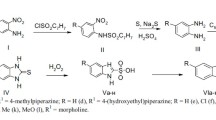
Abstract
New organic nitrate esters, derived from structurally different (cyclo)aliphatic templates, were synthesized and pharmacologically investigated. Theirin vitro vascular smooth muscle relaxing activities and, occasionally,in vivo haemodynamic profiles were studied and compared to those of the clinically important nitrates, glyceryl trinitrate, isosorbide dinitrate and isosorbide-5-mononitrate. A number of compounds appeared to be even more potent than glyceryl trinitrate. Qualitative structure-activity relationships within the series of new compounds are discussed. In flexiblen-alkylene dinitrates, lipophilicity as well as chain length appears to affectin vitro activity. In semi-rigid cyclohexylene dinitrates, the number of atoms between and the configuration of the nitrate groups may play an important role. Finally, in cycloalkylene mononitrates neither the number of ring carbon atoms nor the lipophilicity clearly affects thein vitro activity. It is suggested that, apart from a limited involvement of compound lipophilicity, other factors such as differences in enzymatic conversion to a common putative bioactive species, nitric oxide, are responsible for the observed differences in activity.
Similar content being viewed by others
References
Balard JM. Premier mémoire sur l'alcohol amylique. Ann Clin Phys 1844;12:294–330.
Sobrero A. Sur plusieurs composés détonants producé avec l'acide nitrique et le sucre, la dextrine, la lactine, la mannite et la glycerine. R Acad Sci (Paris) 1847;24:247–8.
Krantz JC, Carr CJ, Forman SE, Ellis FW. Alkylnitrites III. A pharmacologic study of a new series of organic nitrates. J Pharmacol Exp Ther 1939;67:187–90.
Goldberg L. Pharmacological properties of sorbide dinitrate. Acta Phys Scand 1948;15:173–87.
Snatzke G, Laurent H, Wiechert R. Circular dichroism. XXXII. Cotton effect of nitryloxy steroids. Tetrahedron 1969;25:761–9.
Bron J, Sterk GJ, van der Werf JF, Timmerman H. Pharmaceutical composition having relaxing activity which contains a nitrate ester as active substance. Eur Pat Appl 0 359 335 (Bulletin 90-085143/12).
Rekker RF, de Kort HM. The hydrophobic fragmental constant; an extension to a thousand data point set. Eur J Med Chem 1979;14:479–88.
Wildfeuer A, Laufen H, Leitold M. Pharmakologie von Isosorbiddinitrat nach transdermaler Applikation. Arznei mittelforschung/Drug Res 1985;35:1289–91.
Riseman JEF, Altman GE, Koretsky S. Nitroglycerin and other nitrites in the treatment of angina pectoris. Comparison of six preparations and four routes of administration. Circulation 1958;17:22–39.
Noack E. Investigation on structure-activity relationship in organic nitrates. Methods Find Exp Clin Pharmacol 1984;6:583–6.
Stoss P, Erhardt E. Bicycloalkanol-nitrate. Arch Pharm 1987;320:621–4.
Ignarro LJ, Lippton H, Edwards JC, Baricos WH, Hyman AI, Kadowitz PJ, et al. Mechanism of vascular smooth muscle relaxation by organic nitrates, nitrites, nitroprusside and nitric oxide: evidence for the involvement of S-nitrosothiols as active intermediates. J Pharmacol Exp Ther 1981;218:739–49.
Tzeng TB, Fung HL. Structure-activity relationship of organic nitrates: an exploratory hypothesis via molecular models. Med Hypotheses 1992;37:58–62.
Van Woerkens LJ, van der Giessen WJ, Verdouw PD. Cardiovascular profile of 5 novel nitrate esters: a comparative study with nitroglycerin in pigs with and without left ventricular dysfunction. Br J Pharmacol 1991;104:7–14.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bron, J., Sterk, G.J., van der Werf, J.F. et al. Synthesis and pharmacology of a series of new organic nitrate esters. Pharm World Sci 17, 120–125 (1995). https://doi.org/10.1007/BF01872388
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01872388