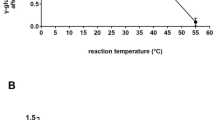
Abstract
Aspergillus nidulans exhibited high γ-glutamyl transpeptidase (γGT) activity in both carbon-starved and carbon-limited cultures. Glucose repressed, but casein peptone increased γGT production. Null mutation of creA did not influence γGT formation, but the functional meaB was necessary for the γGT induction. Deletion of the AN10444 gene (ggtA) completely eliminated the γGT activity, and the mRNA levels of ggtA showed strong correlation with the observed γGT activities. While ggtA does not contain a canonical signal sequence, the γGT activity was detectable both in the fermentation broth and in the hyphae. Deletion of the ggtA gene did not prevent the depletion of glutathione observed in carbon-starved and carbon-limited cultures. Addition of casein peptone to carbon-starved cultures lowered the formation of reactive species (RS). Deletion of ggtA could hinder this decrease and resulted in elevated RS formation. This effect of γGT on redox homeostasis may explain the reduced cleistothecia formation of ΔggtA strains in surface cultures.






Similar content being viewed by others
References
Adamis PD, Mannarino SC, Eleutherio EC (2009) Glutathione and gamma-glutamyl transferases are involved in the formation of cadmium-glutathione complex. FEBS Lett 583:1489–1492
Agblor AA, Josephy PD (2013) Donor substrate specificity of bovine kidney gamma-glutamyltransferase. Chem Biol Interact 203:480–485
Anderson ME (1985) Determination of glutathione and glutathione disulfide in biological samples. Methods Enzymol 113:548–555
Barratt RW, Johnson GB, Ogata WN (1965) Wild-type and mutant stocks of Aspergillus nidulans. Genetics 52:233–246
Bello MH, Epstein L (2013) Clades of γ-glutamyltransferases (GGTs) in the ascomycota and heterologous expression of Colletotrichum graminicola CgGGT1, a member of the pezizomycotina-only GGT clade. J Microbiol 51:88–99
Bello MH, Morin D, Epstein L (2013) γ-Glutamyltransferases (GGT) in Colletotrichum graminicola: mRNA and enzyme activity, and evidence that CgGGT1 allows glutathione utilization during nitrogen deficiency. Fungal Genet Biol 51:72–83
Bodnaryk RP, McGirr L (1973) Purification, properties and function of a unique gamma-glutamyl cyclotransferase from the housefly, Musca domestica. Biochim Biophys Acta 315:352–362
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 2:248–254
Chomczynski P (1993) A reagent for the single-step simultaneous isolation of RNA, DNA and proteins from cell and tissue samples. Biotechniques 15:532–537
Desai PR, Thakur A, Ganguli D, Paul S, Morschhäuser J, Bachhawat AK (2011) Glutathione utilization by Candida albicans requires a functional glutathione degradation (DUG) pathway and OPT7, an unusual member of the oligopeptide transporter family. J Biol Chem 286:41183–41194
Emri T, Pócsi I, Szentirmai A (1997) Glutathione metabolism and protection against oxidative stress caused by peroxides in Penicillium chrysogenum. Free Radical Biol Med 23:809–814
Emri T, Molnár Z, Pusztahelyi T, Pócsi I (2004) Physiological and morphological changes in autolysing Aspergillus nidulans cultures. Folia Microbiol 49:277–284
Emri T, Molnár Z, Veres T, Pusztahelyi T, Dudás G, Pócsi I (2006) Glucose mediated repression of autolysis and conidiogenesis in Emericella nidulans. Mycol Res 110:1172–1178
Emri T, Szilágyi M, László K, M-Hamvas M, Pócsi I (2009) PepJ is a new extracellular proteinase of Aspergillus nidulans. Folia Microbiol 54:105–109
Erdei E, Pusztahelyi T, Miskei M, Barna T, Pócsi I (2008) Characterization and heterologous expression of an age-dependent fungal/bacterial type chitinase of Aspergillus nidulans. Acta Microbiol Immunol Hung 55:351–361
Ganguli D, Kumar C, Bachhawat AK (2006) The alternative pathway of glutathione degradation is mediated by a novel protein complex involving three new genes in Saccharomyces cerevisiae. Genetics 175:1137–1151
