Abstract
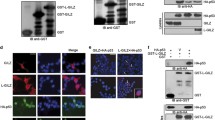
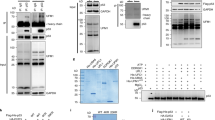
Serum and glucocorticoid regulated kinase 1 (Sgk1) is a serine–threonine kinase that is activated by serum, steroids, insulin, vasopressin, and interleukin 2 at the transcriptional and post-translational levels. Sgk1 is also important in transduction of growth factors and steroid-dependent survival signals and may have a role in the development of resistance to cancer chemotherapy. In the present paper, we demonstrate that Sgk1 activates MDM2-dependent p53 ubiquitylation. The results were obtained in RKO cells and other cell lines by Sgk1-specific RNA silencing and were corroborated in an original mouse model as well as in transiently and in stably transfected HeLa cells expressing wild-type or dominant negative Sgk1 mutant. Sgk1 contributes to cell survival, cell-cycle progression, and epithelial de-differentiation. We also show that the effects of Sgk1 on the clonogenic potential of different cancer cells depend on the expression of wild-type p53. Since transcription of Sgk1 is activated by p53, we propose a finely tuned feedback model where Sgk1 down-regulates the expression of p53 by enhancing its mono- and polyubiquitylation.









Similar content being viewed by others
References
Perrotti N, He RA, Phillips SA, Haft CR, Taylor SI (2001) Activation of serum- and glucocorticoid-induced protein kinase (Sgk) by cyclic AMP and insulin. J Biol Chem 276:9406–9412
Webster MK, Goya L, Ge Y, Maiyar AC, Firestone GL (1993) Characterization of sgk, a novel member of the serine/threonine protein kinase gene family which is transcriptionally induced by glucocorticoids and serum. Mol Cell Biol 13:2031–2040
Hong F, Larrea MD, Doughty C, Kwiatkowski DJ, Squillace R, Slingerland JM (2008) mTOR-Raptor binds and activates SGK1 to regulate p27 phosphorylation. Mol Cell 30:701–711
Faletti CJ, Perrotti N, Taylor SI, Blazer-Yost BL (2002) Sgk: an essential convergence point for peptide and steroid hormone regulation of ENaC-mediated Na+ transport. Am J Physiol Cell Physiol 282:C494–C500
Busjahn A, Aydin A, Uhlmann R, Krasko C, Bahring S, Szelestei T, Feng Y, Dahm S, Sharma AM, Luft FC, Lang F (2002) Serum- and glucocorticoid-regulated kinase (SGK1) gene and blood pressure. Hypertension 40:256–260
von Wowern F, Berglund G, Carlson J, Mansson H, Hedblad B, Melander O (2005) Genetic variance of SGK-1 is associated with blood pressure, blood pressure change over time and strength of the insulin-diastolic blood pressure relationship. Kidney Int 68:2164–2172
Huang DY, Boini KM, Osswald H, Friedrich B, Artunc F, Ullrich S, Rajamanickam J, Palmada M, Wulff P, Kuhl D, Vallon V, Lang F (2006) Resistance of mice lacking the serum- and glucocorticoid-inducible kinase SGK1 against salt-sensitive hypertension induced by a high-fat diet. Am J Physiol Renal Physiol 291:F1264–F1273
Mikosz CA, Brickley DR, Sharkey MS, Moran TW, Conzen SD (2001) Glucocorticoid receptor-mediated protection from apoptosis is associated with induction of the serine/threonine survival kinase gene, sgk-1. J Biol Chem 276:16649–16654
Lang F, Böhmer C, Palmada M, Seebohm G, Strutz-Seebohm N, Vallon V (2006) (Patho)physiological significance of the serum- and glucocorticoid-inducible kinase isoforms. Physiol Rev 286:1151–1178. Review
Amato R, Menniti M, Agosti V, Boito R, Costa N, Bond HM, Barbieri V, Tagliaferri P, Venuta S, Perrotti N (2007) IL-2 signals through Sgk1 and inhibits proliferation and apoptosis in kidney cancer cells. J Mol Med 85:707–721
Iwata S, Nomoto M, Morioka H, Miyata A (2004) Gene expression profiling in the midbrain of striatal 6-hydroxydopamine-injected mice. Synapse 51:279–286
Simon P, Schneck M, Hochstetter T, Koutsouki E, Mittelbronn M, Merseburger A, Weigert C, Niess A, Lang F (2007) Differential regulation of serum- and glucocorticoid-inducible kinase 1 (SGK1) splice variants based on alternative initiation of transcription. Cell Physiol Biochem 20:715–728
Huang M, Kamasani U, Prendergast GC (2006) RhoB facilitates c-Myc turnover by supporting efficient nuclear accumulation of GSK-3. Oncogene 25:1281–1289
