Abstract
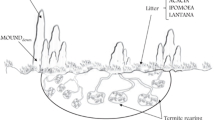
Fungus-farming termites cultivate a mutualistic fungus Termitomyces inside their nest mounds in CO2-rich environments. For sustainable harvests, termites must control weedy parasitic fungi such as Pseudoxylaria that may exploit resources meant for cultivar growth. Earlier, we discovered that termites exploit fungal scents to distinguish between crop and weedy fungi leading to weed burial that could contribute to its control. While chemical antifungals have been reported in termite farms, live burial per se as an antifungal activity has never been investigated. In this study, major and minor worker castes of termites buried the weedy fungus with soil to a significantly greater extent than the crop fungus. This live burial by worker termites led to greater decrease in the survival of the weedy fungus compared to the crop fungus, even after controlling for the differential amount of soil deposition. Such a decrease in parasite survival could result from local hypoxia generated by the burial process. Our experiments with artificial burial revealed that, in the absence of chemical factors such as fungicides, weed survival is indeed negatively affected by the resulting hypoxia alone. However, hypoxia associated with artificial burial also decreased crop survival, explaining why natural burial of crop fungi is minimal. Farmer termites may, therefore, contain weeds in their fungus farms by selectively burying weed-infested areas, resulting in antifungal activity which in part could be due to local hypoxic conditions. These results show how organisms may exploit the abiotic effects of behavioural actions as an effective defence against parasites.


Similar content being viewed by others
References
Abramowski D, Currie CR, Poulsen M (2011) Caste specialization in behavioral defenses against fungus garden parasites in Acromyrmex octospinosus leaf-cutting ants. Insectes Soc 58:65–75. https://doi.org/10.1007/s00040-010-0117-y
Badertscher S, Gerber C, Leuthold RH (1983) Polyethism in food supply and processing in termite colonies of Macrotermes subhyalinus (Isoptera). Behav Ecol Sociobiol 12:115–119. https://doi.org/10.1007/BF00343201
Batra LR, Batra SWT (1979) Termite-fungus mutualism. In: Batra LR (ed) Insect-fungus symbiosis: nutrition, mutualism and commensalism. Allanheld and Osmun, Montclair, pp 117–163
Beemelmanns C, Ramadhar TR, Kim KH et al (2017) Macrotermycins A–D, glycosylated macrolactams from a termite-associated Amycolatopsis sp. M39. Org Lett. https://doi.org/10.1021/acs.orglett.6b03831
Benvenuti S, Macchia M (1995) Effect of hypoxia on buried weed seed germination. Weed Res 35:343–351. https://doi.org/10.1111/j.1365-3180.1995.tb01629.x
Bot ANM, Currie CR, Hart AG, Boomsma JJ (2001) Waste management in leaf-cutting ants. Ethol Ecol Evol 13:225–237. https://doi.org/10.1080/08927014.2001.9522772
Chauhan BS, Johnson DE (2011) Ecological studies on Echinochloa crus-galli and the implications for weed management in direct-seeded rice. Crop Prot 30:1385–1391. https://doi.org/10.1016/j.cropro.2011.07.013
Chouvenc T, Su N-Y (2010) Apparent synergy among defense mechanisms in subterranean termites (Rhinotermitidae) against epizootic events: limits and potential for biological control. J Econ Entomol 103:1327–1337. https://doi.org/10.1603/EC09407
Chouvenc T, Su N-Y (2012) When subterranean termites challenge the rules of fungal epizootics. PLoS ONE 7:e34484. https://doi.org/10.1371/journal.pone.0034484
Chouvenc T, Su N-Y, Elliott ML (2008) Interaction between the subterranean termite Reticulitermes flavipes (Isoptera: Rhinotermitidae) and the entomopathogenic fungus Metarhizium anisopliae in foraging arenas. J Econ Entomol 101:885–893
Chouvenc T, Robert A, Sémon E, Bordereau C (2012) Burial behaviour by dealates of the termite Pseudacanthotermes spiniger (Termitidae, Macrotermitinae) induced by chemical signals from termite corpses. Insectes Soc 59:119–125. https://doi.org/10.1007/s00040-011-0197-3
Cremer S, Armitage SAO, Schmid-Hempel P (2007) Social immunity. Curr Biol 17:R693–R702. https://doi.org/10.1016/j.cub.2007.06.008
