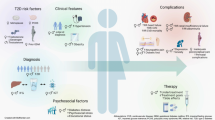
Abstract
Sexual dimorphism greatly influences adipose tissue remodeling, which is characterized by changes in the activity, number, and/or size of adipocytes in response to distinct stimuli, including lifestyle and anti-obesity drugs. This sex dependence seems to be due to the anatomical and endocrine disparities between men and women. At the molecular level, sex hormones are believed to mediate such differences and involve estrogen and androgen receptor-induced gene expression. The signaling pathways that regulate adipose tissue metabolism and function include peroxisome proliferator-activated receptor gamma, uncoupling protein 1 (UCP1), 5’ adenosine monophosphate-activated protein kinase (AMPK), and mitochondrial oxidative phosphorylation (OXPHOS), among other molecular players. Sex hormone-related pathways also interplay with adrenergic signaling, probably the most well-characterized molecular mechanism implicated in the remodeling of white adipose tissue. This review overviews and integrates the signaling pathways behind sexual dimorphism in adipose tissue remodeling, hoping to increase the knowledge on the pathogenesis of diseases, such as obesity and related comorbidities, and consequently, to drive future studies to investigate the regulation of this tissue homeostasis, either in men or women.


Similar content being viewed by others
References
Fitzgerald SJ, Janorkar AV, Barnes A, Maranon RO (2018) A new approach to study the sex differences in adipose tissue. J Biomed Sci 25. https://doi.org/10.1186/s12929-018-0488-3
Bartelt A, Heeren J (2014) Adipose tissue browning and metabolic health. Nat Rev Endocrinol 10:24–36
Choe SS, Huh JY, Hwang IJ, Kim JI, Kim JB (2016) Adipose tissue remodeling: its role in energy metabolism and metabolic disorders. Front Endocrinol (Lausanne) 7. https://doi.org/10.3389/fendo.2016.00030
Chang E, Varghese M, Singer K (2018) Gender and sex differences in adipose tissue. Curr Diabetes Rep 18:69
Kim S-N, Jung Y-S, Kwon H-J, Seong JK, Granneman JG, Lee Y-H (2016) Sex differences in sympathetic innervation and browning of white adipose tissue of mice. Biol Sex Differ 7:1–13
ter Horst KW, Gilijamse PW, de Weijer BA, Kilicarslan M, Ackermans MT, Nederveen AJ, Nieuwdorp M, Romijn JA, Serlie MJ (2015) Sexual dimorphism in hepatic, adipose tissue, and peripheral tissue insulin sensitivity in obese humans. Front Endocrinol (Lausanne) 6. https://doi.org/10.3389/fendo.2015.00182
Benz V, Bloch M, Wardat S, Böhm C, Maurer L, Mahmoodzadeh S, Wiedmer P, Spranger J, Foryst-Ludwig A, Kintscher U (2012) Sexual dimorphic regulation of body weight dynamics and adipose tissue lipolysis. PLoS One 7:e37794
Davis SM, Kaar JL, Ringham BM, Hockett CW, Glueck DH, Dabelea D (2019) Sex differences in infant body composition emerge in the first 5 months of life. J Pediatr Endocrinol Metab. https://doi.org/10.1515/jpem-2019-0243
Zore T, Palafox M, Reue K (2018) Sex differences in obesity, lipid metabolism, and inflammation-a role for the sex chromosomes? Mol Metab 15:35–44
Azrad M, Gower BA, Hunter GR, Nagy TR (2013) Racial differences in adiponectin and leptin in healthy premenopausal women. Endocrine 43:586–592
Brancati FL, Kao WHL, Folsom AR, Watson RL, Szklo M (2000) Incident type 2 diabetes mellitus in African American and white adults: the atherosclerosis risk in communities study. J Am Med Assoc 283:2253–2259
Shungin D, Winkler TW, Croteau-Chonka DC, Ferreira T, Locke AE, Mägi R, Strawbridge RJ, Pers TH, Fischer K, Justice AE et al (2015) New genetic loci link adipose and insulin biology to body fat distribution. Nature 518:187–196