Sherk AB, Frigo DE, Schnackenberg CG, Bray JD, Laping NJ, Trizna W, Hammond M, Patterson JR, Thompson SK, Kazmin D, Norris JD, McDonnell DP (2008) Development of a small-molecule serum- and glucocorticoid- regulated kinase-1 antagonist and its evaluation as a prostate cancer therapeutic. Cancer Res 68:7475–7483
Papadopoulou N, Papakonstanti EA, Kallergi G, Alevizopoulos K, Stournaras C (2009) Membrane androgen receptor activation in prostate and breast tumor cells: molecular signaling and clinical impact. IUBMB Life 61:56–61
Luft FC (2007) SGK1 survival through various lives may save us all. J Mol Med 85:657–659
You H, Jang Y, You-Ten AI, Okada H, Liepa J, Wakeham A, Zaugg K, Mak TW (2004) p53-dependent inhibition of FKHRL1 in response to DNA damage through protein kinase SGK1. Proc Natl Acad Sci U S A 01:14057–14062
Cornez I, Creppe C, Gillard M, Hennuy B, Chapelle JP, Dejardin E, Merville MP, Close P, Chariot A (2008) Deregulated expression of pro-survival and pro-apoptotic p53-dependent genes upon Elongator deficiency in colon cancer cells. Biochem Pharmacol 75:2122–2134
Hollstein M, Sidransky D, Vogelstein B, Harris CC (1991) p53 mutations in human cancers. Science 253:49–53
Jimenez GS, Khan SH, Stommel JM, Wahl GM (1999) p53 regulation by post translational modification and nuclear retention in response to diverse stresses. Oncogene 18:7656–7665
Li M, Brooks CL, Wu-Baer F, Chen D, Baer R, Wei Gu (2003) Mono- versus polyubiquitination: differential control of p53 fate by Mdm2. Science 302:1972–1975
Midgley CA, Lane DP (1997) p53 protein stability in tumour cells is not determined by mutation but is dependent on Mdm2 binding. Oncogene 15:1179–1189
Moll UM, Petrenko O (2003) The MDM2–p53 interaction. Mol Cancer Res 1:1001–1008. Review
Roth J, Dobbelstein M, Freedman DA, Shenk T, Levine AJ (1998) Nucleo-cytoplasmic shuttling of the mdm2 oncoprotein regulates the levels of the p53 protein via a pathway used by the human immunodeficiency virus rev protein. EMBO J 17:554–564
Chen Z, Knutson E, Wang S, Martinez LA, Albrecht T (2007) Stabilization of p53 in human cytomegalovirus-initiated cells is associated with sequestration of MDM2 and decreased p53 ubiquitination. J Biol Chem 282:29284–29295
Li Y, Jenkins CW, Nichols MA, Xiong Y (1994) Cell cycle expression and p53 regulation of the cyclin-dependent kinase inhibitor p21. Oncogene 9:2261–2268
Taylor WR, Stark GR (2001) Regulation of the G2/M transition by p53. Oncogene 20:1803–1815
Soengas MS, Alarcón RM, Yoshida H, Giaccia AJ, Hakem R, Mak TW, Lowe SW (1999) Apaf-1 and caspase-9 in p53-dependent apoptosis and tumor inhibition. Science 284:156–159
Porrello A, Cerone MA, Coen S, Gurtner A, Fontemaggi G, Cimino L, Piaggio G, Sacchi A, Soddu S (2000) p53 regulates myogenesis by triggering the differentiation activity of pRb. J Cell Biol 6:1295–1304
Cam H, Griesmann H, Beitzinger M, Hofmann L, Beinoraviciute-Kellner R, Sauer M, Hüttinger-Kirchhof N, Oswald C, Friedl P, Gattenlöhner S, Burek C, Rosenwald A, Stiewe T (2006) p53 family members in myogenic differentiation and rhabdomyosarcoma development. Cancer Cell 10:281–293
Mies F, Spriet C, Heliot L, Sariban-Sohraby S (2007) Epithelial Na channel stimulation by n-3 fatty acids requires proximity to a membrane-bound A-kinase-anchoring protein complexed with protein kinase A and phosphodiesterase. J Biol Chem 282:18339–18347
Maiyar AC, Phu PT, Huang AJ, Firestone GL (1997) Repression of glucocorticoid receptor transactivation and DNA binding of a glucocorticoid response element within the serum/glucocorticoid-inducible protein kinase (sgk) gene promoter by the p53 tumor suppressor protein. Mol Endocrinol 11:312–329
Rinaldo C, Prodosmo A, Mancini F, Iacovelli S, Sacchi A, Moretti F, Soddu S (2007) MDM2-regulated degradation of HIPK2 prevents p53Ser46 phosphorylation and DNA damage-induced apoptosis. Mol Cell 25:739–750
Treier M, Staszewski LM, Bohmann D (1994) Ubiquitin-dependent c-Jun degradation in vivo is mediated by the delta domain. Cell 78:787–798
Manickan E, Satoi J, Wang TC, Liang TJ (2001) Conditional liver-specific expression of simian virus 40 T antigen leads to regulatable development of hepatic neoplasm in transgenic mice. J Biol Chem 276:13989–13994
Bloom J, Pagano M (2005) Experimental tests to definitively determine ubiquitylation of a substrate. Methods Enzymol 399:249–266