Currie CR, Stuart AE (2001) Weeding and grooming of pathogens in agriculture by ants. Proc R Soc Lond B 268:1033–1039. https://doi.org/10.1098/rspb.2001.1605
Currie CR, Scott JA, Summerbell RC, Malloch D (1999) Fungus-growing ants use antibiotic-producing bacteria to control garden parasites. Nature 398:701–704. https://doi.org/10.1038/19519
Hart AG (2002) Waste management in the leaf-cutting ant Atta colombica. Behav Ecol 13:224–231. https://doi.org/10.1093/beheco/13.2.224
Hart AG, Ratnieks FLW (2001) Task partitioning, division of labour and nest compartmentalisation collectively isolate hazardous waste in the leafcutting ant Atta cephalotes. Behav Ecol Sociobiol 49:387–392. https://doi.org/10.1007/s002650000312
Heil M, McKey D (2003) Protective ant-plant interactions as model systems in ecological and evolutionary research. Annu Rev Ecol Evol Syst 34:425–553. https://doi.org/10.1146/annurev.ecolsys.34.011802.132410
Hinze B, Leuthold RH (1999) Age related polyethism and activity rhythms in the nest of the termite Macrotermes bellicosus (Isoptera, Termitidae). Insectes Soc 46:392–397. https://doi.org/10.1007/s000400050162
Hongoh Y, Ekpornprasit L, Inoue T et al (2006) Intracolony variation of bacterial gut microbiota among castes and ages in the fungus-growing termite Macrotermes gilvus. Mol Ecol 15:505–516. https://doi.org/10.1111/j.1365-294X.2005.02795.x
Hsieh H-M, Lin C-R, Fang M-J et al (2010) Phylogenetic status of Xylaria subgenus Pseudoxylaria among taxa of the subfamily Xylarioideae (Xylariaceae) and phylogeny of the taxa involved in the subfamily. Mol Phylogenet Evol 54:957–969. https://doi.org/10.1016/j.ympev.2009.12.015
Katariya L, Ramesh PB, Gopalappa T et al (2017a) Fungus-farming termites selectively bury weedy fungi that smell different from crop fungi. J Chem Ecol 43:986–995. https://doi.org/10.1007/s10886-017-0902-4
Katariya L, Ramesh PB, Gopalappa T, Borges RM (2017b) Sex and diversity: the mutualistic and parasitic fungi of a fungus-growing termite differ in genetic diversity and reproductive strategy. Fungal Ecol 26:20–27. https://doi.org/10.1016/j.funeco.2016.11.003
Katariya L, Ramesh PB, Borges RM (2018) Dynamic environments of fungus-farming termite mounds exert growth-modulating effects on fungal crop parasites. Environ Microbiol 20:971–979. https://doi.org/10.1111/1462-2920.14026
Kim KH, Ramadhar TR, Beemelmanns C et al (2014) Natalamycin A, an ansamycin from a termite-associated Streptomyces sp. Chem Sci 5:4333–4338. https://doi.org/10.1039/C4SC01136H
Korb J, Linsenmair KE (1999) The architecture of termite mounds: a result of a trade-off between thermoregulation and gas exchange? Behav Ecol 10:312–316. https://doi.org/10.1093/beheco/10.3.312
Kroiss J, Kaltenpoth M, Schneider B et al (2010) Symbiotic streptomycetes provide antibiotic combination prophylaxis for wasp offspring. Nat Chem Biol 6:261–263. https://doi.org/10.1038/nchembio.331
Lamberty M, Zachary D, Lanot R et al (2001) Insect immunity: constitutive expression of a cysteine-rich antifungal and a linear antibacterial peptide in a termite insect. J Biol Chem 276:4085–4092. https://doi.org/10.1074/jbc.M002998200
Little AEF, Murakami T, Mueller UG, Currie CR (2006) Defending against parasites: fungus-growing ants combine specialized behaviours and microbial symbionts to protect their fungus gardens. Biol Lett 2:12–16. https://doi.org/10.1098/rsbl.2005.0371
López-Riquelme GO, Fanjul-Moles ML (2013) The funeral ways of social insects. Social strategies for corpse disposal. Trends Entomol 9:71–129
Meletiadis J, Meis JFGM, Mouton JW et al (2000) Comparison of NCCLS and 3-(4,5-dimethyl-2-thiazyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) methods of in vitro susceptibility testing of filamentous fungi and development of a new simplified method. J Clin Microbiol 38:2949–2954
Morelos-Juárez C, Walker TN, Lopes JFS, Hughes WOH (2010) Ant farmers practice proactive personal hygiene to protect their fungus crop. Curr Biol 20:R553–R554. https://doi.org/10.1016/j.cub.2010.04.047
Myles TG (2002) Alarm, aggregation and defense by Reticulitermes flavipes in response to a naturally occurring isolate of Metarhizium anisopliae. Sociobiology 40:243–256
