Abstract
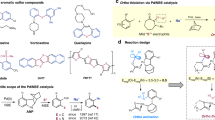
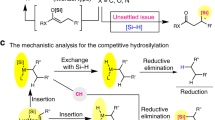
An efficient catalytic system based on easily available palladium acetate was developed for the selective addition of aliphatic thiols to the triple bond of internal alkynes. Formed in situ [M(SR)2] n nanostructured particles were found to be an active form of the catalyst. It was experimentally confirmed for the first time that the most active form of the catalyst for thiol addition to internal alkynes is formed only in the reaction mixture containing the both reactants, namely, alkyne and thiol.
Similar content being viewed by others
References
I. P. Beletskaya, V. P. Ananikov, Chem. Rev., 2011, 111, 1596.
A. Ogawa, Top. Organomet. Chem., 2013, 43, 325.
A. Ishii, N. Nakata, Top. Organomet. Chem., 2013, 43, 21.
P. Bichler, J. A. Love, Top. Organomet. Chem., 2010, 31, 39.
V. P. Ananikov, I. P. Beletskaya, Top. Organomet. Chem., 2013, 43, 1.
V. P. Ananikov, N. V. Orlov, I. P. Beletskaya, Russ. Chem. Bull., Int. Ed., 2005, 54, 576 [Izv. Akad. Nauk. Ser. Khim., 2005, 569].
V. P. Ananikov, N. V. Orlov, M. A. Kabeshov, I. P. Beletskaya, Z. A. Starikova, Organometallics, 2008, 27, 4056.
V. P. Ananikov, D. A. Malyshev, I. P. Beletskaya, G. G. Aleksandrov, I. L. Eremenko, Adv. Synth. Catal., 2005, 347, 1993.
D. A. Malyshev, N. M. Scott, N. Marion, E. D. Stevens, V. P. Ananikov, I. P. Beletskaya, S. P. Nolan, Organometallics, 2006, 25, 4462.
I. Kamiya, E. Nishinaka, A. Ogawa, J. Org. Chem., 2005, 70, 696.
A. Ogawa, T. Ikeda, K. Kimura, T. Hirao, J. Am. Chem. Soc., 1999, 121, 5108.
A. Di Giuseppe, R. Castarlenas, J. J. Pérez-Torrente, M. Crucianelli, V. Polo, R. Sancho, F. J. Lahoz, L. A. Oro, J. Am. Chem. Soc., 2012, 134, 8171.
J. Yang, A. Sabarre, L. R. Fraser, B. O. Patrick, J. A. Love, J. Org. Chem., 2009, 74, 182.
S. Shoai, P. Bichler, B. Kang, H. Buckley, J. A. Love, Organometallics, 2007, 26, 5778.
V. P. Ananikov, D. A. Malyshev, I. P. Beletskaya, G. G. Aleksandrov, I. L. Eremenko, J. Organomet. Chem., 2003, 679, 162.
I. G. Trostyanskaya, I. P. Beletskaya, Synlett, 2012, 535.
U. Koelle, Chr. Rietmann, J. Tjoe, T. Wagner, U. Englert, Organometallics, 1995, 14, 703.
C. Cao, L. R. Fraser, J. A. Love, J. Am. Chem. Soc., 2005, 127, 17614.
V. P. Ananikov, K. A. Gayduk, N. V. Orlov, I. P. Beletskaya, V. N. Khrustalev, M. Yu. Antipin, Chem. Eur. J., 2010, 16, 2063.
V. P. Ananikov, I. P. Beletskaya, Dalton Trans., 2011, 40, 4011.
V. P. Ananikov, I. P. Beletskaya, Organometallics, 2012, 31, 1595.
V. P. Ananikov, N. V. Orlov, S. S. Zalesskiy, I. P. Beletskaya, V. N. Khrustalev, K. Morokuma, D. G. Musaev, J. Am. Chem. Soc., 2012, 134, 6637.
V. P. Ananikov, N. V. Orlov, I. P. Beletskaya, Organometallics, 2007, 26, 740.
V. P. Ananikov, N. V. Orlov, S. S. Zalesskiy, I. P. Beletskaya, Russ. Chem. Bull. (Int. Ed.), 2006, 55, 2109 [Izv. AN. Ser. Khim., 2006, 2030].
A. S. Kashin, V. P. Ananikov, Russ. Chem. Bull. (Int. Ed.), 2011, 60, 2602 [Izv. Akad. Nauk, Ser. Khim., 2011, 2551].
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 1, pp. 0048–0055, January, 2013.
Rights and permissions
About this article
Cite this article
Orlov, N.V., Chistyakov, I.V., Starikova, Z.A. et al. Stereoselective addition of aliphatic thiols to internal alkynes in a catalytic system with palladium “nanosalt” as an active site. Russ Chem Bull 62, 47–54 (2013). https://doi.org/10.1007/s11172-013-0007-3
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-013-0007-3