Abstract
Objective
To understand the epigenetic mechanism underlying the PR-B gene silencing in endometrial cancer (EC) cells, we compared the chromatin composition between transcriptionally active and silenced PR-B genes in EC cell lines and cancer tissues.
Methods
Chromatin Immunoprecipitation (ChIP) assay was performed to measure MBD occupancy and histone acetylation/methylation in transcriptionally active and silenced PR-B genes. PR-B-positive/-negative, as well as epigenetic inhibitor-treated/-untreated EC cells were used as study models. Real-time polymerase chain reaction (PCR) and Western blot analysis were applied to measure the mRNA and protein levels of PR-B, MBD, and histones.
Results
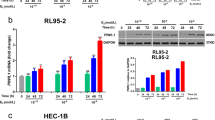
A close association among PR-B methylation, MBD binding and PR-B gene silencing was observed. Treatment with epigenetic inhibitors led to dynamic changes in the PR-B chromatin composition and gene expression. Increased H3/H4 acetylation and H3-K4 methylation, and decreased H3-K9 methylation were found to be associated with re-activation of silenced PR-B genes. MeCP2 knockdown resulted in a decreased MeCP2 binding to PR-B genes and an increased PR-B expression. ChIP analysis of MeCP2 binding to PR-B genes in the PR-B-positive/-negative EC samples confirmed the significant role of MeCP2 in PR-B silencing.
Conclusion
PR-B gene expression is regulated by a concerted action of epigenetic factors including DNA methylation, MBD binding, and histone modifications. MeCP2 occupancy of PR-B genes plays a critical role in PR-B gene silencing. These findings enriched our knowledge of the epigenetic regulation of PR-B expression in EC, and suggested that the epigenetic re-activation of PR-B could be explored as a potential strategy to sensitize the PR-B-negative endometrial cancers to progestational therapy.







Similar content being viewed by others
References
Sitruk-Ware R, Plu-Bureau G (1999) Progestins and cancer. Gynecol Endocrinol 13(Suppl 4):3–9
Markman M (2005) Hormonal therapy of endometrial cancer. Eur J Cancer 41:673–675
Pike MC, Pearce CL, Wu AH (2004) Prevention of cancers of the breast, endometrium and ovary. Oncogene 23:6379–6391
Podratz KC, O’Brien PC, Malkasian GD Jr, Decker DG, Jefferies JA, Edmonson JH (1985) Effects of progestational agents in treatment of endometrial carcinoma. Obstet Gynecol 66:106–110
Figueroa-Casas PR, Ettinger B, Delgado E, Javkin A, Vieder C (2001) Reversal by medical treatment of endometrial hyperplasia caused by estrogen replacement therapy. Menopause 8:420–423
Ramirez PT, Frumovitz M, Bodurka DC, Sun CC, Levenback C (2004) Hormonal therapy for the management of grade 1 endometrial adenocarcinoma: a literature review. Gynecol Oncol 95:133–138
Lentz SS, Brady MF, Major FJ, Reid GC, Soper JT (1996) High-dose megestrol acetate in advanced or recurrent endometrial carcinoma: a Gynecologic Oncology Group Study. J Clin Oncol 14:357–361
Podczaski E, Mortel R (2001) Hormonal treatment of endometrial cancer: past, present and future. Best Pract Res Clin Obstet Gynaecol 15:469–489
van Rijswijk RE, Vermorken JB (2000) Drug therapy for gynaecological cancer in older women. Drugs Aging 17:13–32
Whitney CW, Brunetto VL, Zaino RJ, Lentz SS, Sorosky J, Armstrong DK, Lee RB (2004) Phase II study of medroxyprogesterone acetate plus tamoxifen in advanced endometrial carcinoma: a Gynecologic Oncology Group study. Gynecol Oncol 92:4–9
Fiorica JV, Brunetto VL, Hanjani P, Lentz SS, Mannel R, Andersen W (2004) Phase II trial of alternating courses of megestrol acetate and tamoxifen in advanced endometrial carcinoma: a Gynecologic Oncology Group study. Gynecol Oncol 92:10–14
Burnett AF, Bahador A, Amezcua C, Anastrozole (2004) An aromatase inhibitor, and medroxyprogesterone acetate therapy in premenopausal obese women with endometrial cancer: a report of two cases successfully treated without hysterectomy. Gynecol Oncol 94:832–834
Williams SP, Sigler PB (1998) Atomic structure of progesterone complexed with its receptor. Nature 393:392–396
