Abstract
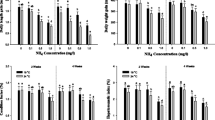
Biomolecule content, dry weight, and wet weight of Atlantic salmon,Salmo salar L., were monitored during 60-day exposure to aluminum, which began before hatch and extended to about the end of the alevin stage. The Atlantic salmon exposed to aluminum concentrations of 0, 33, 71, 124, or 264 μg/L at pH 5.5 for 60 days were evaluated for growth, survival, and biochemical effects on days 15, 30, and 60. Growth and survival were significantly reduced at the two highest aluminum concentrations by day 60. RNA and DNA were the most sensitive biomolecules monitored and were significantly reduced at the three highest aluminum concentrations by day 60. RNA∶protein and RNA∶DNA ratios were no more responsive than growth and survival. The lipid content of salmon was not significantly affected by exposure to aluminum. It was concluded that biomolecule content or ratios were not particularly sensitive indicators of growth effects in Atlantic salmon exposed to aluminum.
Similar content being viewed by others
References
Barron MG, Adelman IR (1984) Nucleic acid, protein content, and growth of larval fish sublethally exposed to various toxicants. Can J Fish Aquat Sci 41:141–150
— (1985) Temporal characterization of growth of fathead minnow(Pimephales promelas) larvae during sublethal hydrogen cyanide exposure. Comp Biochem Physiol 81C:341–344
Bitman J, Wood DL (1982) Detection methods for lipids on TLC. In: Touchstone JC (ed) Advances in thin layer chromatography. John Wiley and Sons, New York, NY, pp 187–218
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Buckler DR (1987) Comparative toxicity and availability of dissociable compounds to fishes as effected by ambient pH. PhD dissertation. Utah State University, Logan, Utah
Buckley LJ (1979) Relationships between RNA-DNA ratio, prey density, and growth rate in Atlantic cod (Gadus morhua) larvae. J Fish Res Board Can 36:1497–1502
— (1984) RNA-DNA ratio: An index of larval fish growth in the sea. Mar Biol 80:291–298
Bulow FJ (1970) RNA-DNA ratios as indicators of recent growth rates of a fish. J Fish Res Board Can 27:2343–2349
Bulow FJ, Zeman ME, Winningham JR, Hudson WF (1981) Seasonal variations in RNA-DNA ratios and in indicators of feeding, reproduction energy storage, and condition in a population of blue-gill,Lepomis macrochirus Rafinesque. J Fish Biol 18:237–244
Burton K (1956) A study of the conditions and mechanism of the diphenylamine reaction for the colorimetric estimation of deoxyribonucleic acid. Biochem J 62:315–323
Cleveland L, Little EE, Hamilton SJ, Buckler DR, Hunn JB (1986) Interactive toxicity of aluminum and acidity to early life stages of brook trout. Trans Am Fish Soc 115:610–620
Daye PG, Garside ET (1979) Development and survival of embryos and alevins of the Atlantic salmon,Salmo solar L., continuously exposed to acidic levels of pH, from fertilization. Can J Zool 57:1713–1718
Haines TA (1973) An evaluation of RNA-DNA ratio as a measure of long-term growth in fish populations. J Fish Res Board Can 30:195–199
— (1980) Seasonal patterns of muscle RNA-DNA ratio and growth in black crappie,Pomoxis nigromaculatus. Environ Biol Fishes 5:67–70
Hayes FR (1949) The growth, general chemistry, and temperature relations of salmonid eggs. Quart Rev Biol 24:281–308
Knowles CO, McKee MJ (1987) Protein and nucleic acid content inDaphnia magna during chronic exposure to cadmium. Ecotoxicol Environ Safety 13:290–300
Lowry OH, Rosebrough NJ, Farr Al, Randall RJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193–275
McKee MJ, Knowles CO (1986) Effects of fenvalerate on biochemical parameters, survival, and reproductionof Daphnia magna. Ecotoxicol Environ Safety 12:70–84
Miller GL (1959) Protein determination for large numbers of samples. Anal Chem 31:964
Mount DI, Brungs WA (1967) A simplified dosing apparatus for fish toxicology studies. Water Res 1:21–29
Munro HN, Fleck A (1966) Recent developments in the measurement of nucleic acids in biological materials. A supplementary review. Analyst 91:78–88
Odonnell AR, Mance G, Norton R (1984) A review of the toxicity of aluminum in freshwater. Water Research Centre, Technical Report TR197, WRC Environment, Marlow, Bucks, SL7ZHD
Pande SV, Khan RP, Venkitasubramanian TA (1963) Micro-determination of lipids and serum total fatty acids. Anal Biochem 6:415–423
Roe JH, Dailey RE (1966) Determination of glycogen with the anthrone reagent. Anal Biochem 15:245–250
SAS Institute, Inc (1982a) SAS users guide: Basics, 1982 edition. SAS Institute, Cary, NC
— (1982b) SAS users guide: Statistics, 1982 edition. SAS Institute, Cary, NC
Schneider WC (1945) Phosphorus compounds in animal tissue. I. Extraction and estimation of desoxypentose nucleic acid and of pentose nucleic acid. J Biol Chem 161:293–303
Wilder IB, Stanley JG (1983) RNA-DNA ratio as an index to growth in salmonid fishes in the laboratory and in streams contaminated by carbaryl. J Fish Biol 22:165–172
Author information
Authors and Affiliations
Additional information
Contribution from the Missouri Agricultural Experiment Station, Columbia. Journal Series No. 10319. Reference to trade names or manufacturers does not imply government endorsement of commercial products.
Rights and permissions
About this article
Cite this article
McKee, M.J., Knowles, C.O. & Buckler, D.R. Effects of aluminum on the biochemical composition of Atlantic salmon. Arch. Environ. Contam. Toxicol. 18, 243–248 (1989). https://doi.org/10.1007/BF01056209
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01056209