Abstract
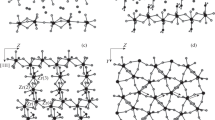
On the basis of precision X-ray diffraction studies of glycinium trifluoroacetate, glycinium monohydrate with l, 5-naphthalenedisulfonic acid, and the 6h-polymorph of glycine, topological analysis of the electron density distribution function in the crystal was carried out. The sublimation energy of glycine topological analysis of the electron density distribution function in the crystal was carried out. The sublimation energy of glycine was estimated and the role of hydrogen bonds in the charge transfer from the anion to the cation was studied. The role of cation—cation and anion—anion contacts in crystal stabilization was considered. Features of the specific interactions C...C, F...O, and F...N and sulfonic acid SO3 - group were analyzed on the basis of the deformation electron density function and the electron localization function (ELF).
Similar content being viewed by others
References
Y. V. Nelyubina, M. Y. Antipin, K. A. Lyssenko, J. Phys. Chem. A., 2007, 111, 1091; (b) V. Luana, A. Costales, P. Mori-Sanchez, A. M. Pendas, J. Phys. Chem. B, 2003, 107, 4912; (c) Y. V. Nelyubina, K. A. Lyssenko, D.G. Golovanov, M. Y. Antipin, Cryst. Eng. Comm., 2007, 9, 991; (d) Y. V. Nelyubina, K. A. Lyssenko, R. G. Kostyanovsky, D. A. Bakulin, M. Y. Antipin, Mendeleev. Commun., 2008, 18, 29; (e) Y. V. Nelyubina, K. A. Lyssenko, V. Yu. Kotov, M. Yu. Antipin, J. Phys. Chem. A, 2008, 112, 8790; (f) Yu. V. Nelyubina, K. A. Lyssenko, A. S. Sigachev, M. Yu. Antipin, A. N. Kravchenko, Izv. Akad. Nauk. Ser. Khim., 2006, 387 [Russ. Chem. Bull., Int. Ed., 2006, 55, 399]; (g) Yu. V. Nelyubina, K. A. Lyssenko, D. G. Golovanov, M. Yu. Antipin, Crysteng. Comm, 2007, 9, 991.
Yu. V. Kotov, S. I. Gorel’skii, Izv. Akad. Nauk. Ser. Khim., 1999, 823 [Russ. Chem. Bull., Int. Ed., 1999, 48, 823].
G. R. Desiraju, Acc. Chem. Res., 2002, 35, 565(a); T. Steiner, Angew. Chem. Int. Ed. Engl., 2002, 41, 48(a).
K. A. Lyssenko, P. Yu. Barzilovich, S. M. Aldoshin, M. Yu. Antipin, Yu. A. Dobrovolsky, Mendeleev Commun., 2008, 18,6, 312; (b) R. Parthasarathi, V. Subramanian, N. Sathyamurthy, J. Phys. Chem. A., 2007, 111, 13287.
K.-D. Kreuer, S. J. Paddison, E. Spohr, M. Schuster, Chem. Rev., 2004, 104, 4637.
A.V. Iogansen, Spectrochimica Acta, 1999, A55, 1585.
E. Espinosa, I. Alkorta, I. Rozas, J. Elguero, E. Molins, Chem. Phys. Lett., 2001, 336, 457.
(a) K. A. Lyssenko, Yu. V. Nelyubina, R. G. Kostyanovsky, M. Yu. Antipin, Chem. Phys. Lett., 2006, 7, 2453; (b) K. A. Lyssenko, A. A. Korlyukov, D. G. Golovanov, S. Yu. Ketkov, M. Yu. Antipin, J. Phys. Chem. A, 2006, 110, 6545; (c) K. A. Lyssenko, A. A. Korlyukov, M. Yu. Antipin, Mendeleev Commun. 2005, 15, 90; (d) I. V. Glukhov, K. A. Lyssenko, A. A. Korlyukov, M. Yu. Antipin, Faraday Discussions, 2007, 135, 203; (e) K. A. Lyssenko, M. Yu. Antipin, Izv. Akad. Nauk. Ser. Khim., 2006, 1 [Russ. Chem. Bull., Int. Ed., 2006, 55, 1]; (f) S. S. Bukalov, L. A. Leites, K. A. Lyssenko, R. R. Aysin, A. A. Korlyukov, J. V. Zubavichus, K. Yu. Chernichenko, E. S. Balenkova, V. G. Nenajdenko, M. Yu. Antipin, J. Phys. Chem. A., 2008, 112 (43), 10949.
S. N. Ngauv, R. Sabbah, M. Laffitte, Thermochim. Acta, 1977, 20, 371.
G. M. Sheldrick, SHELXTL-97, Version 5.10, Bruker AXS Inc., Madison, WI-53719, USA.
A. Volkov, P. Macchi, L. J. Farrugia,. C. Gatti, P. Mallinson, T. Richter, T. Koritsanszky, XD2006 — A Computer Program Package for Multipole Refinement, Topological Analysis of Charge Densities and Evaluation of Intermolecular Energies from Experimental and Theoretical Structure Factors, 2006.