Conte M, Martucci M, Sandri M, Franceschi C, Salvioli S (2019) The dual role of the pervasive “fattish” tissue remodeling with age. Front Endocrinol (Lausanne) 10:114
Bonaccorsi G, Trentini A, Greco P, Tisato V, Gemmati D, Bianchi N, Giganti M, Rossini M, Guglielmi G, Cervellati C (2019) Changes in adipose tissue distribution and association between uric acid and bone health during menopause transition. Int J Mol Sci 20:6321
Regitz-Zagrosek V (2012) Sex and gender differences in health. EMBO Rep 13:596–603
Torgrimson BN, Minson CT (2005) Sex and gender: what is the difference? J Appl Physiol 99:785–787
Heindel JJ, Schug TT (2013) The perfect storm for obesity. Obesity (Silver Spring) 21:1079–1080
Rush EC, Yan MR (2017) Evolution not revolution: nutrition and obesity. Nutrients 9:519
Hruby A, Hu FB (2015) The epidemiology of obesity: a big picture. Pharmacoeconomics 33:673–689
Mauvais-Jarvis F, Arnold AP, Reue K (2017) A guide for the design of pre-clinical studies on sex differences in metabolism. Cell Metab 25:1216–1230
Kanter R, Caballero B (2012) Global gender disparities in obesity: a review. Adv Nutr 3:491–498
Fox A, Feng W, Asal V (2019) What is driving global obesity trends? Globalization or “modernization”? Glob Health 15:32
Pineda E, Sanchez-Romero LM, Brown M, Jaccard A, Jewell J, Galea G, Webber L, Breda J (2018) Forecasting future trends in obesity across Europe: the value of improving surveillance. Obes Facts 11:360–371
Malenfant JH, Batsis JA (2019) Obesity in the geriatric population–a global health perspective. J Glob Health Rep 3:e2019045
Ng M, Fleming T, Robinson M, Thomson B, Graetz N, Margono C, Mullany EC, Biryukov S, Abbafati C, Abera SF et al (2014) Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet 384:766–781
Garawi F, Devries K, Thorogood N, Uauy R (2014) Global differences between women and men in the prevalence of obesity: is there an association with gender inequality? Eur J Clin Nutr 68:1101–1106
Moonen MPB, Nascimento EBM, van Marken Lichtenbelt WD (2019) Human brown adipose tissue: underestimated target in metabolic disease? Biochim Biophys Acta Mol Cell Biol Lipids 1864:104–112
Kwok KHM, Lam KSL, Xu A (2016) Heterogeneity of white adipose tissue: molecular basis and clinical implications. Exp Mol Med 48:e215
Demerath EW, Sun SS, Rogers N, Lee M, Reed D, Choh AC, Couch W, Czerwinski SA, Chumlea WC, Siervogel RM et al (2007) Anatomical patterning of visceral adipose tissue: race, sex, and age variation. Obesity (Silver Spring) 15:2984–2993
Ibrahim MM (2010) Subcutaneous and visceral adipose tissue: structural and functional differences. Obes Rev 11:11–18
Shadis S, Koutsari C, Jensen MD (2007) Direct free fatty acid uptake into human adipocytes in vivo. Diabetes 56:1369–1375
Guglielmi V, Sbraccia P (2018) Obesity phenotypes: depot-differences in adipose tissue and their clinical implications. Eat Weight Disord 23:3–14
Trémollieres FA, Pouilles J-M, Ribot CA (1996) Relative influence of age and menopause on total and regional body composition changes in postmenopausal women. Am J Obstet Gynecol 175:1594–1600
Tchernof A, Després JP, Dupont A, Belanger A, Nadeau A, Prud’homme D, Moorjani S, Lupien PL, Labrie F (1995) Relation of steroid hormones to glucose tolerance and plasma insulin levels in men: importance of visceral adipose tissue. Diabetes Care 18:292–299
Link JC, Reue K (2017) The genetic basis for sex differences in obesity and lipid metabolism. Annu Rev Nutr 37:225–245
Rodríguez AM, Monjo M, Roca P, Palou A (2002) Opposite actions of testosterone and progesterone on UCP1 mRNA expression in cultured brown adipocytes. Cell Mol Life Sci 59:1714–1723
Nedergaard J, Bengtsson T, Cannon B (2007) Unexpected evidence for active brown tissue in adult humans. Am J Physiol Endocrinol Metab 293:E444–E452