Nobre T, Aanen DK (2012) Fungiculture or termite husbandry? The ruminant hypothesis. Insects 3:307–323. https://doi.org/10.3390/insects3010307
Oh D-C, Scott JJ, Currie CR, Clardy J (2009) Mycangimycin, a polyene peroxide from a mutualist Streptomyces sp. Org Lett 11:633–636. https://doi.org/10.1021/ol802709x
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for statistical Computing, Vienna, Austria. https://www.R-project.org/
Renucci M, Tirard A, Provost E (2011) Complex undertaking behavior in Temnothorax lichtensteini ant colonies: from corpse-burying behavior to necrophoric behavior. Insect Soc 58:9–16. https://doi.org/10.1007/s00040-010-0109-y
Rosengaus RB, Guldin MR, Traniello JF (1998) Inhibitory effect of termite fecal pellets on fungal spore germination. J Chem Ecol 24:1697–1706
Rosengaus RB, Lefebvre ML, Traniello JF (2000) Inhibition of fungal spore germination by Nasutitermes: evidence for a possible antiseptic role of soldier defensive secretions. J Chem Ecol 26:21–39
Rosengaus RB, Traniello JFA, Lefebvre ML, Maxmen AB (2004) Fungistatic activity of the sternal gland secretion of the dampwood termite Zootermopsis angusticollis. Insectes Soc 51:259–264. https://doi.org/10.1007/s00040-004-0749-x
RStudio Team (2016) RStudio: integrated development for R. RStudio, Boston, MA. https://www.rstudio.com
Saikkonen K, Gundel PE, Helander M (2013) Chemical ecology mediated by fungal endophytes in grasses. J Chem Ecol 39:962–968. https://doi.org/10.1007/s10886-013-0310-3
Scott JJ, Oh D-C, Yuceer MC et al (2008) Bacterial protection of beetle-fungus mutualism. Science 322:63–63. https://doi.org/10.1126/science.1160423
Um S, Fraimout A, Sapountzis P et al (2013) The fungus-growing termite Macrotermes natalensis harbors bacillaene-producing Bacillus sp. that inhibit potentially antagonistic fungi. Sci Rep 3:3250. https://doi.org/10.1038/srep03250
Visser AA, Ros VID, De Beer ZW et al (2009) Levels of specificity of Xylaria species associated with fungus-growing termites: a phylogenetic approach. Mol Ecol 18:553–567. https://doi.org/10.1111/j.1365-294X.2008.04036.x
Visser AA, Kooij PW, Debets AJM et al (2011) Pseudoxylaria as stowaway of the fungus-growing termite nest: interaction asymmetry between Pseudoxylaria, Termitomyces and free-living relatives. Fungal Ecol 4:322–332. https://doi.org/10.1016/j.funeco.2011.05.003
Visser AA, Nobre T, Currie CR et al (2012) Exploring the potential for actinobacteria as defensive symbionts in fungus-growing termites. Microb Ecol 63:975–985. https://doi.org/10.1007/s00248-011-9987-4
Wickham H (2009) Ggplot2: elegant graphics for data analysis. Springer, New York
Zachariah N, Das A, Murthy TG, Borges RM (2017) Building mud castles: a perspective from brick-laying termites. Sci Rep 7:4692. https://doi.org/10.1038/s41598-017-04295-3
Acknowledgements
This research was funded by the Council of Scientific and Industrial Research (37(1561)/12/EMR-II); the Ministry of Environment, Forest & Climate Change; the Department of Biotechnology; and the Department of Science and Technology-FIST, Government of India. We are grateful to C Viraktamath and Rashmi Shanbhag for termite identifications; Abdul Hakkim for the MTT assay; Tejas Murthy for providing processed soil; Yathiraj Ganesh for field work; Sunitha Murray for administrative support; E S Anupriya for help in weighing; and Thejashwini Gopalappa and Anagha Setlur for help with experiments.
Author information
Authors and Affiliations
Contributions
LK and RMB conceived the study. LK designed the methodology with inputs from PBR, and LK, PBR and AS carried out the experiments. LK analysed all the data. LK wrote the first draft of manuscript and RMB and LK contributed to revision and editing. RMB acquired the funding.
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Katariya, L., Ramesh, P.B., Sharma, A. et al. Local hypoxia generated by live burial is effective in weed control within termite fungus farms. Insect. Soc. 65, 561–569 (2018). https://doi.org/10.1007/s00040-018-0644-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00040-018-0644-5