Abstract
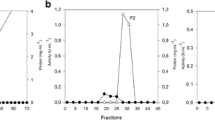
We report the optimum conditions for the degradation of oat spelt arabinoxylan and a preliminary characterisation of the inducible xylan-degrading system of the lignin-degrading white-rot fungus Phanerochaete chrysosporium. Xylanase activity was optimal at pH 5.0 and 50°C; see attached sheet the maximum reaction velocity (Vmax) of the system was 3.86 units (U) mg−1 protein with arabinoxylan as substrate and the substrate concentration giving half Vmax (S0.5) was 0.52 mg ml−1. At concentrations of arabinoxylan greater than 15 mg ml−1 excess substrate inhibition was observed. Xylose at 0.9 mm inhibited activity to the extent of 50%. Xylanase activity increased as a function of the dilution of the enzyme preparation prior to assay. It was resolved into four peaks by using a DEAE-Biogel column; the material in these peaks differed with respect to xylan solubilisation and the formation of reducing sugars. Electrofocusing gels allowed visualisation of several bands of activity corresponding to each peak. The arabinoxylan degradation system of P. chrysosporium is therefore composed of multiple components.
Similar content being viewed by others
References
Bailey M, Puls J, Poutanen K (1991) Purification and properties of two xylanases from Aspergillus oryzae. Biotechnol Appl Biochem 13:380–389
Biely P (1985) Microbial xylanolytic systems. Trends Biotechnol 3:286–290
Birch O (1988) Extracellular enzymes from the lignin-degrading fungus Phanerochaete chrysosporium. Ph.D. Thesis, University of Manchester
Blanchette RA, Abad AR, Cease KR, Farrell RL, Lourien RE, Leather TD (1990) Enzyme immunocytochemistry and ultrastructural localization of cell wall components by enzyme-gold complexes. In: Kirk TK, Chang HM (eds) Biotechnology in pulp and paper manufacture: applications and fundamental investigations. Butterworth Heinemann, Boston, pp 69–81
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:28–254
Comtat J (1983) Isolation, properties and postulated role of some of the xylanases from the basidiomycete Sporotrichum dimorphosporum. Carbohydr Res 118:215–231
Datta A, Battermann A, Kirk T (1991) Identification of a specific manganese peroxidase among ligninolytic enzymes secreted by Phanerochaete chrysosporium during wood decay. Appl Environ Microbiol 57:1453–1460
Dobozi MS, Szakacs G, Bruschi CV (1992) Xulanase activity of Phanerochaete chrysosporium. Appl Environ Microbiol 58:3466–3471
Eriksson KE (1981) Microbial degradation of cellulose and lignin. SCPI International Symposium on Wood and Pulping Chemistry (Stockholm) vol 3, pp 60–65
Eriksson KE, Blanchette R, Ander P (1990) Microbial and enzymatic degradation of wood and wood components. Springer, Berlin Heidelberg New York
Gascon S, Lampen J (1968) Purification of a internal invertase of yeast. J Biol Chem 243:1567–1572
Gilkes NR, Henrissat B, Kilburn DG, Miller RC, Warren RAJ (1991) Domains in microbial β-1,4-glycanases: sequence conservation, function, and enzyme families. Microbiol Rev 55:303–315
John M, Schmidt J (1989) Xylanases and β-xylosidase of Trichoderma lignorum. Methods Enzymol 160:662–671
Johnson WC, Lindsey AJ (1939) An improved universal buffer. Analyst 64:490–492
Jurasek KL, Paice MG (1988) Xylanase A of Schizophyllum commune. Methods Enzymol 160:659–662
Khan AW, Tremblay D, LeDuy A (1986) Assay of xylanase and β-xylosidase activities in bacterial and fungal cultures. Enzyme Microb Technol 8:373–377
Kirk TK, Farrell RL (1987) Enzymatic “combustion”: the microbial degradation of lignin. Annu Rev Microbiol 41:465–505
Kluepfel D, Daigneault N, Morosoli R, Shareck F (1992) Purification and characterization of a new xylanase (xylanase C) produced by Streptomyces lividans 66. Appl Microbiol Biotechnol 36:626–631
Lachke AH (1988) 1,4-β-d-Xylan xylohydrolase of Sclerotium rolfsii. Methods Enzymol 160:679–684
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Llanos-Palop M, Vallés S, Piñaga F, Flors A (1991) Characterization of cellulase and xylanase activities of Clostridium celerecrescens. J Chem Technol Biotechnol 51:105–114
Mason JC, Birch OM, Broda P (1990) Preparation of 14C-radiolabelled lignocelluloses from spring barley of differing maturities and their solubilization by Phanerochaete chrysosporium and Streptomyces cyaneus. J Gen Microbiol 136:227–232
Mishra C, Keskar S, Rao M (1984) Production and properties of extracellular endoxylanase from Neurospora crassa. Appl Environ Microbiol 48:224–228
Mishra C, Forrester I, Kelley B, Burgess R, Leatham G (1990) Lentinula edodes cultures grown on a commercial solid lignocellulose substrate. Appl Microbiol Biotechnol 33:226–232
Mopper R, Gindler J (1973) A new noncorrosive dye reagent for automatic sugar chromatography. Anal Biochem 56:440–442
Morrisey JH (1981) Silver stain for proteins in polyacrylamide gels. A modified procedure with enhanced uniform sensitivity. Anal Biochem 117:307–310
Nakayama M, Tomita Y, Susuki H, Nisizawa K (1976) Partial proteolysis of some cellulase components from Trichoderma viride and the substrate specificity of the modified products. J Biochem 79:955–966
Nawaz M, Gunasekaran M (1988) Effect of peat extract on the hydrolytic enzymes of Phanerochaete chrysosporium. Resour Conserv Recycl 1:197–205
Poutanen K, Puls J (1988) Characteristics of Trichoderma reesei β-xylosidase and its use in the hydrolysis of solubilized xylans. Appl Microbiol Biotechnol 28:425–432
Rapp P, Wagner F (1986) Production and properties of xylan degrading enzymes from Cellulomonas uda. Appl Environ Microbiol 54:746–752
Sinner M, Puls J (1978) Non-corrosive dye reagent for detection of reducing sugars in borate complex ion-exchange chromatography. J Chromatog 156:197–204
Tenkanen M, Puls J, Poutanen K (1992) Two major xylanases of Trichoderma reesei. Enzyme Microb Technol 14:566–574
Tuohy MG, Coughlan MP (1992) Production of thermostable xylan-degrading enzymes by Talaromyces emersonii. Bioresour Technol 39:131–137
Wang P, Broda P (1992) Stable defined substrate for turbidimetric assay of endoxylanases. Appl Environ Microbiol 58:3433–3436
Author information
Authors and Affiliations
Additional information
Correspondence to: P. Broda
Rights and permissions
About this article
Cite this article
Copa-Patiño, J.L., Kim, Y.G. & Broda, P. Production and initial characterisation of the xylan-degrading system of Phanerochaete chrysosporium . Appl Microbiol Biotechnol 40, 69–76 (1993). https://doi.org/10.1007/BF00170431
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00170431