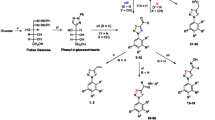
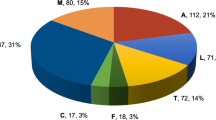
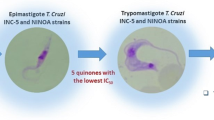
Abstract
Chagas disease or American trypanosomiasis is a major public health problem in Latin America. Approximately seven million people are currently infected worldwide. Despite the efforts to develop new drugs, only two nitroheterocyclic drugs (nifurtimox and benznidazole) are available for the treatment of Chagas disease, These drugs have been available since the 1970s, and no new drugs have been approved. Due to the lack of alternatives for the treatment of this disease, this review describes recent advances (2013–2019) concerning nitroheterocyclic compounds with activity against T. cruzi parasites, as well as new perspectives for future research.















Similar content being viewed by others
References
Almeida TC, Ribeiro LHG, dos Santos LBF et al. (2018) Synthesis, in vtiro and in vivo anti-Trypanosoma cruzi and toxicological activities of nitroaromatic Schiff bases. Biomed Pharmacother 108:1703–1711
Andrade MC, Oliveira MDF, Nagao-Dias AT et al (2013) Clinical and serological evolution in chronic Chagas disease patients in a 4-year pharmacotherapy follow-up: a preliminary study. Rev Soc Bras Med Trop 46: https://doi.org/10.1590/0037-8682-1646-2013
Arias DG, Herrera FE, Garay AS et al. (2017) Rational design of nitrofuran derivatives: Synthesis and valuation as inhibitors of Trypanosoma cruzi trypanothione reductase. Eur J Med Chem 125:1088–1097
Blau L, Menegon RF, Trossini GHG et al. (2013) Design, synthesis and biological evaluation of new aryl thiosemicarbazone as antichagasic candidates. Eur J Med Chem 67:142–151. https://doi.org/10.1016/j.ejmech.2013.04.022
Britta EA, Scariot DB, Falzirolli H et al. (2015) 4-Nitrobenzaldehyde thiosemicarbazone: a new compound derived from S-(-)-limonene that induces mitochondrial alterations in epimastigotes and trypomastigotes of Trypanosoma cruzi. Parasitology 142:978–988. https://doi.org/10.1017/S0031182015000141
Castro JA, DeMecca MM, Bartel LC (2006) Toxic side effects of drugs used to treat chagas’ disease (american trypanosomiasis). Hum Exp Toxicol 25:471–479. https://doi.org/10.1191/0960327106het653oa
Chen M, Borlak J, Tong W (2013) High lipophilicity and high daily dose of oral medications are associated with significant risk for drug-induced liver injury. Hepatology 58:388–396
Chung MC, Carvalho Güido RV, Favarato Martinelli T et al. (2003) Synthesis and in vitro evaluation of potential antichagasic hydroxymethylnitrofurazone (NFOH-121): a new nitrofurazone prodrug. Bioorg Med Chem 11:4779–4783. https://doi.org/10.1016/j.bmc.2003.07.004
Costa LB, De Oliveira Cardoso MV, De Oliveira Filho GB et al. (2016) Compound profiling and 3D-QSAR studies of hydrazone derivatives with activity against intracellular Trypanosoma cruzi. Bioorg Med Chem 24:1608–1618. https://doi.org/10.1016/j.bmc.2016.02.027
Curatolo W (1998) Physical chemical properties of oral drug candidates in the discovery and exploratory development settings. Pharm Sci Technol Today 1:387–393. https://doi.org/10.1016/S1461-5347(98)00097-2
Davies C, Cardozo RM, Negrette OS et al. (2010) Hydroxymethylnitrofurazone is active in a murine model of Chagas’ disease. Antimicrob Agents Chemother 54:3584–3589. https://doi.org/10.1128/AAC.01451-09
Davies C, Dey N, Negrette OS et al. (2014) Hepatotoxicity in mice of a novel anti-parasite drug candidate hydroxymethylnitrofurazone: a comparison with benznidazole. PLoS Negl Trop Dis 8:e3231. https://doi.org/10.1371/journal.pntd.0003231
de Andrade ALSS, Zicker F, de Oliveira RM et al. (1996) Randomised trial of efficacy of benznidazole in treatment of early Trypanosoma cruzi infection. Lancet 348:1407–1413. https://doi.org/10.1016/S0140-6736(96)04128-1
De Andrade P, Galo OA, Carvalho MR et al. (2015) 1,2,3-Triazole-based analogue of benznidazole displays remarkable activity against Trypanosoma cruzi. Bioorganic. Med Chem 23:6815–6826. https://doi.org/10.1016/j.bmc.2015.10.008
Dias LC, Dessoy MA, Silva JJN et al. (2009) Quimioterapia Da Doença de Chagas: Estado da Arte e Perspectivas no Desenvolvimento de Novos fármacos. Quim Nov 32:2444–2457
DNDi (2019) Drugs for neglected diseases initiative (DNDi), Neglected Tropical Diseases. https://www.dndi.org/diseases-projects/chagas/. (Accessed 21 August 2019).