Hagiwara D, Asano Y, Marui J, Furukawa K, Kanamaru K, Kato M, Abe K, Kobayashi T, Yamashino T, Mizuno T (2007) The SskA and SrrA response regulators are implicated in oxidative stress responses of hyphae and asexual spores in the phosphorelay signaling network of Aspergillus nidulans. Biosci Biotechnol Biochem 71:1003–1014
Halliwell B, Gutteridge JMC (2007) Measurement of reactive species. In: Halliwell B, Gutteridge JMC (eds) Free radicals in biology and medicine, 4th edn. Oxford University Press, Oxford, pp 268–330
Hara T, Yokoo Y, Furukawa T (1992) Potential of γ-l-glutamyl-l-cystine and bis-γ-l-glutamyl-l-cystine as a cystine-containing peptide for parental nutrition. In: Takai K (ed) Frontiers and new horizons in amino acid research. Elsevier, Amsterdam, pp 607–611
Heisterkamp N, Groffen J, Warburton D, Sneddon TP (2008) The human gamma-glutamyltransferase gene family. Hum Genet 123:321–332
Holdom MD, Hay RJ, Hamilton AJ (1996) The Cu, Zn superoxide dismutases of Aspergillus flavus, Aspergillus niger, Aspergillus nidulans, and Aspergillus terreus: purification and biochemical comparison with the Aspergillus fumigatus Cu, Zn superoxide dismutase. Infect Immun 64:3326–3332
Kang HJ, Kim BC, Park EH, Ahn K, Lim CJ (2005) The gene encoding gamma-glutamyl transpeptidase II in the fission yeast is regulated by oxidative and metabolic stress. J Biochem Mol Biol 38:609–618
Kaur H, Ganguli D, Bachhawat AK (2012) Glutathione degradation by the alternative pathway (DUG pathway) in Saccharomyces cerevisiae is initiated by (Dug2p-Dug3p)2 complex, a novel glutamine amidotransferase (GATase) enzyme acting on glutathione. J Biol Chem 287:8920–8931
Kawasaki L, Sánchez O, Shiozaki K, Aguirre J (2002) SakA MAP kinase is involved in stress signal transduction, sexual development and spore viability in Aspergillus nidulans. Mol Microbiol 45:1153–1163
Keserű JS, Szabó I, Gál Z, Massidda O, Mingoia M, Kaszanyitzky E, Jánosi S, Hulvely J, Csorba A, Buzás K, Hunyadi-Gulyás E, Medzihradszky KF, Biró S (2011) Identification of β-lactamases in human and bovine isolates of Staphylococcus aureus strains having borderline resistance to penicillinase-resistant penicillins (PRPs) with proteomic methods. Vet Microbiol 147:96–102
Kimura K, Tran LS, Uchida I, Itoh Y (2004) Characterization of Bacillus subtilis γ-glutamyltranspeptidase and its involvement in the degradation of capsule poly-gammaglutamate. Microbiol 150:4115–4123
Kovács Z, Szarka M, Kovács S, Boczonádi I, Emri T, Abe K, Pócsi I, Pusztahelyi T (2013) Effect of cell wall integrity stress and RlmA transcription factor on asexual development and autolysis in Aspergillus nidulans. Fungal Genet Biol 54:1–14
Krappmann S, Braus GH (2005) Nitrogen metabolism of Aspergillus and its role in pathogenicity. Med Mycol 43(Suppl 1):S31–S40
Kumar C, Sharma R, Bachhawat AK (2003) Utilization of glutathione as an exogenous sulfur source is independent of gamma-glutamyl transpeptidase in the yeast Saccharomyces cerevisiae: evidence for an alternative glutathione degradation pathway. FEMS Microbiol Lett 219:187–194
Kumar A, Tikoo S, Maity S, Sengupta S, Sengupta S, Kaur A, Bachhawat AK (2012) Mammalian proapoptotic factor ChaC1 and its homologues function as γ-glutamyl cyclotransferases acting specifically on glutathione. EMBO Rep 13:1095–1101
Kwon NJ, Garzia A, Espeso EA, Ugalde U, Yu JH (2010) FlbC is a putative nuclear C2H2 transcription factor regulating development in Aspergillus nidulans. Mol Microbiol 77:1203–1219
Li M, Liang X, Rollins JA (2012) Sclerotinia sclerotiorum γ-glutamyl transpeptidase (Ss-Ggt1) is required for regulating glutathione accumulation and development of sclerotia and compound appressoria. Mol Plant Microbe Interact 25:412–420. doi:10.1094/MPMI-06-11-0159
Margelis S, D’Souza C, Small AJ, Hynes MJ, Adams TH, Davis MA (2001) Role of glutamine synthetase in nitrogen metabolite repression in Aspergillus nidulans. J Bacteriol 183:5826–5833
