Abstract
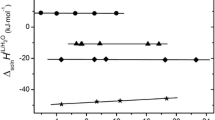
The tetra-alkyl-ammonium halides are electrolytes that have special characteristics due to the presence of hydrocarbon chains. They have been used asmodel compounds to study hydrophobic interactions due to their fairly large solubility in water and because of the possibility of change in the length and geometry of the alkyl chains. In this work the solution enthalpies of tetra-n-butyl-ammonium bromide, tetra-sec-butyl ammonium bromide, tetra-iso-butyl-ammonium bromide and di-n-butyl-di-sec-butyl ammonium bromide were determined at 298.15 K at concentrations between 0.001 and 0.01 mol kg−1. The experimental method used was isoperibol calorimetry. The results were extrapolated fitting the experimental data by least squares to obtain the solution enthalpies at infinite dilution. The hydrocarbon chains studied were so chosen because the tetra-n-butyl-ammonium ion is considered to be a structure maker for water. The results show that the geometry of the ion and of the hydrocarbon chains play an important role in the behavior properties of the solutions.
Similar content being viewed by others
References
L. H. Blanco, A. Gómez and G. Bermúdez, Acta. Sud. Quím., 1 (1981) 107.
E. Amado and L. H. Blanco, Phys. Chem. Liq., 38 (2000) 451.
L. H. Blanco and E. F. Vargas, J. Sol. Chem., 35 (2006) 21.
H. S. Franks and M.W. Evans, J. Chem. Phys., 13 (1945) 507.
H. S. Franks and W. Y. Wen, Discuss. Faraday Soc., 24 (1957) 133.
W. Y. Wen, Water and Aqueous solutions, R. A. Horne, Ed., Wiley-Interscience, New York 1972, p. 613.
R. L. Kay and D. F. Evans, J. Phys. Chem., 70 (1966) 2325.
R. M. Diamond, J. Phys. Chem., 67 (1963) 2513.
E. F. Vargas, J. C. Moreno, J. Forero and D. F. Parra, J. Therm. Anal. Cal., 91 (2008) 659.
S. J. Lindenbaum, J. Phys. Chem., 70 (1965) 814.
V. P. Korolev, N. L. Smirnova and A. V. Kustov, Thermochim. Acta, 427 (2005) 43.
H. T. Clarke, H. B. Gillespie and S. Z. Weishaus, J. Am. Chem. Soc., 55 (1933) 4571.
A. I. Vogel, Text-book of Practical Organic Chemistry Including Qualitative Organic Analysis, Logman Green and Co., London 1963.
A. K. R. Unni, L. Elias and H. I. Schift, J. Phys. Chem., 67 (1963) 1216.
B. Yff, P. G. Royall, M. B. Brown and G. P. Martin, Int. J. Pharm., 269 (2004) 361.
A. Weinsbergen, Ed., Physical Methods of Chemistry, Vol. 1, Part 4, 4th Ed., Wiley Interscience, New York 1972.
I. Wadso and R. N. Goldberg, Pure Appl. Chem., 73 (2001) 1625.
S. R. Gunn, J. Phys. Chem., 69 (1965) 2902.
C. de Visser and G. Somsen, J. Phys. Chem., 78 (1974) 1719.
C. V. Krishnan and H. L. Friedman, J. Phys. Chem., 74 (1970) 3900.
C. Shin, I. Worsley and C. M. Criss, J. Solution. Chem., 5 (1976) 867.
O. N. Bhatnagar and C. M. Criss, J. Phys. Chem., 73 (1969) 174.
E. M. Arnett and D. R. McKelvey, J. Am. Chem. Soc., 88 (1966) 2598.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Blanco, L.H., Salamanca, Y.P. & Vargas, E.F. Heats of solution in water of salts of the type R4NBr and R2R′2NBr. J Therm Anal Calorim 92, 683–686 (2008). https://doi.org/10.1007/s10973-008-9014-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-008-9014-2