Wagner BL, Norris JD, Knotts TA, Weigel NL, McDonnell DP (1998) The nuclear corepressors NCoR and SMRT are key regulators of both ligand- and 8-bromo-cyclic AMP-dependent transcriptional activity of the human progesterone receptor. Mol Cell Biol 18:1369–1378
Charnock-Jones DS, Macpherson AM, Archer DF, Leslie S, Makkink WK, Sharkey AM, Smith SK (2000) The effect of progestins on vascular endothelial growth factor, oestrogen receptor and progesterone receptor immunoreactivity and endothelial cell density in human endometrium. Hum Reprod 15(Suppl 3):85–95
Shiozawa T, Horiuchi A, Kato K, Obinata M, Konishi I, Fujii S, Nikaido T (2001) Up-regulation of p27Kip1 by progestins is involved in the growth suppression of the normal and malignant human endometrial glandular cells. Endocrinology 142:4182–4188
Kester HA, Sonneveld E, van der Saag PT, van der Burg B (2003) Prolonged progestin treatment induces the promoter of CDK inhibitor p21 Cip1, Waf1 through activation of p53 in human breast and endometrial tumor cells. Exp Cell Res 284:264–273
Amezcua CA, Lu JJ, Felix JC, Stanczyk FZ, Zheng W (2000) Apoptosis may be an early event of progestin therapy for endometrial hyperplasia. Gynecol Oncol 79:169–176
Quinn MA, Cauchi M, Fortune D (1985) Endometrial carcinoma: steroid receptors and response to medroxyprogesterone acetate. Gynecol Oncol 21:314–319
Kleine W, Maier T, Geyer H, Pfleiderer A (1990) Estrogen and progesterone receptors in endometrial cancer and their prognostic relevance. Gynecol Oncol 38:59–65
Podratz KC (1990) Hormonal therapy in endometrial carcinoma. Recent Results Cancer Res 118:242–251
Dai D, Wolf DM, Litman ES, White MJ, Leslie KK (2002) Progesterone inhibits human endometrial cancer cell growth and invasiveness: down-regulation of cellular adhesion molecules through progesterone B receptors. Cancer Res 62:881–886
Kumar NS, Richer J, Owen G, Litman E, Horwitz KB, Leslie KK (1998) Selective down-regulation of progesterone receptor isoform B in poorly differentiated human endometrial cancer cells: implications for unopposed estrogen action. Cancer Res 58:1860–1865
Sakaguchi H, Fujimoto J, Hong BL, Nakagawa Y, Tamaya T (2004) Drastic decrease of progesterone receptor form B but not A mRNA reflects poor patient prognosis in endometrial cancers. Gynecol Oncol 93:394–399
Hanekamp EE, Kuhne EC, Smid-Koopman E, de Ruiter PE, Chadha-Ajwani S, Brinkmann AO, Burger CW, Grootegoed JA, Huikeshoven FJ, Blok LJ (2002) Loss of progesterone receptor may lead to an invasive phenotype in human endometrial cancer. Eur J Cancer 38(Suppl 6):S71–S72
Pijnenborg JM, Romano A, Dam-de Veen GC, Dunselman GA, Fischer DC, Groothuis PG, Kieback DG (2005) Aberrations in the progesterone receptor gene and the risk of recurrent endometrial carcinoma. J Pathol 205:597–605
Sasaki M, Dharia A, Oh BR, Tanaka Y, Fujimoto S, Dahiya R (2001) Progesterone receptor B gene inactivation and CpG hypermethylation in human uterine endometrial cancer. Cancer Res 61:97–102
Jin F, Dowdy SC, Xiong Y, Eberhardt NL, Podratz KC, Jiang SW (2005) Up-regulation of DNA methyltransferase 3B expression in endometrial cancers. Gynecol Oncol 96:531–538
Xiong Y, Dowdy SC, Xue A, Shujuan J, Eberhardt NL, Podratz KC, Jiang SW (2005) Opposite alterations of DNA methyltransferase gene expression in endometrioid and serous endometrial cancers. Gynecol Oncol 96:601–609
Baylin SB, Esteller M, Rountree MR, Bachman KE, Schuebel K, Herman JG (2001) Aberrant patterns of DNA methylation, chromatin formation and gene expression in cancer. Hum Mol Genet 10:687–692
Esteller M, Herman JG (2002) Cancer as an epigenetic disease: DNA methylation and chromatin alterations in human tumours. J Pathol 196:1–7
Jones PA, Baylin SB (2002) The fundamental role of epigenetic events in cancer. Nat Rev Genet 3:415–428
Bird AP, Wolffe AP (1999) Methylation-induced repression—belts, braces, and chromatin. Cell 99:451–454
Irvine RA, Lin IG, Hsieh CL (2002) DNA methylation has a local effect on transcription and histone acetylation. Mol Cell Biol 22:6689–6696
Shi Y (2007) Histone lysine demethylases: emerging roles in development, physiology and disease. Nat Rev Genet 8:829–833
Dhasarathy A, Wade PA (2008) The MBD protein family-reading an epigenetic mark? Mutat Res 647:39–43
