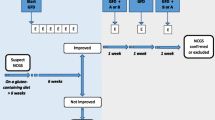
Abstract
Introduction
Food intolerance/malabsorption is caused by food ingredients, carbohydrates (mainly lactose and fructose), proteins (gluten), and biogenic amines (histamine) which cause nonspecific gastrointestinal and extra-intestinal symptoms. Here we focus on possible etiologic factors of intolerance/malabsorption especially in people with non-celiac gluten sensitivity (NCGS) or the so-called people without celiac disease avoiding gluten (PWCDAG) and histamine intolerance.
Methods
Recognizing the recently described symptoms of NCGS (PWCDAG) we review correlations and parallels to histamine intolerance (HIT).
Results
We show that intestinal and extra-intestinal NCGS (PWCDAG) symptoms are very similar to those which can be found in histamine intolerance.
Conclusions
After a detailed diagnostic workup for all possible etiologic factors in every patient, a targeted dietary intervention for single or possibly combined intolerance/malabsorption might be more effective than a short-term diet low in fermentable oligo-, di- and monosaccharides and polyols (FODMAP) or the untargeted uncritical use of gluten-free diets.
Similar content being viewed by others
References
Ortolani C, Pastorello EA. Food allergies and food intolerances. Best Pract Res Clin Gastroenterol. 2006;20:467–83.
Lomer MCE. Review article: the aetiology, diagnosis, mechanisms and clinical evidence for food intolerance. Aliment Pharmacol Ther. 2015;41:262–75.
Enko D, Meinitzer A, Mangge H, Kriegshäuser G, Halwachs-Baumann G, Reininghaus EZ, et al. Concomitant prevalence of low serum diamine oxidase activity and carbohydrate malabsorption. Can J Gastroenterol Hepatol. 2016;2016:4893501.
Valenti S, Corica D, Ricciardi L, Romano C. Gluten-related disorders: certainties, questions and doubts. Ann Med. 2017. https://doi.org/10.1080/07853890.2017.1325968.
Fasano A, Sapone A, Zevallos V, Schuppan D. Non-celiac gluten sensitivity. Gastroenterology. 2015;148:1195 – 204.
Czaja-Bulsa G. Non coeliac gluten sensitivity—a new disease with gluten intolerance. Dig Dis Sci. 2015;34:189 – 94.
Tavakkoli A, Lewis SK, Tennyson CA, Lebwohl B, Green PHR. Characteristics of patients who avoid wheat and/or gluten in the absence of celiac disease. Dig Dis Sci. 2014:59:1255–61.
van Gils T, Nijeboer P, IJssennagger CE, Sanders DS, Mulder CJJ, Bouma G. Prevalence and characterization of self-reported gluten sensitivity in the Netherlands. Nutrients. 2016;8:714.
Ontiveros N, López-Gallardo JA, Vergara-Jiménez MJ, Cabrera-Chávez F. Self-reported prevalence of symptomatic adverse reactions to gluten and adherence to gluten-free diet in an adult Mexican population. Nutrients. 2015;7:6000–15.
Choung RS, Unalp-Arida A, Ruhl MD, Brantner CE, Everhart TL, Murray JE. JA. Less hidden celiac disease but increased gluten avoidance without a diagnosis in the United States: Findings from the National Health and Nutrition Examination Surveys from 2009 to 2014. Mayo Clin Proc. 2017;92:30 – 3.
El-Salhy M, Hatlebakk JG, Gilja OH, Hausken T. The relation between celiac disease, nonceliac gluten sensitivity and irritable bowel syndrome. Nutr J. 2015;14:92.
Verdu EF, Armstrong D, Murray JA. Between celiac disease and irritable bowel syndrome: the “no man’s land” of gluten sensitivity. Am J Gastroenterol. 2009;104:1587–94.
Zevallos VF, Raker V, Tenzer S, Jimenez-Calvente C, Ashfaq-Khan M, Rüssel N, et al. Nutritional wheat amylase-trypsin inhibitors promote intestinal inflammation via activation of myeloid cells. Gastroenterology. 2017;152:1100–13.
Elli L, Branchi F, Tomba C, Villalta D, Norsa L, Ferretti F, et al. Diagnosis of gluten related disorders: Celiac disease, wheat allergy and non-celiac gluten sensitivity. World J Gastroenterol. 2015;21:7110–9.
De Giorgio R, Volta U, Gibson PR. Sensitivity to wheat, gluten and FODMAPs in IBS: facts or fiction? Gut. 2016;65:169–78.
Zanwar VG, Pawar SV, Gambhire PA, Jain SS, Surude RG, Shah VB, et al. Symptomatic improvement with gluten restriction in irritable bowel syndrome: a prospective, randomized, double blinded placebo controlled trial. Intest Res. 2016;14:343 – 50.