Hany TF, Gharehpapagh E, Kamel EM, Buck A, Himms-Hagen J, von Schulthess GK (2002) Brown adipose tissue: a factor to consider in symmetrical tracer uptake in the neck and upper chest region. Eur J Nucl Med 29:1393–1398
van Marken Lichtenbelt WD, Vanhommerig JW, Smulders NM, Drossaerts JMAFL, Kemerink GJ, Bouvy ND, Schrauwen P, Teule GJJ (2009) Cold-activated brown adipose tissue in healthy men. N Engl J Med 360:1500–1508
Cypess AM, Lehman S, Williams G, Tal I, Rodman D, Goldfine AB, Kuo FC, Palmer EL, Tseng Y-H, Doria A et al (2009) Identification and importance of BAT in adult humans. N Engl J Med 360:1509–1517
Rothwell NJ, Stock MJ (1982) Effect of chronic food restriction on energy balance, thermogenic capacity, and brown-adipose-tissue activity in the rat. Biosci Rep 2:543–549
Perkins AC, Mshelia DS, Symonds ME, Sathekge M (2013) Prevalence and pattern of brown adipose tissue distribution of 18F-FDG in patients undergoing PET-CT in a subtropical climatic zone. Nucl Med Commun 34:168–174
Law J, Bloor I, Budge H, Symonds ME (2014) The influence of sex steroids on adipose tissue growth and function. Horm Mol Biol Clin Invest 19:13–24
Pfannenberg C, Werner MK, Ripkens S, Stef I, Deckert A, Schmadl M, Reimold M, Häring H-U, Claussen CD, Stefan N (2010) Impact of age on the relationships of brown adipose tissue with sex and adiposity in humans. Diabetes 59:1789–1793
Kleinert M, Clemmensen C, Hofmann SM, Moore MC, Renner S, Woods SC, Huypens P, Beckers J, de Angelis MH, Schürmann A et al (2018) Animal models of obesity and diabetes mellitus. Nat Rev Endocrinol 14:140–162
Bryda EC (2013) The mighty mouse: the impact of rodents on advances in biomedical research. Mo Med 110:207–211
Chusyd DE, Wang D, Huffman DM, Nagy TR (2016) Relationships between rodent white adipose fat pads and human white adipose fat depots. Front Nutr 3:10
Amengual-Cladera E, Lladó I, Gianotti M, Proenza AM (2012) Sex differences in the effect of high-fat diet feeding on rat white adipose tissue mitochondrial function and insulin sensitivity. Metabolism 61:1108–1117
Rodríguez-Cuenca S, Pujol E, Justo R, Frontera M, Oliver J, Gianotti M, Roca P (2002) Sex-dependent thermogenesis, differences in mitochondrial morphology and function, and adrenergic response in brown adipose tissue. J Biol Chem 277:42958–42963
Justo R, Frontera M, Pujol E, Rodríguez-Cuenca S, Lladó I, García-Palmer FJ, Roca P, Gianotti M (2005) Gender-related differences in morphology and thermogenic capacity of brown adipose tissue mitochondrial subpopulations. Life Sci 76:1147–1158
López M, Alvarez CV, Nogueiras R, Diéguez C (2013) Energy balance regulation by thyroid hormones at central level. Trends Mol Med 19:418–427
Kooijman S, van den Heuvel JK, Rensen PCN (2015) Neuronal control of brown fat activity. Trends Endocrinol Metab 26:657–668
Kalicińska E, Wojtas K, Majda J, Zacharski M, Skiba J, Śliwowski J, Banasiak W, Ponikowski P, Jankowska EA (2018) Expression of sex steroid receptors and aromatase in adipose tissue in different body regions in men with coronary artery disease with and without ischemic systolic heart failure. Aging Male 7:1–13
Wang X, Xian T, Jia X, Zhang L, Liu L, Man F, Zhang X, Zhang J, Pan Q, Guo L (2017) A cross-sectional study on the associations of insulin resistance with sex hormone, abnormal lipid metabolism in T2DM and IGT patients. Medicine (Baltimore) 96:e7378
Simpson ER (2004) Aromatase: biologic relevance of tissue-specific expression. Semin Reprod Med 22:11–23
O’Reilly MW, House PJ, Tomlinson JW (2014) Understanding androgen action in adipose tissue. J Steroid Biochem Mol Biol 143:277–284
Stocco C (2012) Tissue physiology and pathology of aromatase. Steroids 77:27–35
