Abstract
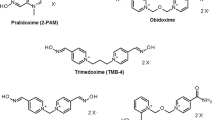
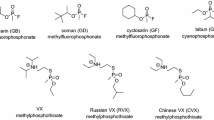
Oxime-type acetylcholinesterase reactivators (oxime-AChER) are used as an adjunct in the treatment for organophosphorus anticholinesterase poisoning. Because of the widespread usage and exposure of organophosphorus compounds (OPCs), its poisoning and fatalities is obvious in pregnant women, embryos and fetuses. OPCs irreversibly inhibit acetylcholinesterase (AChE) at nerve synapses. Furthermore, the role of AChE other than neurotransmission termination has been defined in the literature. The growing evidences show that cholinergic mechanisms are involved during growth and development of other organ systems. In contrary to the fact, the data on the use of oxime-AChER in OPC poisoning in pregnancy are scanty. The present review aimed to comprehend the status of oximes in pregnancy in lieu of the published literature. A thorough literature search was performed in January 2013, using ten popular search engines including Medline/PubMed, Google scholar, etc., using nine standard keywords. The search period was set from 1966 to present. The search did not reveal substantial data. No considerable studies were retrieved which could really demonstrate either the beneficial, harmful or even null effect of oxime-AChER usage in pregnancy. Only eighteen relevant articles were obtained for a period of about 47 years. In the literature, there is no report available to demonstrate the risk of using oxime-AChER in pregnancy for the treatment of OPC poisoning. The study reveals that the use of oxime-AChER in pregnancy is largely un-addressed, inconclusive and based on speculation albeit the incidences of OPC poisoning are quite prevalent. Well-designed studies are warranted for a tangible conclusion.



Similar content being viewed by others
References
Adhikari K, Ghosh A, Alauddin MD, Moitra A, Datta AK (2011) Organophosphate poisoning in pregnancy. J Obstet Gynaecol 31(4):290–292
Andersen RA, Barstad JA (1974) Passage of tertiary and quaternary nitrogen compounds through the rat placenta. Arch Int Pharmacodyn Ther 210(2):232–240
Antonijevic B, Stojiljkovic MP (2007) Unequal efficacy of pyridinium oximes in acute organophosphate poisoning. Clin Med Res 5:71–82
Arbuckle TE, Lin Z, Mery LS (2001) An exploratory analysis of the effect of pesticide exposure on the risk of spontaneous abortion in an Ontario farm population. Environ Health Perspect 109(8):851–857
Astroff AB, Young AD (1998) The relationship between maternal and fetal effects following maternal organophosphate exposure during gestation in the rat. Toxicol Ind Health 14(6):869–889
Bell JU, Van Petten GR, Taylor PJ, Aiken MJ (1979) The inhibition and reactivation of human maternal and fetal plasma cholinesterase following exposure to the organophosphate, dichlorvos. Life Sci 24(3):247–254
Bellinger DC (2013) Prenatal exposures to environmental chemicals and children’s neurodevelopment: an update. Saf Health Work 4:1–11
Berkowitz GS, Wetmur JG, Birman-Deych E, Obel J, Lapinski RH, Godbold JH, Holzman IR, Wolff MS (2004) In utero pesticide exposure, maternal paraoxonase activity, and head circumference. Environ Health Perspect 112(3):388–917
Berman T, Hochner-Celnikier D, Barr DB, Needham LL, Amitai Y, Wormser U, Richter E (2011) Pesticide exposure among pregnant women in Jerusalem, Israel: results of a pilot study. Environ Int 37(1):198–203
Bewan M (2009) Proposal for the inclusion of pralidoxime in the WHO model list of essential medicines. 17th Expert Committee on the Selection and Use of Essential Medicines,Geneva,http://www.who.int/selection_medicines/committees/expert/17/application/Pralidoxime_web.pdf (Last accessed on 16.03.2013)