Bhushan S, Malik F, Kumar A, Kaur Isher H, Pal Kaur I, Taneja SC, Singh J (2009) Activation of p53/p21/PUMA alliance and disruption of PI-3/Akt in multimodal targeting of apoptotic signaling cascades in cervical cancer cells by a pentacyclic triterpenediol from Boswellia serrata. Mol Carcinog. doi:10.1002/mc.20559
Chen F, Chang D, Goh M, Klibanov SA, Ljungman M (2000) Role of p53 in cell cycle regulation and apoptosis following exposure to proteasome inhibitors. Cell Growth Differ 11:239–246
Lober C, Lenz-Stoppler C, Dobbelstein M (2002) Adenovirus E1-transformed cells grow despite the continuous presence of transcriptionally active p53. J Gen Virol 83:2047–2057
Geyer RK, Yu ZK, Maki CG (2000) The MDM2 RING-finger domain is required to promote p53 nuclear export. Nat Cell Biol 2:569–573
Lee JH, Kim HS, Lee SJ, Kim KT (2007) Stabilization and activation of p53 induced by Cdk5 contributes to neuronal cell death. J Cell Sci 120:2259–2271
Feng J, Tamaskovic R, Yang Z, Brazil DP, Merlo A, Hess D, Hemmings BA (2004) Stabilization of Mdm2 via decreased ubiquitination is mediated by protein kinase B/Akt-dependent phosphorylation. J Biol Chem 279:35510–35517
Zhou BP, Liao Y, Xia W, Zou Y, Spohn B, Hung MC (2001) HER-2/neu induces p53 ubiquitination via Akt-mediated MDM2 phosphorylation. Nat Cell Biol 3:973–982
Weber HO, Ludwig RL, Morrison D, Kotlyarov A, Gaestel M, Vousden KH (2005) HDM2 phosphorylation by MAPKAP kinase 2. Oncogene 24:1965–1972
Ashcroft M, Ludwig RL, Woods DB, Copeland TD, Weber HO, MacRae EJ, Vousden KH (2002) Phosphorylation of MDM2 by Akt. Oncogene 21:1955–1962
Wulff P, Vallon V, Huang DY, Völkl H, Yu F, Richter K, Jansen M, Schlünz M, Klingel K, Loffing J, Kauselmann G, Bösl MR, Lang F, Kuhl D (2002) Impaired renal Na(+) retention in the sgk1-knockout mouse. J Clin Invest 110:1263–1268
Bera TK, Pastan I (2000) Mesothelin is not required for normal mouse development or reproduction. Mol Cell Biol 20:2902–2906
Zhang BH, Tang ED, Zhu T, Greenberg ME, Vojtek AB, Guan KL (2001) Serum- and glucocorticoid-inducible kinase SGK phosphorylates and negatively regulates B-Raf. J Biol Chem 276:31620–31626
Waldegger S, Barth P, Raber G, Lang F (1997) Cloning and characterization of a putative human serine/threonine protein kinase transcriptionally modified during anisotonic and isotonic alterations of cell volume. Proc Natl Acad Sci 94:4440–4445
Chung EJ, Sung YK, Farooq M, Kim Y, Im S, Tak WY, Hwang YJ, Kim YI, Han HS, Kim JC, Kim MK (2002) Gene expression profile analysis in human hepatocellular carcinoma by cDNA microarray. Mol Cell 14:382–387
Leong ML, Maiyar AC, Kim B, O'Keeffe BA, Firestone GL (2003) Expression of the serum- and glucocorticoid-inducible protein kinase, Sgk, is a cell survival response to multiple types of environmental stress stimuli in mammary epithelial cells. J Biol Chem 278:5871–5882
Kojima K, Shimanuki M, Shikami M, Samudio IJ, Ruvolo V, Corn P, Hanaoka N, Konopleva M, Andreeff M, Nakakuma H (2008) The dual PI3 kinase/mTOR inhibitor PI-103 prevents p53 induction by Mdm2 inhibition but enhances p53-mediated mitochondrial apoptosis in p53 wild-type AML. Leukemia 22:1728–1736
Feng Z, Zhang H, Levine AJ, Jin S (2005) The coordinate regulation of the p53 and mTOR pathways in cells. Proc Natl Acad Sci U S A 102:8204–8209
Acknowledgments
We thank the American Journal Experts for the editorial revision of the manuscript. This work was supported by the Fondazione Carical, Cofin 2005/068017_004, Interlink II04C0G4EM, Cofin 2006/prot. 2006065339_005, and Firb 2001/RBNE01724C_007. Rosario Amato was partially supported by the Fondazione “Lilli Funaro”.
Conflict of interest statement
The authors declare that they have no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary materials
Below is the link to the electronic supplementary material.
Supplementary 1
(PDF 439 kb)
Rights and permissions
About this article
Cite this article
Amato, R., D’Antona, L., Porciatti, G. et al. Sgk1 activates MDM2-dependent p53 degradation and affects cell proliferation, survival, and differentiation. J Mol Med 87, 1221–1239 (2009). https://doi.org/10.1007/s00109-009-0525-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-009-0525-5