Bélanger C, Hould F-S, Lebel S, Biron S, Brochu G, Tchernof A (2006) Omental and subcutaneous adipose tissue steroid levels in obese men. Steroids 71:674–682
Lee H-K, Lee JK, Cho B (2013) The role of androgen in the adipose tissue of males. World J Mens Health 31:136–140
Gao H, Dahlman-Wright K (2013) Implications of estrogen receptor alpha and estrogen receptor beta for adipose tissue functions and cardiometabolic complications. Horm Mol Biol Clin Invest 15:81–90
Arnal J-F, Lenfant F, Metivier R, Flouriot G, Henrion D, Adlanmerini M, Fontaine C, Gourdy P, Chambon P, Katzenellenbogen B et al (2017) Membrane and nuclear estrogen receptor alpha actions: from tissue specificity to medical implications. Physiol Rev 97:1045–1087
Gustafsson KL, Farman H, Henning P, Lionikaite V, Movérare-Skrtic S, Wu J, Ryberg H, Koskela A, Gustafsson J-Å, Tuukkanen J et al (2016) The role of membrane ERα signaling in bone and other major estrogen responsive tissues. Sci Rep 6:1–11
Amisten S, Neville M, Hawkes R, Persaud SJ, Karpe F, Salehi A (2015) An atlas of G-protein coupled receptor expression and function in human subcutaneous adipose tissue. Pharmacol Ther 146:61–93
Goedecke JH, Tootla M, Keswell D (2019) Ethnic differences in regional adipose tissue oestrogen receptor gene expression. Endocr Connect 8:32–38
Naaz A, Zakroczymski M, Heine P, Taylor J, Saunders P, Lubahn D, Cooke PS (2002) Effect of ovariectomy on adipose tissue of mice in the absence of estrogen receptor alpha (ERα): a potential role for estrogen receptor beta (ERβ). Horm Metab Res 34:758–763
González-Granillo M, Savva C, Li X, Fitch M, Pedrelli M, Hellerstein M, Parini P, Korach-André M, Gustafsson J-Å (2019) ERβ activation in obesity improves whole body metabolism via adipose tissue function and enhanced mitochondria biogenesis. Mol Cell Endocrinol 479:147–158
Brown EL, Hazen BC, Eury E, Wattez J-S, Gantner ML, Albert V, Chau S, Sanchez-Alavez M, Conti B, Kralli A (2018) Estrogen-related receptors mediate the adaptive response of brown adipose tissue to adrenergic stimulation. iScience 2:221–237
Moore JT, McKee DD, Slentz-Kesler K, Moore LB, Jones SA, Horne EL, Su J-L, Kliewer SA, Lehmann JM, Willson TM (1998) Cloning and characterization of human estrogen receptor β isoforms. Biochem Biophys Res Commun 247:75–78
Pedersen SB, Bruun JM, Hube F, Kristensen K, Hauner H, Richelsen B (2001) Demonstration of estrogen receptor subtypes α and β in human adipose tissue: influences of adipose cell differentiation and fat depot localization. Mol Cell Endocrinol 182:27–37
Leung Y-K, Mak P, Hassan S, Ho S-M (2006) Estrogen receptor (ER)-β isoforms: a key to understanding ER-β signaling. Proc Natl Acad Sci U S A 103:13162–13167
Rodriguez-Cuenca S, Monjo M, Frontera M, Gianotti M, Proenza AM, Roca P (2007) Sex steroid receptor expression profile in brown adipose tissue. Effects of hormonal status. Cell Physiol Biochem 20:877–886
Roncari DAK, Van RLR (1978) Promotion of human adipocyte precursor replication by 17β-estradiol in culture. J Clin Invest 62:503–508
Dieudonne MN, Pecquery R, Leneveu MC, Giudicelli Y (2000) Opposite effects of androgens and estrogens on adipogenesis in rat preadipocytes: evidence for sex and site-related specificities and possible involvement of insulin-like growth factor 1 receptor and peroxisome proliferator-activated receptor γ2. Endocrinology 141:649–656
Pedersen SB, Kristensen K, Hermann PA, Katzenellenbogen JA, Richelsen B (2004) Estrogen controls lipolysis by up-regulating α2A-adrenergic receptors directly in human adipose tissue through the estrogen receptor α. Implications for the female fat distribution. J Clin Endocrinol Metab 89:1869–1878
