Abstract
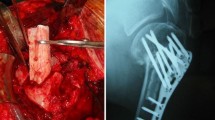
Avascular osteonecrosis (AVN) due to local ischemia leads to an inhomogeneous osseous defect, which can be treated by resection and with bone substitute materials in a joint-preserving treatment. Due to the underlying risk profile of AVN, the mostly subchondral localization and the size of the local bone defect, bone regeneration is impaired. Therefore, bioactivation of the applied bone substitute materials prior to application is highly desirable. Apart from the use of growth factors and other soluble substances, the autologous application of location-typical cells and tissue is a useful alternative to support the bone healing properties of scaffolds. This article presents various methods to activate scaffolds for bone stimulation and discusses these techniques with respect to recent data from the literature.
Zusammenfassung
Knochennekrosen entstehen durch eine ossäre Minderperfusion und führen zu einer inhomogenen ossären Defektzone, die im Fall einer gelenkerhaltenden Therapie entfernt und mit Knochenersatzstoffen behandelt werden kann. Aufgrund des zugrunde liegenden Risikoprofils der AVN,, der meist subchondralen Lokalisation und der Defektgröße wird knöcherne Heilung beeinträchtigt. Daher ist eine Bioaktivierung der verwendeten Knochenersatzmaterialien erstrebenswert. Neben dem Einsatz von Wachstumsfaktoren und anderen löslichen Substanzen stellt die autologe Applikation von lokotypischen Zellen und Geweben eine weitere Möglichkeit dar, die Knochenheilungspotenz von Scaffolds zu erhöhen. In der vorliegenden Arbeit werden die verschiedenen Möglichkeiten der Osteostimulation vorgestellt und diese unter dem Hintergrund der wissenschaftlichen Literatur diskutiert.




Similar content being viewed by others
Abbreviations
- ARCO:
-
Association Research Circulation Osseous classification
- AVN:
-
Avascular osteonecrosis
- BMP:
-
Bone morphogenetic proteins
- CD40 :
-
Cluster of differentiation 40
- ECM:
-
Extracellular matrix
- EPR:
-
Endoplasmic reticulum
- EV:
-
Extracellular vesicles
- HA:
-
Hydroxyapatite
- IFU:
-
Instruction for use
- IGF:
-
Insulin-like growth factor
- IL:
-
Interleukin
- MFGE8:
-
Human Milk Fat Globule EGF Factor 8
- MMP:
-
Matrix-Metalloprotease
- MSC:
-
Mesenchymale Stromazelle
- MTT:
-
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- PCL:
-
Poly(ε-caprolactone)
- PDCD6IP:
-
Programmed Cell Death 6 Interacting Protein
- PDGF:
-
Platelet-derived growth factor
- PEGMC:
-
Poly(ethylene glycol) maleate citrate
- PLA:
-
Polylactide
- PLGA:
-
Poly(lactide-co-glycolide)
- PLGA-mPEG:
-
PLGA-methoxy poly(ethylene glycol)
- PMMA:
-
Polymethylmethacrylat
- PRF:
-
Platelet-rich fibrin
- PRP:
-
Platelet-rich plasma
- rhBMP‑2:
-
Recombinant human BMP
- SEM:
-
Scanning electron microscope
- TCP:
-
Tricalcium phosphate
- TGF:
-
Transforming growth factor
- TNF:
-
Tumor necrosis factor
- TSG101:
-
Tumor Susceptibility Gene 101
- VEGF:
-
Vascular endothelial growth factor
- XACB:
-
Xenogeneic antigen-extracted cancellous bone
References
Ando W, Sakai T, Fukushima W et al (2021) Japanese Orthopaedic Association 2019 Guidelines for osteonecrosis of the femoral head. J Orthop Sci 26:46–68
Andriolo L, Merli G, Tobar C et al (2018) Regenerative therapies increase survivorship of avascular necrosis of the femoral head: a systematic review and meta-analysis. Int Orthop 42:1689–1704
Busch A, Herten M, Haversath M et al (2020) Ceramic scaffolds in a vacuum suction handle for Intraoperative stromal cell enrichment. Int J Mol Sci 21. https://doi.org/10.3390/ijms21176393
Chen WH, Kong XY, Wan R et al (2012) Effects of huogu I formula (I) on correlated factors of bone regeneration in chickens with steroid-induced necrosis of femoral head. Chin J Integr Med 18:378–384
Egle K, Salma I, Dubnika A (2021) From blood to regenerative tissue: how autologous platelet-rich fibrin can be combined with other materials to ensure controlled drug and growth factor release. Int J Mol Sci 22(21):11553. https://doi.org/10.3390/ijms222111553
Elgaz S, Bonig H, Bader P (2020) Mesenchymal stromal cells for osteonecrosis. J Transl Med 18:399
Feddahi N, Herten M, Tassemeier T et al (2021) Does needle design affect the regenerative potential of Bone marrow aspirate? An in vitro study. Life 11. https://doi.org/10.3390/life11080748
Han J, Gao F, Li Y et al (2020) The use of platelet-rich plasma for the treatment of osteonecrosis of the femoral head: a systematic review. Biomed Res Int 2020:2642439