Tsukada Y, Ishitani T, Nakayama KI (2010) KDM7 is a dual demethylase for histone H3 Lys 9 and Lys 27 and functions in brain development. Genes Dev 24:432–437
Nishida M, Kasahara K, Oki A, Satoh T, Arai Y, Kubo T (1996) Establishment of eighteen clones of Ishikawa cells. Hum Cell 9:109–116
Xiong Y, Dowdy SC, Podratz KC, Jin F, Attewell JR, Eberhardt NL, Jiang SW (2005) Histone deacetylase inhibitors decrease DNA methyltransferase-3B messenger RNA stability and down-regulate de novo DNA methyltransferase activity in human endometrial cells. Cancer Res 65:2684–2689
Dobosy JR, Selker EU (2001) Emerging connections between DNA methylation and histone acetylation. Cell Mol Life Sci 58:721–727
Vaissiere T, Sawan C, Herceg Z (2008) Epigenetic interplay between histone modifications and DNA methylation in gene silencing. Mutat Res 659:40–48
Ikegami K, Ohgane J, Tanaka S, Yagi S, Shiota K (2009) Interplay between DNA methylation, histone modification and chromatin remodeling in stem cells and during development. Int J Dev Biol 53:203–214
Tamaru H, Selker EU (2001) A histone H3 methyltransferase controls DNA methylation in Neurospora crassa. Nature 414:277–283
Cosgrove DE, Cox GS (1990) Effects of sodium butyrate and 5-azacytidine on DNA methylation in human tumor cell lines: variable response to drug treatment and withdrawal. Biochim Biophys Acta 1087:80–86
Hu JF, Oruganti H, Vu TH, Hoffman AR (1998) The role of histone acetylation in the allelic expression of the imprinted human insulin-like growth factor II gene. Biochem Biophys Res Commun 251:403–408
Selker EU (1998) Trichostatin A causes selective loss of DNA methylation in Neurospora. Proc Natl Acad Sci USA 95:9430–9435
Cameron EE, Bachman KE, Myohanen S, Herman JG, Baylin SB (1999) Synergy of demethylation and histone deacetylase inhibition in the re-expression of genes silenced in cancer. Nat Genet 21:103–107
El-Osta A, Kantharidis P, Zalcberg JR, Wolffe AP (2002) Precipitous release of methyl-CpG binding protein 2 and histone deacetylase 1 from the methylated human multidrug resistance gene (MDR1) on activation. Mol Cell Biol 22:1844–1857
Robert MF, Morin S, Beaulieu N, Gauthier F, Chute IC, Barsalou A, MacLeod AR (2003) DNMT1 is required to maintain CpG methylation and aberrant gene silencing in human cancer cells. Nat Genet 33:61–65
Ng HH, Bird A (1999) DNA methylation and chromatin modification. Curr Opin Genet Dev 9:158–163
Ballestar E, Wolffe AP (2001) Methyl-CpG-binding proteins. Targeting specific gene repression. Eur J Biochem 268:1–6
Chen ZJ, Pikaard CS (1997) Epigenetic silencing of RNA polymerase I transcription: a role for DNA methylation and histone modification in nucleolar dominance. Genes Dev 11:2124–2136
Maass N, Biallek M, Rosel F, Schem C, Ohike N, Zhang M, Jonat W, Nagasaki K (2002) Hypermethylation and histone deacetylation lead to silencing of the maspin gene in human breast cancer. Biochem Biophys Res Commun 297:125–128
Fournier C, Goto Y, Ballestar E, Delaval K, Hever AM, Esteller M, Feil R (2002) Allele-specific histone lysine methylation marks regulatory regions at imprinted mouse genes. EMBO J 21:6560–6570
Bannister AJ, Kouzarides T (2004) Histone methylation: recognizing the methyl mark. Methods Enzymol 376:269–288
Schotta G, Lachner M, Peters AH, Jenuwein T (2004) The indexing potential of histone lysine methylation. Novartis Found Symp 259:22–37 (discussion 37–47, 163–169)
Peters AH, Schubeler D (2005) Methylation of histones: playing memory with DNA. Curr Opin Cell Biol 17:230–238
Kondo Y, Shen L, Issa JP (2003) Critical role of histone methylation in tumor suppressor gene silencing in colorectal cancer. Mol Cell Biol 23:206–215
Fujita H, Fujii R, Aratani S, Amano T, Fukamizu A, Nakajima T (2003) Antithetic effects of MBD2a on gene regulation. Mol Cell Biol 23:2645–2657
Li J, Lin Q, Yoon HG, Huang ZQ, Strahl BD, Allis CD, Wong J (2002) Involvement of histone methylation and phosphorylation in regulation of transcription by thyroid hormone receptor. Mol Cell Biol 22:5688–5697