Bigbee JW, Sharma KV, Chan EL, Bögler O (2000) Evidence for the direct role of acetylcholinesterase in neurite outgrowth in primary dorsal root ganglion neurons. Brain Res 861(2):354–362
Bouchard MF, Bellinger DC, Wright RO, Weisskopf MG (2010) Attention deficit/hyperactivity disorder and urinary metabolites of organophosphate pesticides. Pediatrics 125(6):e1270–e1277. doi:10.1542/peds.2009-3058
Bradman A, Eskenazi B, Barr DB, Bravo R, Castorina R, Chevrier J, Kogut K, Harnly ME, McKone TE (2005) Organophosphate urinary metabolite levels during pregnancy and after delivery in women living in an agricultural community. Environ Health Perspect 113(12):1802–1807
Buckley NA, Eddleston M, Li Y, Bevan M, Robertson J (2011) Oximes for acute organophosphate pesticide poisoning. Cochrane Database Syst Rev 2:CD005085. doi:10.1002/14651858.CD005085.pub2
Bytyqi AH, Lockridge O, Duysen E, Wang Y, Wolfrum U, Layer PG (2004) Impaired formation of the inner retina in an AChE knockout mouse results in degeneration of all photoreceptors. Eur J Neurosci 20(11):2953–2962
Campanharo F, Caetano A, Lopes C, Cavalcante R, Lopes M, Mattar R, Sun S (2009) Organophosphates and carbamates intoxication in pregnancy: a case-report. Int J Gynecol Obstet 107(Supplement 2):S502
Carlton FB, Simpson WM, Haddad LM (1998) The organophosphates and other insecticides. In: Haddad LM, Shannon MW, Winchester JF (eds) Clinical management of poisoning and drug overdose, 3rd edn. Saunders, Philadelphia, pp. 836–842
Cherian MA, Roshini C, Peter JV, Cherian AM (2005) Oximes in organophosphorus poisoning. Indian J Crit Care Med 9(3):155–163
Clinical: Atnaa (Atropine and Pralidoxime Chloride). http://wiki.medpedia.com/Clinical:Atnaa_(Atropine_and_Pralidoxime_Chloride) (Last accessed on 16.03.2013)
Carrington da Costa RB, Maul ER, Pimentel J, Gonçalves JS, Rebelo A, Oliveira LC, Rebelo A (1982) A case of acute poisoning by methyl demeton in a female 5 months pregnant. Arch Toxicol Suppl 5:202–204
Eddleston M, Buckley NA, Eyer P, Dawson AH (2008) Management of acute organophosphorus pesticide poisoning. Lancet 371:597–607
Edery H, Gila P, Zahavy J (1966) Passage of 2-hydroxyiminomethyl-N-methylpyridinium methanesulfonate to the fetus and cerebral spaces. Toxicol Appl Pharmacol 9(2):341–346
Eskenazi B, Harley K, Bradman A, Weltzien E, Jewell NP, Barr DB, Furlong CE, Holland NT (2004) Association of in utero organophosphate pesticide exposure and fetal growth and length of gestation in an agricultural population. Environ Health Perspect 112(10):1116–1124
Flaskos J (2012) The developmental neurotoxicity of organophosphorus insecticides: a direct role for the oxon metabolites. Toxicol Lett 209(1):86–93
Gadoth N, Fisher A (1978) Late onset of neuromuscular block in organophosphorus poisoning. Ann Intern Med 88(5):654–655
Gilboa-Geffen A, Hartmann G, Soreq H (2012) Stressing hematopoiesis and immunity: an acetylcholinesterase window into nervous and immune system interactions. Front Mol Neurosci 5:30. doi:10.3389/fnmol.2012.00030
Gunnell D, Eddleston M, Phillips MR, Konradsen F (2007) The global distribution of fatal pesticide self-poisoning: systematic review. BMC Public Health 7:357
Gupta RC (2007) Placental toxicity. In: Veterinary Toxicology, Basic and Clinical Principles. pp 245–262. doi:10.1016/B978-012370467-2/50112-7
Jajoo M, Saxena S, Pandey M (2010) Transplacentally acquired organophosphorus poisoning in a newborn: case report. Ann Trop Paediatr 30(2):137–139
Jones PEH, McCance RA (1949) Enzyme activities in the blood of infants and adults. Biochem J 45:464–467
Kalász H, Szöko E, Tábi T, Petroianu GA, Lorke DE, Omar A, Alafifi S, Jasem A, Tekes K (2009) Analysis of pralidoxime in serum, brain and CSF of rats. Med Chem 5(3):237–241