Henze K, Herten M, Haversath M et al (2019) Surgical vacuum filter-derived stromal cells are superior in proliferation to human bone marrow aspirate. Stem Cell Res Ther 10:338
Jager M, Jennissen HP, Haversath M et al (2019) Intrasurgical protein layer on titanium arthroplasty explants: from the big twelve to the implant proteome. Proteomics Clin Appl 13:e1800168
Jäger M, Latosinska A, Herten M et al (2022) The implant proteome—the right surgical glue to fix titanium implants in situ. J Funct Biomater 13(2):44. https://doi.org/10.3390/jfb13020044
Jäger MS, Haversath M, Busch A (2022) Surgical-site released tissue for bone regeneration? The BoneFlo concept. In: 23rd EFORT Lisbon
Kang M, Lee CS, Lee M (2021) Bioactive scaffolds integrated with liposomal or extracellular vesicles for bone regeneration. Bioengineering 88(10):137. https://doi.org/10.3390/bioengineering8100137
Landgraeber S, Jäger M (2020) Modified advanced core decompression (mACD). Operat Orthop Traumatol 32:96–106
Li GQ, Wang ZY (2019) MiR-20b promotes osteocyte apoptosis in rats with steroid-induced necrosis of the femoral head through BMP signaling pathway. Eur Rev Med Pharmacol Sci 23:4599–4608
Lieberman JR, Conduah A, Urist MR (2004) Treatment of osteonecrosis of the femoral head with core decompression and human bone morphogenetic protein. Clin Orthop Relat Res: 139–145. https://doi.org/10.1097/01.blo.0000150312.53937.6f
Martinot P, Dartus J, Leclerc JT et al (2020) Hip survival after plain core decompression alone versus bone morphogenetic protein and/or bone marrow reinjection with core decompression for avascular osteonecrosis of the femoral head: a retrospective case control study in ninety two patients. Int Orthop 44:2275–2282
Poeran J, Opperer M, Rasul R et al (2016) Change in off-label use of bone morphogenetic protein in spine surgery and associations with adverse outcome. Global Spine J 6:650–659
Roth A, Beckmann J, Bohndorf K et al (2018) Update of the German S3 guideline on atraumatic femoral head necrosis in adults. Orthopade 47:757–769
Samara S, Dailiana Z, Varitimidis S et al (2013) Bone morphogenetic proteins (BMPs) expression in the femoral heads of patients with avascular necrosis. Mol Biol Rep 40:4465–4472
Serong S, Haversath M, Jäger M et al (2019) Prevalence of CAM deformity and its influence on therapy success in patients with osteonecrosis of the femoral head. J Tissue Eng Regen Med 13:546–554
Sun W, Li Z, Gao F et al (2014) Recombinant human bone morphogenetic protein‑2 in debridement and impacted bone graft for the treatment of femoral head osteonecrosis. Plos One 9:e100424
Tran TN, Wolf M, Winter P et al (2022) Hip joint mechanics in patients with osteonecrosis of the femoral head following treatment by advanced core decompression. Clin Biomech 94:105635
Wang ZL, He RZ, Tu B et al (2018) Drilling combined with adipose-derived stem cells and bone morphogenetic protein‑2 to treat femoral head epiphyseal necrosis in juvenile rabbits. Curr Med Sci 38:277–288
Wu J, Piao Y, Liu Q et al (2021) Platelet-rich plasma-derived extracellular vesicles: a superior alternative in regenerative medicine? Cell Prolif 54:e13123
Yan HC, Yu TT, Li J et al (2020) The delivery of extracellular vesicles loaded in biomaterial scaffolds for bone regeneration. Front Bioeng Biotechnol 8:1015
Zhang XL, Wang YM, Chu K et al (2018) The application of PRP combined with TCP in repairing avascular necrosis of the femoral head after femoral neck fracture in rabbit. Eur Rev Med Pharmacol Sci 22:903–909
Zhu T, Cui Y, Zhang M et al (2020) Engineered three-dimensional scaffolds for enhanced bone regeneration in osteonecrosis. Bioact Mater 5:584–601
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
M. Jäger, A. Busch and A. Sowislok declare that they have no competing interests.
For this article no studies with human participants or animals were performed by any of the authors. All studies mentioned were in accordance with the ethical standards indicated in each case.
Additional information

Scan QR code & read article online
Rights and permissions
About this article
Cite this article
Jäger, M., Busch, A. & Sowislok, A. Bioactivation of scaffolds in osteonecrosis. Orthopädie 51, 808–814 (2022). https://doi.org/10.1007/s00132-022-04303-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00132-022-04303-z