Shi Y, Lan F, Matson C, Mulligan P, Whetstine JR, Cole PA, Casero RA, Shi Y (2004) Histone demethylation mediated by the nuclear amine oxidase homolog LSD1. Cell 119:941–953
Hendrich B, Bird A (1998) Identification and characterization of a family of mammalian methyl-CpG binding proteins. Mol Cell Biol 18:6538–6547
Wade PA (2001) Methyl CpG-binding proteins and transcriptional repression. Bioessays 23:1131–1137
Nan X, Bird A (2001) The biological functions of the methyl-CpG-binding protein MeCP2 and its implication in Rett syndrome. Brain Dev 23(Suppl 1):S32–S37
Shahbazian MD, Antalffy B, Armstrong DL, Zoghbi HY (2002) Insight into Rett syndrome: MeCP2 levels display tissue- and cell-specific differences and correlate with neuronal maturation. Hum Mol Genet 11:115–124
Hite KC, Adams VH, Hansen JC (2009) Recent advances in MeCP2 structure and function. Biochem Cell Biol 87:219–227
Urdinguio RG, Sanchez-Mut JV, Esteller M (2009) Epigenetic mechanisms in neurological diseases: genes, syndromes, and therapies. Lancet Neurol 8:1056–1072
Brero A, Easwaran HP, Nowak D, Grunewald I, Cremer T, Leonhardt H, Cardoso MC (2005) Methyl CpG-binding proteins induce large-scale chromatin reorganization during terminal differentiation. J Cell Biol 169:733–743
Collins AL, Levenson JM, Vilaythong AP, Richman R, Armstrong DL, Noebels JL, Sweatt JD, Zoghbi HY (2004) Mild overexpression of MeCP2 causes a progressive neurological disorder in mice. Hum Mol Genet 13:2679–2689
Bracaglia G, Conca B, Bergo A, Rusconi L, Zhou Z, Greenberg ME, Landsberger N, Soddu S, Kilstrup-Nielsen C (2009) Methyl-CpG-binding protein 2 is phosphorylated by homeodomain-interacting protein kinase 2 and contributes to apoptosis. EMBO Rep 10:1327–1333
Fujita N, Shimotake N, Ohki I, Chiba T, Saya H, Shirakawa M, Nakao M (2000) Mechanism of transcriptional regulation by methyl-CpG binding protein MBD1. Mol Cell Biol 20:5107–5118
Jorgensen HF, Ben-Porath I, Bird AP (2004) Mbd1 is recruited to both methylated and nonmethylated CpGs via distinct DNA binding domains. Mol Cell Biol 24:3387–3395
Nguyen CT, Weisenberger DJ, Velicescu M, Gonzales FA, Lin JC, Liang G, Jones PA (2002) Histone H3-lysine 9 methylation is associated with aberrant gene silencing in cancer cells and is rapidly reversed by 5-aza-2′-deoxycytidine. Cancer Res 62:6456–6461
Liang G, Lin JC, Wei V, Yoo C, Cheng JC, Nguyen CT, Weisenberger DJ, Egger G, Takai D, Gonzales FA, Jones PA (2004) Distinct localization of histone H3 acetylation and H3-K4 methylation to the transcription start sites in the human genome. Proc Natl Acad Sci USA 101:7357–7362
Acknowledgments
The authors would like thank Mrs. Lynn Caflisch for her strong secretarial support and Mrs. Ying Zhao for her superb technical assistance. Shi-Wen Jiang is supported by the Distinguished Cancer Scholarship of the Georgia Research Alliance (GRA). This work was partially funded by research grants from the National Institute of Health (NIH) (R01 HD 41577, Shi-Wen Jiang); the NIH/National Cancer Institute (NCI)-MD Anderson Uterine Cancer SPORE (Jinping Li, Shi-Wen Jiang); the NIH K12 training program (Sean Dowdy, Shi-Wen Jiang); a research grant from Merck Pharmaceiticals (Sean Dowdy, Shi-Wen Jiang); the research supplement from the Department of Obstetrics and Gynecology, Mayo Clinic and Mayo Medical School (Shi-Wen Jiang); the National Natural Science Foundation of China 81200420 and the Yantai Science Development Fund 2011219 (Yongli Chu); the Shangdong Natural Science Foundation ZR2012HL03 and 2011YD21014 (Yanlin Wang); the research start-up from Mercer University School of Medicine (Jinping Li); and seed grants from Mercer University (Jinping Li, Shi-Wen Jiang).
Conflict of interest
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Y. Chu, Y. Wang, G. Zhang and H. Chen contributed equally.
Rights and permissions
About this article
Cite this article
Chu, Y., Wang, Y., Zhang, G. et al. Chromatin composition alterations and the critical role of MeCP2 for epigenetic silencing of progesterone receptor-B gene in endometrial cancers. Cell. Mol. Life Sci. 71, 3393–3408 (2014). https://doi.org/10.1007/s00018-014-1580-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-014-1580-9