Kamha AA, Al Omary IY, Zalabany HA, Hanssens Y, Adheir FS (2005) Organophosphate poisoning in pregnancy: a case report. Basic Clin Pharmacol Toxicol 96(5):397–398
Karalliedde L, Senanayake N, Ariaratnam A (1988) Acute organophosphorus insecticide poisoning during pregnancy. Human Toxicology 7(4):363–364
Kavalci C, Durukan P, Ozer M, Cevik Y, Kavalci G (2009) Organophosphate poisoning due to a wheat bagel. Inter Med 48:85–88
Landauer W (1977) Cholinomimetic teratogens V. The effect of oximes and related cholinesterase reactivators. Teratology 15(1):33–42
Landgraf D, Barth M, Layer PG, Sperling LE (2010) Acetylcholine as a possible signaling molecule in embryonic stem cells: studies on survival, proliferation and death. Chem Biol Interact 187(1–3):115–119
Layer PG, Klaczinski J, Salfelder A, Sperling LE, Thangaraj G, Tuschl C, Vogel-Höpker A (2013) Cholinesterases in development: AChE as a firewall to inhibit cell proliferation and support differentiation. Chem Biol Interact 203(1):269–276
Lima CS, Dutra-Tavares AC, Nunes F, Nunes-Freitas AL, Ribeiro-Carvalho A, Filgueiras CC, Manhães AC, Meyer A, Abreu-Villaça Y (2013) Methamidophos exposure during the early postnatal period of mice: Immediate and late-emergent effects on the cholinergic and serotonergic systems and on behavior. Toxicol Sci [Epub ahead of print]
Lorke DE, Petroianu GA (2009) Minireview: does in vitro testing of oximes help predict their in vivo action after paraoxon exposure? J Appl Toxicol 29(6):459–469
Lorke DE, Kalasz H, Petroianu GA, Tekes K (2008) Entry of oximes into the brain: a review. Curr Med Chem 15(8):743–753
Marks AR, Harley K, Bradman A, Kogut K, Barr DB, Johnson C, Calderon N, Eskenazi B (2010) Organophosphate pesticide exposure and attention in young Mexican-American children: the CHAMACOS study. Environ Health Perspect 118(12):1768–1774
Meiniel R (1974) Protective action of pralidoxime against the teratogenic effects of parathion on the axial skeleton of the quail embryo. C R Acad Sci Hebd Seances Acad Sci D 279(7):603–606
Meiniel R (1975) Plurality in the determinism of organophosphorus teratogenic effects (author’s transl. Experientia 32(7):920–922
Meiniel R (1976) Prevention of abnormalities induced by 2 organophosphate insecticides (parathion and bidrin) in quail embryos. Arch Anat Morphol Exp 65(1):1–6
Meiniel R (1978) Pralidoxime prevents certain teratogenic effects induced by bidrin in quail embryos
Meiniel R (1979) On the plurifactorial determinism of the organophosphorus-induced teratogenesis on bird embryos; trials of protection by various compounds: oximes, hydroxamic acids and nicotinamide analogs. Arch Anat Histol Embryol 62:29–44
Meneguz A, Bisso GM, Michalek H (1989) Alterations in the distribution of cholinesterase molecular forms in maternal and fetal brain following diisopropyl fluorophosphate treatment of pregnant rats. Neurochem Res 14(3):285–291
Okumura T, Takasu N, Ishimatsu S, Miyanoki S, Mitsuhashi A, Kumada K, Tanaka K, Hinohara S (1996) Report on 640 victims of the Tokyo subway sarin attack. Ann Emerg Med 28:129–135
Olivera S, Rodriguez-Ithurralde D, Henley JM (2003) Acetylcholinesterase promotes neurite elongation, synapse formation, and surface expression of AMPA receptors in hippocampal neurones. Mol Cell Neurosci 23(1):96–106
Paraoanu LE, Layer PG (2008) Acetylcholinesterase in cell adhesion, neurite growth and network formation. FEBS J 275(4):618–624
Patocka J, Cabal J, Kuca K, Jun D (2005) Oxime reactivation of acetylcholinesterase inhibited by toxic phosphorus esters: in vitro kinetics and thermodynamics. J Appl Biomed 3:91–99
Peiris-John RJ, Wickremasinghe R (2008) Impact of low-level exposure to organophosphates on human reproduction and survival. Trans R Soc Trop Med Hyg 102(3):239–245