Martins I, Garcia H, Varela A, Núñez O, Planchon S, Galceran MT, Renaut J, Rebelo LP, Silva Pereira C (2014) Investigating Aspergillus nidulans secretome during colonisation of cork cell walls. J Proteomics 98:175–188
Matsuyama A, Arai R, Yashiroda Y, Shirai A, Kamata A, Sekido S, Kobayashi Y, Hashimoto A, Hamamoto M, Hiraoka Y, Horinouchi S, Yoshida M (2006) ORFeome cloning and global analysis of protein localization in the fission yeast Schizosaccharomyces pombe. Nat Biotechnol 24:841–847
McCluskey K (2003) The fungal genetics stock center: from molds to molecules. Adv Appl Microbiol 52:245–262
Mehdi K, Penninckx MJ (1997) An important role for glutathione and γ-glutamyltranspeptidase in the supply of growth requirements during nitrogen starvation in the yeast Saccharomyces cerevisiae. Microbiology 143:885–1889
Molnár Z, Mészáros E, Szilágyi Z, Rosén S, Emri T, Pócsi I (2004) Influence of fadA G203R and ΔflbA mutations on the morphology and physiology of submerged Aspergillus nidulans cultures. Appl Biochem Biotech 118:349–360
Molnár Z, Emri T, Zavaczki E, Pusztehelyi T, Pócsi I (2006) Effects of mutations in the GanB/RgsA G protein mediated signaling on the autolysis of Aspergillus nidulans. J Basic Microbiol 46:495–503
Ohkama-Ohtsu N, Oikawa A, Zhao P, Xiang C, Saito K, Oliver DJ (2008) A gamma-glutamyl transpeptidase-independent pathway of glutathione catabolism to glutamate via 5-oxoproline in Arabidopsis. Plant Physiol 148:1603–1613
Orlowski M, Richman PG, Meister A (1969) Isolation and properties of gamma-L-glutamylcyclotransferase from human brain. Biochemistry 8:1048–1055
Osmani AH, May GS, Osmani SA (1999) The extremely conserved pyroA gene of Aspergillus nidulans is required for pyridoxine synthesis and is required indirectly for resistance to photosensitizers. J Biol Chem 274:23565–23569
Park HJ, Lim HW, Kim K, Kim IH, Park EH, Lim CJ (2004) Characterization and regulation of the γ-glutamyl transpeptidase gene from the fission yeast Schizosaccharomyces pombe. Can J Microbiol 50:61–66
Patananan AN, Palmer JM, Garvey GS, Keller NP, Clarke SG (2013) A novel automethylation reaction in the Aspergillus nidulans LaeA protein generates S-methylmethionine. J Biol Chem 288:14032–14045
Penninckx MJ, Elskens MT (1993) Metabolism and functions of glutathione in micro-organisms. Adv Microb Physiol 34:239–301
Petersen TN, Brunak S, von Heijne G, Nielsen H (2011) SignalP 4.0: discriminating signal peptides from transmembrane regions. Nat Methods 8:785–786
Pócsi I, Prade RA, Penninckx MJ (2004) Glutathione, altruistic metabolite in fungi. Adv Microb Physiol 49:1–76
Pusztahelyi T, Klement E, Szajli E, Klem J, Miskei M, Karányi Z, Emri T, Kovács S, Orosz G, Kovács KL, Medzihradszky KF, Prade RA, Pócsi I (2011) Comparison of transcriptional and translational changes caused by long-term menadione exposure in Aspergillus nidulans. Fungal Genet Biol 48:92–103
Sámi L, Emri T, Pócsi I (2001) Autolysis and aging of Penicillium chrysogenum cultures under carbon starvation: III: glutathione metabolism and formation of reactive oxygen species. Mycol Res 105:1246–1250
Saykhedkar S, Ray A, Ayoubi-Canaan P, Hartson SD, Prade R, Mort AJ (2012) A time course analysis of the extracellular proteome of Aspergillus nidulans growing on sorghum stover. Biotechnol Biofuels 5:52
Shibayama K, Wachino J, Arakawa Y, Saidijam M, Rutherford NG, Henderson PJ (2007) Metabolism of glutamine and glutathione via gamma-glutamyltranspeptidase and glutamate transport in Helicobacter pylori: possible significance in the pathophysiology of the organism. Mol Microbiol 64:396–406
Shroff RA, O’Connor SM, Hynes MJ, Lockington RA, Kelly JM (1997) Null alleles of creA, the regulator of carbon catabolite repression in Aspergillus nidulans. Fungal Genet Biol 22:28–38
