) Aryl hydrocarbon receptor modulation of tumor necrosis factor-alpha-induced apoptosis and lysosomal disruption in a hepatoma model that is caspase-8-independent. J Biol Chem 281(16):10954–10967. doi:10.1074/jbc.M508383200
Elferink CJ, Ge NL, Levine A (2001) Maximal aryl hydrocarbon receptor activity depends on an interaction with the retinoblastoma protein. Mol Pharmacol 59(4):664–673
Elizondo G, Fernandez-Salguero P, Sheikh MS et al (2000) Altered cell cycle control at the G(2)/M phases in aryl hydrocarbon receptor-null embryo fibroblast. Mol Pharmacol 57(5):1056–1063
Esser C, Rannug A (2015) The aryl hydrocarbon receptor in barrier organ physiology, immunology, and toxicology. Pharmacol Rev 67(2):259–279. doi:10.1124/pr.114.009001
Esser C, Rannug A, Stockinger B (2009) The aryl hydrocarbon receptor in immunity. Trends Immunol 30(9):447–454. doi:10.1016/j.it.2009.06.005
Ge NL, Elferink CJ (1998) A direct interaction between the aryl hydrocarbon receptor and retinoblastoma protein. Linking dioxin signaling to the cell cycle. J Biol Chem 273(35):22708–22713
Hankinson O, Andersen RD, Birren BW, Sander F, Negishi M, Nebert DW (1985) Mutations affecting the regulation of transcription of the cytochrome P1-450 gene in the mouse Hepa-1 cell line. J Biol Chem 260(3):1790–1795
Hushka DR, Greenlee WF (1995) 2,3,7,8-Tetrachlorodibenzo-p-dioxin inhibits DNA synthesis in rat primary hepatocytes. Mutat Res 333(1–2):89–99
Ito T, Tsukumo S, Suzuki N et al (2004) A constitutively active arylhydrocarbon receptor induces growth inhibition of jurkat T cells through changes in the expression of genes related to apoptosis and cell cycle arrest. J Biol Chem 279(24):25204–25210. doi:10.1074/jbc.M402143200
Jonsson ME, Mattsson A, Shaik S, Brunstrom B (2016) Toxicity and cytochrome P450 1 A mRNA induction by 6-formylindolo[3,2-b]carbazole (FICZ) in chicken and Japanese quail embryos. Comp Biochem Physiol C Toxicol Pharmacol 179:125–136. doi:10.1016/j.cbpc.2015.09.014
Jurgensmeier JM, Xie Z, Deveraux Q, Ellerby L, Bredesen D, Reed JC (1998) Bax directly induces release of cytochrome c from isolated mitochondria. Proc Natl Acad Sci USA 95(9):4997–5002
Kamath AB, Camacho I, Nagarkatti PS, Nagarkatti M (1999) Role of Fas-Fas ligand interactions in 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD)-induced immunotoxicity: increased resistance of thymocytes from Fas-deficient (lpr) and Fas ligand-defective (gld) mice to TCDD-induced toxicity. Toxicol Appl Pharmacol 160(2):141–155. doi:10.1006/taap.1999.8753
Kluck RM, Bossy-Wetzel E, Green DR, Newmeyer DD (1997) The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science 275(5303):1132–1136
Kolluri SK, Weiss C, Koff A, Gottlicher M (1999) p27(Kip1) induction and inhibition of proliferation by the intracellular Ah receptor in developing thymus and hepatoma cells. Genes Dev 13(13):1742–1753
Krammer PH (2000) CD95’s deadly mission in the immune system. Nature 407(6805):789–795. doi:10.1038/35037728
Kroemer G (1997) Mitochondrial implication in apoptosis. Towards an endosymbiont hypothesis of apoptosis evolution. Cell Death Differ 4(6):443–456. doi:10.1038/sj.cdd.4400266
Liu X, Kim CN, Yang J, Jemmerson R, Wang X (1996) Induction of apoptotic program in cell-free extracts: requirement for dATP and cytochrome c. Cell 86(1):147–157
Ma Q, Whitlock JP Jr (1996) The aromatic hydrocarbon receptor modulates the Hepa 1c1c7 cell cycle and differentiated state independently of dioxin. Mol Cell Biol 16(5):2144–2150
Marlowe JL, Puga A (2005) Aryl hydrocarbon receptor, cell cycle regulation, toxicity, and tumorigenesis. J Cell Biochem 96(6):1174–1184. doi:10.1002/jcb.20656
McConkey DJ, Hartzell P, Duddy SK, Hakansson H, Orrenius S (1988) 2,3,7,8-Tetrachlorodibenzo-p-dioxin kills immature thymocytes by Ca2+-mediated endonuclease activation. Science 242(4876):256–259