Peter JV, Moran JL, Graham P (2006) Oxime therapy and outcomes in human organophosphate poisoning: an evaluation using meta-analytic techniques. Crit Care Med 34:502–510
Petit C, Chevrier C, Durand G, Monfort C, Rouget F, Garlantezec R, Cordier S (2010) Impact on fetal growth of prenatal exposure to pesticides due to agricultural activities: a prospective cohort study in Brittany, France. Environ Health 9:71. doi:10.1186/1476-069X-9-71
Petroianu GA (2012) The history of cholinesterase reactivation: hydroxylamine and pyridinium aldoximes. Die Pharmazie 67(10):874–879
Pope CN (1999) Organophosphorus pesticides: do they all have same mechanism of toxicity? J Toxicol Environ Health B Crit Rev 2(2):161–181
FDA Approved Labeling Text dated September 8 (2010) PROTOPAM Chloride (pralidoxime chloride) for Injection. NDA 014134/S-022
Rauh VA, Perera FP, Horton MK, Whyatt RM, Bansal R, Hao X, Liu J, Barr DB, Slotkin TA, Peterson BS (2012) Brain anomalies in children exposed prenatally to a common organophosphate pesticide. Proc Natl Acad Sci USA 109(20):7871–7876
Solomon GM, Moodley J (2007) Acute chlorpyrifos poisoning in pregnancy: a case report. Clin Toxicol (Phila) 45(4):416–419
Soreq H, Seidman S (2001) Acetylcholinesterase—new roles for an old actor. Nat Rev Neurosci 2(4):294–302
Sperling LE, Klaczinski J, Schütz C, Rudolph L, Layer PG (2012) Mouse acetylcholinesterase enhances neurite outgrowth of rat R28 cells through interaction with laminin-1. PLoS ONE 7(5):e36683. doi:10.1371/journal.pone.0036683
Teixidó E, Piqué E, Gómez-Catalán J, Llobet JM (2013) Assessment of developmental delay in the zebrafish embryo teratogenicity assay. Toxicol In Vitro 27(1):469–478
Tenenbein M (1996) Acute poisonings in pregnancy. In: Descotes J. (ed) Human Toxicology. Elsevier, France, pp 247–257
van Helden HP, Busker RW, Melchers BP, Bruijnzeel PL (1996) Pharmacological effects of oximes: how relevant are they? Arch Toxicol 70(12):779–786
Vera B, Santa Cruz S, Magnarelli G (2012) Plasma cholinesterase and carboxylesterase activities and nuclear and mitochondrial lipid composition of human placenta associated with maternal exposure to pesticides. Reprod Toxicol 34(3):402–407
Vogel-Hopker A, Sperling LE, Layer PG (2012) Co-opting functions of cholinesterases in neural, limb and stem cell development. Protein Pept Lett 19(2):155–164
Wang P, Tian Y, Wang XJ, Gao Y, Shi R, Wang GQ, Hu GH, Shen XM (2012) Organophosphate pesticide exposure and perinatal outcomes in Shanghai, China. Environ Int 42:100–104
Weis OF, Müller FO, Lyell H, Badenhorst CH, van Niekerk P (1983) Materno-fetal cholinesterase inhibitor poisoning. Anesth Analg 62(2):233–235
Weitman SD, Vodicnik MJ, Lech JJ (1983) Influence of pregnancy on parathion toxicity and disposition. Toxicol Appl Pharmacol 71(2):215–224
Worek F, von der Wellen J, Musilek K, Kuca K, Thiermann H (2012) Reactivation kinetics of a homologous series of bispyridinium bis-oximes with nerve agent-inhibited human acetylcholinesterase. Arch Toxicol 86(9):1379–1386
Wyttenbach CR, Hwang JD (1984) Relationship between insecticide-induced short and wry neck and cervical defects visible histologically shortly after treatment of chick embryos. J Exp Zool 229(3):437–446
Zhang XJ, Yang L, Zhao Q, Caen JP, He HY, Jin QH, Guo LH, Alemany M, Zhang LY, Shi YF (2002) Induction of acetylcholinesterase expression during apoptosis in various cell types. Cell Death Differ 9(8):790–800
Conflict of interest
The authors declare that there is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nurulain, S.M., Kornelia, T., Naqvi, S.N.H. et al. Oxime-type acetylcholinesterase reactivators in pregnancy: an overview. Arch Toxicol 88, 575–584 (2014). https://doi.org/10.1007/s00204-013-1160-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-013-1160-z