Do Amaral AT, Andrade CH, Kümmerle AE, Guido RVC (2017) A evolução da Química Medicinal no Brasil: Avanços nos 40 anos da Sociedade Brasileira de Química. Quim Nova 40:694–700. https://doi.org/10.21577/0100-4042.20170075
Ekins S, de Siqueira-Neto JL, McCall L-I et al. (2015) Machine learning models and pathway genome data base for trypanosoma cruzi drug discovery. PLoS Negl Trop Dis 9:e0003878. https://doi.org/10.1371/journal.pntd.0003878
Elias PR, Coelho GS, Xavier VF et al. (2016) Synthesis of xylitan derivatives and preliminary evaluation of in vitro trypanocidal activity. Molecules 21:1–13. https://doi.org/10.3390/molecules21101342
Fernandez ML, Riarte AR, Marson ME et al. (2016) Pharmacokinetic and pharmacodynamics responses in adult patients with Chagas disease treated with a new formulation of benznidazole. Mem Inst Oswaldo Cruz 111:218–221. https://doi.org/10.1590/0074-02760150401
Ferreira EI (2012) Planejamento de Fármacos na Área de Doença de Chagas: Avanços e Desafios Planejamento de Fármacos na Área de Doença de Chagas. Avanços e Desafios 4:225–246
Fonseca-Berzal C, Ibáñez-Escribano A, Reviriego F et al. (2016) Antichagasic and trichomonacidal activity of 1-substituted 2-benzyl-5-nitroindazolin-3-ones and 3-alkoxy-2-benzyl-5-nitro-2H-indazoles. Eur J Med Chem 115:295–310. https://doi.org/10.1016/j.ejmech.2016.03.036
Francisco AF, Jayawardhana S, Lewis MD et al. (2016) Nitroheterocyclic drugs cure experimental Trypanosoma cruzi infections more effectively in the chronic stage than in the acute stage. Sci Rep 6:35351. https://doi.org/10.1038/srep35351
Francisco AF, Lewis MD, Jayawardhana S et al. (2015) Limited ability of posaconazole to cure both acute and chronic trypanosoma cruzi infections revealed by highly sensitive in vivo imaging. Antimicrob Agents Chemother 59:4653–4661. https://doi.org/10.1128/AAC.00520-15
Gomes PAT, de M, Oliveira AR, De Oliveira Cardoso MV et al. (2016) Phthalimido-thiazoles as building blocks and their effects on the growth and morphology of Trypanosoma cruzi. Eur J Med Chem 111:46–57. https://doi.org/10.1016/j.ejmech.2016.01.010
Guido RVC, Ferreira EI, Nassute JC, Varanda EA, Chung MC (2001) Diminuição da atividade mutagênica do pró-fármaco NFOH-121 em relação ao nitrofural (nitrofurazona). Rev Ciências Farm 22:319–333
Lancaster JR, (2015) Nitric oxide: a brief overview of chemical and physical properties relevant to therapeutic applications Futur Sci OA 1:FSO59. https://doi.org/10.4155/fso.15.59
Leeson PD, Springthorpe B (2007) The influence of drug-like concepts on decisionmaking in medicinal chemistry. Nat Rev Drug Disco 6:881–902. https://doi.org/10.1038/nrd2445
Lipinski CA, Lombardo F, Dominy BW, Feeney PJ (2001) Experimental and computational approaches to estimate solubility and permeability in discovery and development Settings. Adv Drug Deliv Rev 46:3–26
Lipinski CA, Lombardo F, Dominy BW, Feeney PJ (1997) Experimental and computational approaches to estimate solubility and permeability in drug discovery and developmental settings. Adv Drug Deliv Rev 23:3–25. https://doi.org/10.1016/S0169-409X(96)00423-1