Mohammadi-Bardbori A, Rannug A (2014) Arsenic, cadmium, mercury and nickel stimulate cell growth via NADPH oxidase activation. Chem Biol Interact 224:183–188. doi:10.1016/j.cbi.2014.10.034
Mohammadi-Bardbori A, Bengtsson J, Rannug U, Rannug A, Wincent E (2012) Quercetin, resveratrol, and curcumin are indirect activators of the aryl hydrocarbon receptor (AHR). Chem Res Toxicol 25(9):1878–1884. doi:10.1021/tx300169e
Mohammadi-Bardbori A, Vikstrom Bergander L, Rannug U, Rannug A (2015) NADPH oxidase-dependent mechanism explains how arsenic and other oxidants can activate aryl hydrocarbon receptor signaling. Chem Res Toxicol 28(12):2278–2286. doi:10.1021/acs.chemrestox.5b00415
Mohammadi-Bardbori A, Akbarizadeh AR, Delju F, Rannug A (2016) Chromatin remodeling by curcumin alters endogenous aryl hydrocarbon receptor signaling. Chem Biol Interact 252:19–27. doi:10.1016/j.cbi.2016.03.037
Munzel P, Bock-Hennig B, Schieback S, Gschaidmeier H, Beck-Gschaidmeier S, Bock KW (1996) Growth modulation of hepatocytes and rat liver epithelial cells (WB-F344) by 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD). Carcinogenesis 17(2):197–202
Nebert DW (1990) Drug metabolism. Growth signal pathways. Nature 347(6295):709–710. doi:10.1038/347709a0
Nebert DW (1991) Proposed role of drug-metabolizing enzymes: regulation of steady state levels of the ligands that effect growth, homeostasis, differentiation, and neuroendocrine functions. Mol Endocrinol 5(9):1203–1214. doi:10.1210/mend-5-9-1203
Nebert DW, Roe AL, Dieter MZ, Solis WA, Yang Y, Dalton TP (2000) Role of the aromatic hydrocarbon receptor and [Ah] gene battery in the oxidative stress response, cell cycle control, and apoptosis. Biochem Pharmacol 59(1):65–85
Oberg M, Bergander L, Hakansson H, Rannug U, Rannug A (2005) Identification of the tryptophan photoproduct 6-formylindolo[3,2-b]carbazole, in cell culture medium, as a factor that controls the background aryl hydrocarbon receptor activity. Toxicol Sci 85(2):935–943. doi:10.1093/toxsci/kfi154
Pohjanvirta R (2011) The AH receptor in biology and toxicology. Wiley, Hoboken
Puga A, Barnes SJ, Chang C et al (2000a) Activation of transcription factors activator protein-1 and nuclear factor-kappaB by 2,3,7,8-tetrachlorodibenzo-p-dioxin. Biochem Pharmacol 59(8):997–1005
Puga A, Barnes SJ, Dalton TP, Chang C, Knudsen ES, Maier MA (2000b) Aromatic hydrocarbon receptor interaction with the retinoblastoma protein potentiates repression of E2F-dependent transcription and cell cycle arrest. J Biol Chem 275(4):2943–2950
Puga A, Xia Y, Elferink C (2002) Role of the aryl hydrocarbon receptor in cell cycle regulation. Chem Biol Interact 141(1–2):117–130 pii]
Rannug A, Rannug U, Rosenkranz HS et al (1987) Certain photooxidized derivatives of tryptophan bind with very high affinity to the Ah receptor and are likely to be endogenous signal substances. J Biol Chem 262(32):15422–15427
Rhile MJ, Nagarkatti M, Nagarkatti PS (1996) Role of Fas apoptosis and MHC genes in 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD)-induced immunotoxicity of T cells. Toxicology 110(1–3):153–167
Scarlett JL, Sheard PW, Hughes G, Ledgerwood EC, Ku HH, Murphy MP (2000) Changes in mitochondrial membrane potential during staurosporine-induced apoptosis in Jurkat cells. FEBS Lett 475(3):267–272
Schrenk D, Karger A, Lipp HP, Bock KW (1992) 2,3,7,8-Tetrachlorodibenzo-p-dioxin and ethinylestradiol as co-mitogens in cultured rat hepatocytes. Carcinogenesis 13(3):453–456
Sherr CJ (1996) Cancer cell cycles. Science 274(5293):1672–1677
Sherr CJ, Roberts JM (1999) CDK inhibitors: positive and negative regulators of G1-phase progression. Genes Dev 13(12):1501–1512
Silverstone AE, Frazier DE Jr, Gasiewicz TA (1994) Alternate immune system targets for TCDD: lymphocyte stem cells and extrathymic T-cell development. Exp Clin Immunogenet 11(2–3):94–101
Smirnova A, Wincent E, Vikstrom Bergander L et al (2016) Evidence for new light-independent pathways for generation of the endogenous aryl hydrocarbon receptor agonist FICZ. Chem Res Toxicol 29(1):75–86. doi:10.1021/acs.chemrestox.5b00416
