Abstract
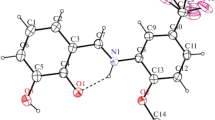
The condensation of 1,2,5-trimethylpiperidine-4-one with phenol and isomeric cresols yields 1,2,5-trimethyl-4-(p-hydroxyphenyl)- and (p-hydroxytolyl)-δ3-tetrahydropyridines, the structure and conformation of which have been studied by proton NMR spectroscopy.
Similar content being viewed by others
Literature Cited
D. M. Zimmerman, R. Nikander, J. S. Horng, and D. T. Wong, Nature, 275, 332 (1978).
M. M. A. Hassan and A. F. Casy, Org. Magn. Reson., 2, 197 (1970).
M. Karplus, J. Chem. Phys., 30, 11 (1959).
M. Barfield and B. Chakrabarti, Chem. Rev., 69, 757 (1969).
F. A. L. Anet and I. Yavari, J. Am. Chem. Soc., 99, 2794 (1977).
M. Anteunis, Bull. Soc. Chim. Belg., 75, 413 (1966).
F. R. Jensen and C. H. Bushweller, Adv. Alicyclic Chem., 3, 139 (1971).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 10, pp. 1367–1370, October, 1986.
Rights and permissions
About this article
Cite this article
Rezakov, V.A., Das, S.K., Fomichev, A.A. et al. 1,2,5-Trimethyl-4-(p-hydroxyaryl)-δ3-tetrahydropyridines and their spatial structure. Chem Heterocycl Compd 22, 1107–1110 (1986). https://doi.org/10.1007/BF00473489
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00473489