Tanner GJ, Blundel MJ, Colgrave ML, Howitt CA. Creation of the first ultra-low gluten barley (Hordeum vulgare L.) for coeliac and gluten-intolerant populations. Plant Biotechnol J. 2016;14:1139–50.
Low gluten diets may be associated with higher risk of type 2 diabetes. American Heart Association Meeting Report Presentation 11. http://newsroom.heart.org/news/low-gluten-diets-may-be-associated-with-higher-risk-of-type-2-diabetes?preview=b2db. Accessed Nov 2017.
Kovacova-Hanuskova E, Buday T, Gavliakova S, Plevkova J. Histamine, histamine intoxication and intolerance. Allergol Immunopathol (Madr). 2015;43:498–506.
Qi W, Hou LH, Guo HL, Wang CL, Fan ZC, Liu JF, et al. Effect of salt-tolerant yeast of Candida versatilis and Zygosaccharomyces rouxii on the production of biogenic amines during soy sauce fermentation. J Sci Food Agric. 2014;94:1537–42.
Verheyen C, Albrecht A, Herrmann J, Strobl M, Jekle M, Becker T. The contribution of glutathione to the destabilizing effect of yeast on wheat dough. Food Chem. 2015;173:243–9.
Chefkoch.de. http://www.chefkoch.de/rs/s60/bulgur/Rezepte.html; 593 Rezepte mit Bulgur. Accessed June 2017.
Kohn JB. Is there a diet for histamine intolerance? J Acad Nutr Diet. 2014;114:1860.
San Mauro Martin I, Brachero S, Garicano Vilar E. Histamine intolerance and dietary management: a complete review. Allergol Immunopathol (Madr). 2016;44:475 – 83.
Maintz L, Novak N. Histamine and histamine intolerance. Am J Clin Nutr. 2007;85:1185–96.
Smolinska S, Jutel M, Crameri R, O’Mahony L. Histamine and gut mucosal immune regulation. Allergy. 2014;69:273–81.
García-Martín E, Martínez C, Serrador M, Alonso-Navarro H, Ayuso P, Navacerrada F, et al. Diamine oxidase rs10156191 and rs2052129 variants are associated with the risk for migraine. Headache. 2015;55:276 – 86.
Cacabelos R, Torrellas C, Fernández-Novoa L, López-Muñoz F. Histamine and immune biomarkers in CNS disorders. Mediators Inflamm. 2016;2016:1924603.
Romero SA, Hocker AD, Mangum JE, Luttrell MJ, Turnbull DW, Struck AJ, et al. Evidence of a broad histamine footprint on the human exercise transcriptome. J Physiol. 2016;594:5009–23.
Worm M, Fiedler EM, Dölle S, Schink T, Hemmer W, Jarisch R, et al. Exogenous histamine aggravates eczema in a subgroup of patients with atopic dermatitis. Acta Derm Venerol. 2009;89:52–6.
Neumann D. Role of the histamine H4-receptor in bronchial asthma. Handb Exp Pharmacol. 2017;241:347 – 59.
Hudak L, Jaraisy A, Haj S, Muhsen K. An updated systematic review and meta-analysis on the association between Helicobacter pylori infection and iron deficiency anemia. Helicobacter. 2017;22:e12330.
Mentis A, Lehours P, Francis Megraud F. Epidemiology and diagnosis of Helicobacter pylori infection. Helicobacter. 2015;20(Suppl. 1):1–7.
Jones BL, Kearns GL. Histamine. New thoughts about a familiar mediator. Clin Pharm Therap. 2011;89:189 – 97.
Schwelberger HG. Histamine intolerance: a metabolic disease? Inflamm Res. 2010;59(Suppl 2):S219–21.
Ji Y, Sakata Y, Li X, Zhang C, Yang Q, Xu M, et al. Lymphatic diamine oxidase secretion stimulated by fat absorption is linked with histamine release. Am J Physiol Gastrointest Liver Physiol. 2013;304:G732-G740.
Wollin A, Wang X, Tso P. Nutrients regulate diamine oxidase release from intestinal mucosa. Am J Physiol. 1988;275:R969-R975.
Elsenhans B, Hunder G, Strugala G, Schümann K. Longitudinal pattern of enzymatic and absorptive functions in the small intestine of rats after short-term exposure to dietary cadmium chloride. Arch Environ Contam Toxicol. 1999;36:341–6.
Maintz L, Yu CF, Rodríguez E, Baurecht H, Bieber T, Illig T, et al. Association of single nucleotide polymorphisms in the diamine oxidase gene with diamine oxidase serum activities. Allergy. 2011;66:893–902.