Smirnova GV, Oktyabrsky ON (2005) Glutathione in bacteria. Biochemistry 70:1199–1211
Springael JY, Penninckx MJ (2003) Nitrogen-source regulation of yeast gamma-glutamyl transpeptidase synthesis involves the regulatory network including the GATA zinc-finger factors Gln3, Nil1/Gat1 and Gzf3. Biochem J 371:589–595
Suzuki H, Yamada C, Kato K (2007) Gamma-glutamyl compounds and their enzymatic production using bacterial gamma-glutamyltranspeptidase. Amino Acids 32:333–340
Szilágyi M, Pócsi I, Forgács K, Emri T (2010) MeaB dependent nutrition sensing regulates autolysis in carbon starving Aspergillus nidulans cultures. Indian J Microbiol 50:104–108
Szilágyi M, Anton F, Forgács K, Yu JH, Pócsi I, Emri T (2012) Antifungal activity of extracellular hydrolases produced by autolysing Aspergillus nidulans cultures. J Microbiol 50:849–854
Szilágyi M, Miskei M, Karányi Z, Lenkey B, Pócsi I, Emri T (2013) Transcriptome changes initiated by carbon starvation in Aspergillus nidulans. Microbiology 159:176–190
Thön M, Al-Abdallah Q, Hortschansky P, Brakhage AA (2007) The thioredoxin system of the filamentous fungus Aspergillus nidulans: impact on development and oxidative stress response. J Biol Chem 282:27259–27269
Todd RB, Fraser JA, Wong KH, Davis MA, Hynes MJ (2005) Nuclear accumulation of the GATA factor AreA in response to complete nitrogen starvation by regulation of nuclear export. Eukaryot Cell 4:1646–1653
Viña JR, Puertes IR, Montoro JB, Saez GT, Viña J (1985) Gamma-glutamyl-amino acids as signals for the hormonal regulation of amino acid uptake by the mammary gland of the lactating rat. Biol Neonate 48:250–256
Winderickx J, Holsbeeks I, Lagatie O, Giots F, Thevelein J, de Winde H (2003) From feast to famine; adaptation to nutrient availability in yeast. Top Curr Genet 1:305–386
Wong KH, Hynes MJ, Todd RB, Davis MA (2007) Transcriptional control of nmrA by the bZIP transcription factor MeaB reveals a new level of nitrogen regulation in Aspergillus nidulans. Mol Microbiol 66:534–551
Wünschmann J, Krajewski M, Letzel T, Huber EM, Ehrmann A, Grill E, Lendzian KJ (2010) Dissection of glutathione conjugate turnover in yeast. Phytochemistry 71:54–61
Yu JH, Hamari Z, Han KH, Seo JA, Reyes-Domínguez Y, Scazzocchio C (2004) Double-joint PCR: a PCR-based molecular tool for gene manipulations in filamentous fungi. Fungal Genet Biol 41:973–981
Zarnowski R, Cooper KG, Brunold LS, Calaycay J, Woods JP (2008) Histoplasma capsulatum secreted gamma-glutamyltransferase reduces iron by generating an efficient ferric reductant. Mol Microbiol 70:352–368
Zhang H, Forman HJ (2009) Redox regulation of gamma-glutamyl transpeptidase. Am J Respir Cell Mol Biol 41:509–515
Zhang H, Forman HJ, Choi J (2005) Gamma-glutamyl transpeptidase in glutathione biosynthesis. Methods Enzymol 401:468–483
Acknowledgments
This project was supported financially by the Hungarian Research Found OTKA-K100464 and TÁMOP-4.2.2.A-11/1/KONV-2012-0045 project, which is co-financed by the European Union and the European Social Fund. The work at UW-Madison was supported by the Intelligent Synthetic Biology Center of Global Frontier Project (2011-0031955) funded by the Ministry of Education, Science and Technology, Republic of Korea. Protein identification was carried out at the BMBI Proteomics Core Facility, University of Debrecen, Department of Biochemistry and Molecular Biology, and was supported in part by KMA 0149/3.0 Grants from the Research Fund Management and Research Exploitation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Erko Stackebrandt.
Rights and permissions
About this article
Cite this article
Spitzmüller, Z., Kwon, NJ., Szilágyi, M. et al. γ-Glutamyl transpeptidase (GgtA) of Aspergillus nidulans is not necessary for bulk degradation of glutathione. Arch Microbiol 197, 285–297 (2015). https://doi.org/10.1007/s00203-014-1057-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-014-1057-0