Palin SL, McTernan PG, Anderson LA, Sturdee DW, Barnett AH, Kumar S (2003) 17β-Estradiol and anti-estrogen ICI: compound 182,780 regulate expression of lipoprotein lipase and hormone-sensitive lipase in isolated subcutaneous abdominal adipocytes. Metabolism 52:383–388
Lundholm L, Zang H, Hirschberg AL, Gustafsson J-Å, Arner P, Dahlman-Wright K (2008) Key lipogenic gene expression can be decreased by estrogen in human adipose tissue. Fertil Steril 90:44–48
Pedersen SB, Bruun JM, Kristensen K, Richelsen B (2001) Regulation of UCP1, UCP2, and UCP3 mRNA expression in brown adipose tissue, white adipose tissue, and skeletal muscle in rats by estrogen. Biochem Biophys Res Commun 288:191–197
Nadal-Casellas A, Proenza AM, Lladó I, Gianotti M (2011) Effects of ovariectomy and 17-β estradiol replacement on rat brown adipose tissue mitochondrial function. Steroids 76:1051–1056
Babaei P, Mehdizadeh R, Ansar MM, Damirchi A (2010) Effects of ovariectomy and estrogen replacement therapy on visceral adipose tissue and serum adiponectin levels in rats. Menopause Int 16:100–104
Wohlers LM, Spangenburg EE (2010) 17β-estradiol supplementation attenuates ovariectomy-induced increases in ATGL signaling and reduced perilipin expression in visceral adipose tissue. J Cell Biochem 110:420–427
Kim J-Y, Jo K-J, Kim O-S, Kim B-J, Kang D-W, Lee K-H, Baik H-W, Han MS, Lee S-K (2010) Parenteral 17beta-estradiol decreases fasting blood glucose levels in non-obese mice with short-term ovariectomy. Life Sci 87:358–366
Greenberg AS, Shen W-J, Muliro K, Patel S, Souza SC, Roth RA, Kraemer FB (2001) Stimulation of lipolysis and hormone-sensitive lipase via the extracellular signal-regulated kinase pathway. J Biol Chem 276:45456–45461
Bartz R, Zehmer JK, Zhu M, Chen Y, Serrero G, Zhao Y, Liu P (2007) Dynamic activity of lipid droplets: protein phosphorylation and GTP-mediated protein translocation. J Proteome Res 6:3256–3265
Blouin K, Richard C, Brochu G, Hould F-S, Lebel S, Marceau S, Biron S, Luu-The V, Tchernof A (2006) Androgen inactivation and steroid-converting enzyme expression in abdominal adipose tissue in men. J Endocrinol 191:637–649
Blouin K, Veilleux A, Luu-The V, Tchernof A (2009) Androgen metabolism in adipose tissue: recent advances. Mol Cell Endocrinol 301:97–103
Fan W, Yanase T, Nomura M, Okabe T, Goto K, Sato T, Kawano H, Kato S, Nawata H (2005) Androgen receptor null male mice develop late-onset obesity caused by decreased energy expenditure and lipolytic activity but show normal insulin sensitivity with high adiponectin secretion. Diabetes 54:1000–1008
Bianchi VE, Locatelli V (2018) Testosterone a key factor in gender related metabolic syndrome. Obes Rev 19:557–575
Veilleux A, Blouin K, Tchernof A (2009) Mechanisms of androgenic action in adipose tissue. Clin Lipidol 4:367–378
Valencak TG, Osterrieder A, Schulz TJ (2017) Sex matters: the effects of biological sex on adipose tissue biology and energy metabolism. Redox Biol 12:806–813
Rodríguez-Cuenca S, Monjo M, Gianotti M, Proenza AM, Roca P (2007) Expression of mitochondrial biogenesis-signaling factors in brown adipocytes is influenced specifically by 17β-estradiol, testosterone, and progesterone. Am J Physiol Metab 292:E340–E346
Gnad T, Scheibler S, von Kügelgen I, Scheele C, Kilić A, Glöde A, Hoffmann LS, Reverte-Salisa L, Horn P, Mutlu S et al (2014) Adenosine activates brown adipose tissue and recruits beige adipocytes via A2Areceptors. Nature 516:395–399
Rodríguez E, Monjo M, Rodríguez-Cuenca S, Pujol E, Amengual B, Roca P, Palou A (2001) Sexual dimorphism in the adrenergic control of rat brown adipose tissue response to overfeeding. Pflugers Arch - Eur J Physiol 442:396–403
