Abstract
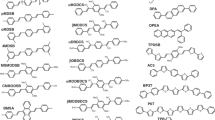
We have shown that the lasing properties of naphthyl 1,3,4 oxadiazole derivatives are directly related to the position of the forbidden transitionS 0→1 L b of naphthalene with respect to the first allowed transitions. The combination of theoretical and experimental results allows us to predict which compounds are most likely to exhibit a laser effect according to the nature and the position of their substituants. We have successfully applied this approach to the following compounds: αNPD, βNPD, αNND, βNND, βNBD, and αNBD. In particular we reported the first observation of a laser effect for αNBD and βNBD in the UV at 3830 Å and 3758 Å.
Similar content being viewed by others
References
K.H.Drexage: InDye Lasers (Springer, Berlin, Heidelberg, New York 1973)
T.G.Pavlopoulos: IEEE J. Quant. Elect.QE-9, 510 (1973)
C.Rullière: P.H.D. Thesis—Laval University (June 1974)
C.Rullière, M.M.Denariez-Roberge: Can. J. Phys.51, 418 (1973)
C.Rullière, J.C.Rayez, M.M.Denariez-Roberge, J.Joussot-Dubien: InLasers in Physical Chemistry and Biophysics (Elsevier, Amsterdam 1975) p. 117
J.Langelaar: Appl. Phys.6, 61 (1975)
J.Langelaar: InLasers in Physical Chemistry and Biophysics (Elsevier, Amsterdam 1975) p. 89
M.J.Weber, M.Bass: IEEE J. Quant. Electr.QE-4, 175 (1969)
For our knowledge, 9, 10 diphenyl anthracene is the laser molecule which have the longest radiative lifetime τ f ≃ ns [10, 11]
B.G.Huth, G.I.Farmer: IEEE J. Quant. Elect. QE-4, 427 (1968)
I.B.Berlmann:Handbook of Fluorescence Spectra of Aromatic Molecules (Academic Press, New York 1973)
R.Pariser: J. Chem. Phys.24, 250 (1956)
R.Phan Tan Luu, L.Bouscasse, E.J.Vincent, J.Metzger: Bull. Soc. Chim. France 3238 (1967)
C.Rullière, P.C.Roberge: ACFAS Meeting, Quebec (June 1974)
H.W.Furmoto, H.L.Ceccon: IEEE J. Quant. Elect.QE-6, 262 (1970)
C.C.J.Roothaan: Rev. Mod. Phys.23, 69 (1951)
R.Pariser, R.G.Parr: J. Chem. Phys.21, 466, 767 (1953)
N.Mataga, K.Nishimoto: Z. Phys. Chem.12, 335 (1957)
J.Hinze, H.H.Jaffe: J. Am. Chem. Soc.84, 540 (1962)
H.O.Pridchard, H.A.Skinner: J. Inorg. Nocl. Chem.24, 937 (1962)
The oxadiazole ring and the phenyl group are taken in the same plane [14]
R.G.Parr, B.L.Crawford, Jr.: J. Chem. Phys.16, 526 (1948)
M.J.S.Dewar: J. Am. Chem. Soc.74, 3345 (1952)
J.N.Murell:Theory of Electronic Spectra of Organic Molecules (John Wiley, New York 1963)
B.S.Hudson, B.E.Kohler: J. Chem. Phys.59, 4984 (1973)
M.Maeda, Y.Miyazae: Jap. J. Appl. Phys.13, 827 (1974)
G.A.Abakumov, A.P.Somonov, V.V.Fadeev, L.A.Kharitonov, R.V.Khokholov: JETP Lett.9, 9 (1969)
C.Rullière, M.M.Denariez-Roberge: Opt. Comm.7, 166 (1973)
D.Basting, F.P.Schäfer, B.Steyer: Appl. Phys.3, 81 (1974)
C.Rulliere, J.P.Morand, J.Joussot-Dubien: Opt. Comm.15, 263 (1975)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rullière, C., Rayez, JC. Lasing ability of naphthyl 1,3,4 oxadiazole molecules in relation with their structures: Application to the design of new UV dye laser. Appl. Phys. 11, 377–382 (1976). https://doi.org/10.1007/BF00919367
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00919367