Abstract
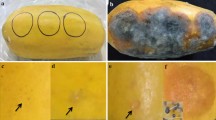
Plants ofLycopersicon esculentum (grown in greenhouses) andAnona cherimolia cultivated in southeastern Spain were examined for the presence of trypanosomatid flagellates. Kinetoplastid protozoa were found in the fruits but not in the phloem or other plant tissues. Parasites were detected from the onset of fruiting. Isolates were adapted to in vitro culturing in monophase media. The form and the structural organization was studied by scanning and transmission electron microscopy. The parasites showed an ultrastructural pattern similar to that of other species of the genusPhytomonas. In tomatoes experimentally inoculated with flagellates cultivated in vitro, we observed that the parasites did not lose their infectious capacity. Three strains of trypanosomatids of the genusPhytomonas, isolated from different species ofEuphorbia (E. characias andE. hyssopifolia) and fromCocos nucifera, were compared with our isolates by lectinagglutination tests. Our isolates were different from the two strains isolated fromEuphorbia, but with this technique we could not differentiate our isolates from those of the coconut, nor could we differentiate between the isolates, their ultrastructural similarity together with their similar behavior in the lectin-agglutination test suggesting that these isolates have a common origin.
Similar content being viewed by others
References
Attias M, Roitman I, Camargo EP, Dollet M, Souza W de (1988) Comparative analysis of fine structure of four isolates of trypanosomatids of the genusPhytomonas. J Protozool 35:365–370
Camargo EP, Kastelein P, Roitman I (1990) Trypanosomatid parasites of plants (Phytomonas). Parasitol Today 6:22–25
Conchon I, Campaner M, Sbravate C, Camargo EP (1989) Trypanosomatids, other thanPhytomonas spp., isolated and cultured from fruit. J Protozool 36:412–414
Dollet M (1984) Plant diseases caused by flagellate protozoa (Phytomonas). Annu Rev Phytopathol 22:115–132
Dollet M, Gargani D (1989) Mixed infection ofAsclepias curassavica L. in Ecuador:Rhabdovirus-like particles andPhytomonas sp. J Phytopathol 125:269–273
Donovan C (1909) Kala azar in Madras, especially with regard to its connexion with the dog and the bug (Cnorhinus). Lancet 177:1495–1496
Forbes MS, Plantholt BA, Sperelakis N (1977) Cytochemical staining procedures selective for sarcotubular systems of muscle: modifications and applications. J Ultrastruct Res 60:306–327
Gibbs AJ (1957)Leptomonas serpens n. sp. parasitic in the digestive tract and salivary glands ofNezara viridula (Pentatomidae) and in the sap ofSolanum lycopersicum (tomato) and other plants. Parasitology 47:297–303
Jankevicius JV, Jankevicius SI, Campaner M, Conchon I, Maeda LA, Camargo EP (1989) Life cycle and culturing ofPhytomonas serpens (Gibbs), a trypanosomatid parasite of tomatoes. J Protozool 36:265–271
Kastelein P, Camargo EP (1990) Trypanosomatid protozoa in fruit of solanaceae in southeastern Brazil. Mem Inst Oswaldo Cruz 85:413–417
Kitajima EW, Vainstein MH, Silveira JSM (1986) Flagellate protozoan associated with poor development of the root system of cassava in the Espiritu Santo State, Brazil. Phytopathology 96:638–642
Lafont A (1909) Sur la présence d'un parasite de la classe des flagellés dans le latex de l'Euphorbia pilluliferal. C R Soc Biol 66:1011–1013
Linder JC, Staehelim LA (1980) The membrane lattice: a novel organelle of the trypanosomatid flagellateLeptomonas collosoma. J Ultrastruct Res 72:200–205
Menara A, Dollet M, Gargani D, Louise C (1988) Culture in vitro sur cellules d'invertébrés, desPhytomonas sp. (trypanosomatidae) associés au Hartrot, maladie du cocotier. C R Acad Sci [III] 307:597–602
Opperdoes FR (1987) Compartmentation of carbohydrate metabolism in trypanosomes. Annu Rev Microbiol 41:127–152
Parthasarathy MV, Van Slobbe WG, Soudant C (1976) Trypanosomatid flagellate in the phloem of diseased coconut palm. Science 192:1346–1348
Petry K, Gargani D, Baltz T, Kastelein P, Dollet M (1985) Use of monoclonal antibodies for different isolates ofPhytomonas (plant trypanosomatids). J Phytopathol 126:59–68
Petry K, Schottelius J, Dollet M (1987) Differentiation ofPhytomonas sp. lower trypanosomatids (Herpetomonas, Crithidia) by agglutination test with lectins. Parasitol Res 74:1–4
Riou JF, Dollet M, Ahomadgbe JC, Coulaud D, Riou G (1987) Characterization ofPhytomonas sp. kinetoplast DNA, a plant pathogenic trypanosomal species. FEBS Lett 213:304–308
Sánchez-Moreno M, Lasztity D, Coppens I, Opperdoes FR (1992) Characterization of carbohydrate metabolism and demonstration of glycosomes in aPhytomonas sp. isolated fromEuphorbia characias. Mol Biochem Farasitol 54:185–200
Santos-Padron T, Souza W de (1982) Fine structure and cytochemistry of peroxisomes (microbodies) inLeptomonas samueli. Cell Tissue Res 222:153–158
Souza W de (1984) Cell biology ofTrypanosoma cruzi. Int Rev Cytol 86:197–283
Stahel G (1931) Zur Kenntnis der Siebröhr-Krankheit (Phoemnekrose) des Kaffeebaumes in Surinam. Phytopathol 1-Z 4:65–82
Wallace FG, Camargo EP, McGhee RB, Roitman I (1983) Guidelines for the description of new species of lower trypanosomatids. J Protozool 30:308–313
Zubiaur F, Alonso P (1985) Behaviour ofNaegleria lavaniensis andN. gruberi against different lectins. Protistologica 2:267–272
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sanchez-Moreno, M., Fernandez-Becerra, C., Mascaro, C. et al. Isolation, in vitro culture, ultrastructure study, and characterization by lectin-agglutination tests of Phytomonas isolated from tomatoes (Lycopersicon esculentum) and cherimoyas (Anona cherimolia) in southeastern Spain. Parasitol Res 81, 575–581 (1995). https://doi.org/10.1007/BF00932024
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00932024