Manjunatha UH, Smith OW (2015) Perspective: challenges and opportunities in TB drug discovery from phenotypic screening. Bioorg Med Chem 23:5087–5097. https://doi.org/10.1016/j.bmc.2014.12.031
Maya JD, Orellana M, Ferreira J et al (2010) Chagas disease: present status of pathogenic mechanisms and chemotherapy. Biol Res 323–231. https://doi.org/10.4067/S0716-97602010000300009.
Moreno-Rodríguez A, Salazar-Schettino PM, Bautista JL et al. (2014) In vitro antiparasitic activity of new thiosemicarbazones in strains of Trypanosoma cruzi. Eur J Med Chem 87:23–29. https://doi.org/10.1016/j.ejmech.2014.09.027
Moreno-Viguri E, Jiménez-Montes C, Martín-Escolano R et al. (2016) In vitro and in vivo anti-trypanosoma cruzi activity of new arylamine mannich base-type derivatives. J Med Chem 59:10929–10945. https://doi.org/10.1021/acs.jmedchem.6b00784
Morillo CA, Marin-Neto JA, Avezum A et al. (2015) Randomized trial of benznidazole for chronic chagas’ cardiomyopathy. N Engl J Med 2015:1295–1306. 10.1056/NEJMoa1507574
Olmo F, Marin C, Rosales MJ et al. (2015) Synthesis and evaluation of in vitro and in vivo trypanocidal properties of a new imidazole-containing nitrophthalazine derivative. Eur J Med Chem 106:106–119. https://doi.org/10.1016/j.ejmech.2015.10.034
Palace-Berl F, Pasqualoto KFM, Zingales B et al. (2018) Investigating the structure-activity relationships of N’-[(5-nitrofuran-2-yl) methylene] substituted hydrazides against Trypanosoma cruzi to design novel active compounds. Eur J Med Chem 144:29–40
Pan P, Vermelho AB, Rodrigues GC et al. (2013) Cloning, characterization, and sulfonamide and thiol inhibition studies of an a-carbonic anhydrase from Trypanosoma cruzi, the causative agent of chagas disease. J Med Chem 56:1761–1771. https://doi.org/10.1021/jm4000616
Papadopoulou MV, Bloomer WD, Rosenzweig HS et al. (2014) Novel nitro(triazole/imidazole)-based heteroarylamides/sulfonamides as potential antitrypanosomal agents. Eur J Med Chem 87:79–88. https://doi.org/10.1371/journal.pntd.0000440
Papadopoulou MV, Bloomer WD, Rosenzweig HS et al. (2015) Discovery of potent nitrotriazole-based antitrypanosomal agents: in vitro and in vivo evaluation. Bioorg Med Chem 23:6467–6476. https://doi.org/10.1016/j.bmc.2015.08.014
Paula FR, Serrano SHP, Tavares LC (2009) Aspectos mecanísticos da bioatividade e toxicidade de nitrocompostos. Quim Nova 32:1013–1020
Rassi A, Rassi A, Marin-Neto JA (2010) Chagas disease. Lancet 375:1388–1402. https://doi.org/10.1016/S0140-6736(10)60061-X
Scarim CB, de Andrade CR, da Rosa JA et al. (2018) Hydroxymethylnitrofurazone treatment in indeterminate form of chronic Chagas disease: reduced intensity of tissue parasitism and inflammation—a histopathological study. Int J Exp Pathol 99:236–248. https://doi.org/10.1111/iep.12289
Serafim RAM, Gonçalves JE, de Souza FP et al. (2014) Design, synthesis and biological evaluation of hybrid bioisoster derivatives of N-acylhydrazone and furoxan groups with potential and selective anti-trypanosoma cruzi activity. Eur J Med Chem 82:418–425