Susin SA, Zamzami N, Castedo M et al (1996) Bcl-2 inhibits the mitochondrial release of an apoptogenic protease. J Exp Med 184(4):1331–1341
Wang H, Joseph JA (1999) Quantifying cellular oxidative stress by dichlorofluorescein assay using microplate reader. Free Radic Biol Med 27(5–6):612–616
Wang HC, Zhou Y, Huang SK (2016) SHP-2 phosphatase controls aryl hydrocarbon receptor-mediated ER stress response in mast cells. Arch Toxicol. doi:10.1007/s00204-016-1861-1
Weiss C, Kolluri SK, Kiefer F, Gottlicher M (1996) Complementation of Ah receptor deficiency in hepatoma cells: negative feedback regulation and cell cycle control by the Ah receptor. Exp Cell Res 226(1): 154–163. doi:10.1006/excr.1996.0214
Wincent E, Amini N, Luecke S et al (2009) The suggested physiologic aryl hydrocarbon receptor activator and cytochrome P4501 substrate 6-formylindolo[3,2-b]carbazole is present in humans. J Biol Chem 284(5):2690–2696. doi:10.1074/jbc.M808321200
Wincent E, Bengtsson J, Mohammadi Bardbori A et al (2012) Inhibition of cytochrome P4501-dependent clearance of the endogenous agonist FICZ as a mechanism for activation of the aryl hydrocarbon receptor. Proc Natl Acad Sci USA 109(12):4479–4484. doi:10.1073/pnas.1118467109
Wincent E, Kubota A, Timme-Laragy A, Jonsson ME, Hahn ME, Stegeman JJ (2016) Biological effects of 6-formylindolo[3,2-b]carbazole (FICZ) in vivo are enhanced by loss of CYP1A function in an Ahr2-dependent manner. Biochem Pharmacol. doi:10.1016/j.bcp.2016.04.012
Wu R, Zhang L, Hoagland MS, Swanson HI (2007) Lack of the aryl hydrocarbon receptor leads to impaired activation of AKT/protein kinase B and enhanced sensitivity to apoptosis induced via the intrinsic pathway. J Pharmacol Exp Ther 320(1):448–457. doi:10.1124/jpet.106.111773
Yang JC, Cortopassi GA (1998) Induction of the mitochondrial permeability transition causes release of the apoptogenic factor cytochrome c. Free Radic Biol Med 24(4):624–631
Yang J, Liu X, Bhalla K et al (1997) Prevention of apoptosis by Bcl-2: release of cytochrome c from mitochondria blocked. Science 275(5303):1129–1132
Zamzami N, Marchetti P, Castedo M et al (1995) Sequential reduction of mitochondrial transmembrane potential and generation of reactive oxygen species in early programmed cell death. J Exp Med 182(2):367–377
Zamzami N, Marchetti P, Castedo M et al (1996) Inhibitors of permeability transition interfere with the disruption of the mitochondrial transmembrane potential during apoptosis. FEBS Lett 384(1):53–57
Zeytun A, McKallip RJ, Fisher M, Camacho I, Nagarkatti M, Nagarkatti PS (2002) Analysis of 2,3,7,8-tetrachlorodibenzo-p-dioxin-induced gene expression profile in vivo using pathway-specific cDNA arrays. Toxicology 178(3):241–260
Zhou Y, Tung HY, Tsai YM et al (2013) Aryl hydrocarbon receptor controls murine mast cell homeostasis. Blood 121(16):3195–3204. doi:10.1182/blood-2012-08-453597
Acknowledgements
The authors of this manuscript wish to express their appreciation to Agneta Rannug at Karolinska Instiutet, Stockholm, Sweden, for scientific discussions and suggestions and to Shiraz University of Medical Sciences, Shiraz, Iran. This work was supported by the Research Grants from the Shiraz University of Medical Sciences, Iran, (Grant Numbers 93-01-36-8817; 93-01-36-8818).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Author of this manuscript have no competing financial, professional, or personal interests.
Rights and permissions
About this article
Cite this article
Mohammadi-Bardbori, A., Bastan, F. & Akbarizadeh, AR. The highly bioactive molecule and signal substance 6-formylindolo[3,2-b]carbazole (FICZ) plays bi-functional roles in cell growth and apoptosis in vitro. Arch Toxicol 91, 3365–3372 (2017). https://doi.org/10.1007/s00204-017-1950-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-017-1950-9