Petersen J, Raithel M, Schwelberger HG. Characterisation of functional polymorphisms of the human diamine oxidase gene. Inflamm Res. 2005;54(Suppl 1):S58–9.
Sadek B, Stark H. Cherry-picked ligands at histamine receptor subtypes. Neuropharmacology. 2015;106:56–73.
Mušič E, Korošec P, Šilar M, Adamič K, Košnik M, Rijavec M. Serum diamine oxidase activity as a diagnostic test for histamine intolerance. Wien Klin Wochenschr. 2013;125:239 – 43.
Rosell-Camps A, Zibetti S, Pérez-Esteban G, Vila-Vidal M, Ferrés-Ramis L, García-Teresa-García E. Histamine intolerance as a cause of chronic digestive complaints in pediatric patients. Rev Esp Enferm Dig. 2013;105:201–6.
Lionetti E, Pulvirenti A, Vallorani M, Catassi G, Verma AK, Gatti S, Catassi C. Re-challenge studies in non-celiac gluten sensitivity: a systematic review and meta-analysis. Front Physiol. 2017;8:621.
Casellas F, Aparici A, Casaus M, Rodríguez P, Malagelada JR. Subjective perception of lactose intolerance does not always indicate lactose malabsorption. Clin Gastroenterol Hepatol. 2010;8:581–6.
Enko D, Rezanka E, Stolba R, Halwachs-Baumann G. Lactose malabsorption testing in daily clinical practice: a critical retrospective analysis and comparison of the hydrogen/methane breath test and genetic test (c/t-13910 polymorphism) results. Gastroenterol Res Pract. 2014;2014:464382.
Reese I, Ballmer-Weber B, Beyer K, Fuchs T, Kleine-Tebbe J, Klimek L, et al. German guideline for the management of adverse reactions to ingested histamine. Allergo J Int. 2017;26:72–9.
Pimentel M, Mathur R, Chang C. Gas and the microbiome. Curr Gastroenterol Rep. 2013;15:356.
Scott KP, Gratz SW, Sheridon PO, Flint HJ, Duncan SH. The influence of diet on the gut microbiota. Pharmacol Res. 2013;69:52–60.
Gibson PR, Newnham E, Barrett JS, Shepherd SJ, Muir JG. Review article: fructose malabsorption and the bigger picture. Aliment Pharm Ther. 2007;25:349 – 63.
Hovde O, Farup PG. A comparison of diagnostic tests for lactose malabsorption—which one is the best? BMC Gastroenterol. 2009;9:82.
Goebel-Stengel M, Stengel A, Schmidtmann M, van der Voort I, Kobelt P, Mönnikes H. Unclear abdominal discomfort: Pivotal role of carbohydrate malabsorption. J Neurogastroenterol Motil. 2014;20:228–35.
Catassi G, Lionetti E, Gatti S, Catassi C. The low FODMAP diet: many question marks for a catchy acronym. Nutrients. 2017;9:292.
Järvelä I, Torniainen S, Kolho KL. Molecular genetics of human lactase deficiencies. Ann Med. 2009;41:568 – 75.
Hunt KA, van Heel DA. Recent advances in coeliac disease genetics. Gut. 2009;58:473–6.
Patel C, Douard V, Yu S, Tharabenjasin P, Gao N, Ferraris RP. Fructose-induced increases in expression of intestinal fructolytic and gluconeogenic genes are regulated by GLUT5 and KHK. Am J Physiol Regul Integr Comp Physiol. 2015;309:R499–R509.
Schnedl WJ, Enko D, Mangge H, Schenk M, Lackner S, Holasek SJ. Bening pancreatic hyperenzymemia (Gullo syndrome), histamine intolerance, and carbohydrate malabsorption. Proc (Bayl Univ Med Cent). 2017;30:177–8.
Schnedl WJ, Lipp RW, Wallner-Liebmann S, Kalmar P, Szolar DH, Mangge H. Primary epiploic appendagitis and fructose intolerance. Eur J Clin Nutr. 2014;68:1359–61.
Albenberg LG, Wu GD. Diet and the intestinal microbiome: Associations, functions and implications for health and disease. Gastroenterology. 2014;146:1564–72.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Wolfgang J. Schnedl received speaking honoraria from Sciotec. The other authors declare no competing interests.
Additional information
Responsible Editor: Artur Bauhofer.
Rights and permissions
About this article
Cite this article
Schnedl, W.J., Lackner, S., Enko, D. et al. Non-celiac gluten sensitivity: people without celiac disease avoiding gluten—is it due to histamine intolerance?. Inflamm. Res. 67, 279–284 (2018). https://doi.org/10.1007/s00011-017-1117-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-017-1117-4