D. A. Kirzhnits, Yu. E. Lozovik, G. V. Shpatakovskaya, Usp. Fiz. Nauk, 1975, 117, 3 [Physics Uspekhi, 1975, 117].
A. Stash, V. Tsirelson, WinXPRO, A program for Calculation of the Crystal and Molecular properties Using the Model Electron Density, 2001; (b) A. Stash, V. G. Tsirelson, Acta Cryst., 2002, 35, 371.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, J. A. Montgomery, J., T. Vreven, K. N. Kudin, J. C. Burant, J. M. Millam, S. S. Iyengar, J. Tomasi, V. Barone, B. Mennucci, M. Cossi, G. Scalmani, N. Rega, G. A. Petersson, H. Nakatsuji, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, M. Klene, X. Li, J. E. Knox, H. P. Hratchian, J. B. Cross, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, P. Y. Ayala, K. Morokuma, G. A. Voth, P. Salvador, J. J. Dannenberg, V. G. Zakrzewski, S. Dapprich, A. D. Daniels, M. C. Strain, O. Farkas, D. K. Malick, A.D. Rabuck, K. Raghavachari, J. B. Foresman, J. V. Ortiz, Q. Cui, A. G. Baboul, S. Clifford, J. Cioslowski, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. L. Martin, D. J. Fox, T. Keith, M. A. A1-Laham, C. Y. Peng, A. Nanayakkara, M. Challacombe, P. M. W. Gill, B. Johnson, W. Chen, M. W. Wong, C. Gonzalez, and J. A. Pople, Gaussian03, Rev.B.02, Gaussian, Inc., Pittsburgh PA, 2003.
A. Keith, AIMAll (Version 08.01.25), 2008, http://aim.tkgristmill.com.
B. M. Grafov, E. A. Ukshe, Elektrokhimicheskie tsepi peremennogo toka [Electrochemical Alternating Current Circuits], Nauka, Moscow, 127 pp. (in Russian).
V. H. Rodrigues, J. A. Paixaäo, M. M. R. R. Costa, A. Matos Beja, Acta Cryst., 2002, C58, 658.
E. A. Sudbeck, W. B. Gleason, M. C. Etter, J. Chem. Cryst., 1995, 25, 555.
R. L. Papoular, Y. Vekhter, P. Coppens, Acta Cryst., 1996, A52, 397; (b) R. Destro, P. Roversim, M. Barzaghi, R. E. Marsh, J. Phys. Chem., 2000, A104, 1047; (c) R. Flaig, T. Koritsanszky, B. Dittrich, A. Wagner, P. Luger, J. Am. Chem. Soc., 2002, 124, 3407; (d) J. Netzel, A. Hofmann, S. van Smaalen, Cryst. Eng. Comm., 2008, 10, 335.
K. Fumino, A. Wulf, R. Ludwig, Angew. Chem. Int. Ed., 2008, 47, 3830.
Yu. V. Nelyubina, M. Yu. Antipin, K. A. Lyssenko, J. Phys. Chem. A., 2009.
M. A. Spackman, Chem. Rev., 1992, 92, 1769.
A. Gavezzotti, J. Phys. Chem. B, 2002, 106, 4145.
J. Chocholousova, J. Vacek, F. Huisken, O. Werhahn, P. Hobza, J. Phys. Chem. A, 2002, 106, 1540.
E. Kassab, J. Langlet, E. Evleth, Y. Akacem, J. Mol. Structure (Theochem), 2000, 531, 267.
J. S. Gaffney, R. C. Pierce, L. Friedman, J. Am. Chem. Soc., 1977, 99, 4293.
K. A. Lyssenko, M. Yu. Antipin, Izv. Akad. Nauk. Ser. Khim., 2004, 11 [Russ. Chem. Bull., Int. Ed., 2004, 53, 10].
P. I. Dem’yanov, R. M. Gschwind, Organometallics, 2006, 25, 5709.
V. Tsirelson, A. Stash, Chem. Phys. Lett., 2002, 351, 142.
(a) K. A. Lyssenko, G. V. Grintselev-Knyazev, M. Yu. Antipin, Mendeleev Commun., 2002, 12, 128; (b) K. A. Lyssenko, M. Yu. Antipin, M. E. Gurskii, Yu. N. Bubnov, A. L. Karionova, R. Boese, Chem. Phys. Lett., 2004, 384, 40; (c) K. A. Lyssenko, S. M. Aldoshin, M. Yu. Antipin, Mendeleev Commun., 2004, 14, 98.
(a) J. A. Dabado, H. Martinez-Garcia, J. M. Molina, M. R. Sundeberg, J. Am. Chem. Soc., 1998, 120, 8461; (b) D. B. Chesnut, A. Savin, J. Amer. Chem. Soc., 1999, 121, 2335.
R. F. W. Bader, Atoms in Molecules. A Quantum Theory, Clarendon Press, Oxford, 1990.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Academician A. I. Konovalov on his 75th birthday.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 1, pp. 31—40, January, 2009.
Rights and permissions
About this article
Cite this article
Lyssenko, K.A., Barzilovich, P.Y., Nelyubina, Y.V. et al. Charge transfer and hydrogen bond energy in glycinium salts. Russ Chem Bull 58, 31–40 (2009). https://doi.org/10.1007/s11172-009-0005-7
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-009-0005-7