Monjo M, Rodríguez AM, Palou A, Roca P (2003) Direct effects of testosterone, 17β-estradiol, and progesterone on adrenergic regulation in cultured brown adipocytes: potential mechanism for gender-dependent thermogenesis. Endocrinology 144:4923–4930
Lisbôa PC, Curty FH, Moreira RM, Oliveira KJ, Pazos-Moura CC (2001) Sex steroids modulate rat anterior pituitary and liver iodothyronine deiodinase activities. Horm Metab Res 33:532–535
Bisschop PH, Toorians AW, Endert E, Wiersinga WM, Gooren LJ, Fliers E (2006) The effects of sex-steroid administration on the pituitary–thyroid axis in transsexuals. Eur J Endocrinol 155:11–16
Donda A, Reymond F, Lemarchand-Béraud T (1990) Sex steroids modulate the pituitary parameters involved in the regulation of TSH secretion in the rat. Acta Endocrinol 122:577–584
Coker RH, Williams RH, Yeo SE, Kortebein PM, Bodenner DL, Kern PA, Evans WJ (2009) The impact of exercise training compared to caloric restriction on hepatic and peripheral insulin resistance in obesity. J Clin Endocrinol Metab 94:4258–4266
Valle A, García-Palmer FJ, Oliver J, Roca P (2007) Sex differences in brown adipose tissue thermogenic features during caloric restriction. Cell Physiol Biochem 19:195–204
Zügel M, Qiu S, Laszlo R, Bosnyák E, Weigt C, Müller D, Diel P, Steinacker JM, Schumann U (2016) The role of sex, adiposity, and gonadectomy in the regulation of irisin secretion. Endocrine 54:101–110
Scalzo RL, Peltonen GL, Giordano GR, Binns SE, Klochak AL, Paris HLR, Schweder MM, Szallar SE, Wood LM, Larson DG et al (2014) Regulators of human white adipose browning: evidence for sympathetic control and sexual dimorphic responses to sprint interval training. PLoS One 9:e90696
Després JP, Bouchard C, Savard R, Tremblay A, Marcotte M, Thériault G (1984) The effect of a 20-week endurance training program on adipose-tissue morphology and lipolysis in men and women. Metabolism 33:235–239
Cataldi M, Muscogiuri G, Savastano S, Barrea L, Guida B, Taglialatela M, Colao A (2019) Gender-related issues in the pharmacology of new anti-obesity drugs. Obes Rev 20:375–384
Association AD (2019) 8. Obesity management for the treatment of type 2 diabetes: standards of medical care in diabetes-2019. Diabetes Care 42:S81–S89
Paccosi S, Cresci B, Pala L, Rotella CM, Parenti A (2019) Obesity therapy: how and why? Curr Med Chem. https://doi.org/10.2174/0929867326666190124121725
Datz FL, Christian PE, Moore J (1987) Gender-related differences in gastric emptying. J Nucl Med 28:1204–1207
Rubio MA (2014) Pharmacological treatment of obesity in Europe: waiting for the arrival of the white blackbird. Endocrinol Nutr 61:501–504
Srivastava G, Apovian C (2018) Future pharmacotherapy for obesity: new anti-obesity drugs on the horizon. Curr Obes Rep 7:147–161
Acknowledgments
This work was supported by the University of Aveiro and Portuguese Foundation for Science and Technology/Ministry of Science and Technology, FCT/MCT, for the financial support for the LAQV (FCT UIDB/50006/2020), CIAFEL (UIDB/00617/2020), iBiMED (UID/BIM/04501/2019), and UnIC (UID/IC/00051/2019) research units and the research projects RUNawayPCa (POCI-01-0145-FEDER-006958 and PTDC/DTP-DES/6077/2014) and NETDIAMOND (POCI-01-0145-FEDER-016385) and the post-graduation student (grant number SFRH/BD/144396/2019) to A.M.P. through national funds and co-financed by the European Regional Development Fund (ERDF), within the PT2020 Partnership Agreement.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Moreira-Pais, A., Ferreira, R., Neves, J.S. et al. Sex differences on adipose tissue remodeling: from molecular mechanisms to therapeutic interventions. J Mol Med 98, 483–493 (2020). https://doi.org/10.1007/s00109-020-01890-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-020-01890-2