Sherlock IA (1999) Epidemiology and dinamics of the vectorial transmission of Chagas disease. Mem Inst Oswaldo Cruz 4:385–386
Da Silva GMS, Mediano MFF, Brasil PEAA Do et al. (2014) A clinical adverse drug reaction prediction model for patients with chagas disease treated with benznidazolE. Antimicrob Agents Chemother 58:6371–6377. https://doi.org/10.1128/AAC.02842-14
Silva FT, Franco CH, Favaro DC et al. (2016) Design, synthesis and antitrypanosomal activity of some nitrofurazone 1,2,4-triazolic bioisosteric analogues. Eur J Med Chem 121:553–560. https://doi.org/10.1016/j.ejmech.2016.04.065
Sosa-Estani S, Segura E, Ruiz A et al. (1998) Efficacy of chemotherapy with benznidazole in children in the indeterminate phase of Chagas’ disease. Am J Trop Med Hyg 59:526–9
Soy D, Aldasoro E, Guerrero L et al. (2015) Population pharmacokinetics of benznidazole in adult patients with Chagas disease. Antimicrob Agents Chemother 59:3342–3349. https://doi.org/10.1128/AAC.05018-14
Tarcsay A, Keseru GM (2013) Contributions of molecular properties to drug promiscuity. J Med Chem 56:1789–1795. https://doi.org/10.1021/jm301514n
Tetko IV (2005) Computing chemistry on the web. Drug Discov Today 10:1497–1500. https://doi.org/10.1016/S1359-6446(05)03584-1
Thompson AM, Blaser A, Palmer BD et al. (2017) 6-Nitro-2,3-dihydroimidazo[2,1-b][1,3]thiazoles: Facile synthesis and comparative appraisal against tuberculosis and neglected tropical diseases. Bioorg Med Chem Lett 27:2583–2589. https://doi.org/10.1016/j.bmcl.2017.03.069
Trossini GHG, Malvezzi A, T-do Amaral A et al. (2010) Cruzain inhibition by hydroxymethylnitrofurazone and nitrofurazone: investigation of a new target in Trypanosoma cruzi. J Enzym Inhib Med Chem 25:62–67. https://doi.org/10.3109/14756360902941058
Waring MJ (2010) Lipophilicity in drug discovery. Expet Opin Drug Disco 5:235–248. https://doi.org/10.1517/17460441003605098
Wenlock MC, Barton P (2013) In silico physicochemical parameter predictions. Mol Pharm 10:1224–1235. https://doi.org/10.1021/mp300537k
World and Health Organization (WHO) (2019) Neglected tropical diseases. World and Health Organization (WHO). http://www.who.int/neglected_diseases/diseases/en/. Accessed 21 August 2019
WHO (2002). Control of Chagas disease: second report of the WHO expert committee. World Health Organization (2000: Brasilia, Brazil). Geneva, World Health Organization. 905:109
Acknowledgements
This work was financed in part by the Coordenação de Aperfeiçoamento Pessoal de Nível Superior—Brasil (CAPES)—Finance code 001. Moreover, the authors would like to thank the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP—2016/10847-9) for the research fellowship assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
About this article
Cite this article
Benito Scarim, C., Man Chin, C. Nitroheterocyclic derivatives: privileged scaffold for drug development against Chagas disease. Med Chem Res 28, 2099–2108 (2019). https://doi.org/10.1007/s00044-019-02453-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-019-02453-y